The Effects of Synbiotic Supplementation on Blood Pressure and Other Maternal Outcomes in Pregnant Mothers with Mild Preeclampsia: A Triple-Blinded Randomized Controlled Trial
Received Date: September 06, 2025 Accepted Date: September 20, 2025 Published Date: September 25, 2025
doi:10.17303/ejmrc.2025.7.104
Citation: Rouhina Movaghar, Shamci Abbasalizadeh, Shabnam Vazifekhah, Azizeh Farshbaf-Khalili, Mahnaz Shahnazi (2025) The Effects of Synbiotic Supplementation on Blood Pressure and Other Maternal Outcomes in Pregnant Mothers with Mild Preeclampsia: A Triple.Blinded Randomized Controlled Trial Eur J Med Res Clin Trials 7: 1-16
Abstract
Introduction: Preeclampsia affects 2-8% of all pregnancies and is the main cause of premature childbirth. Probiotics can influence inflammatory factors and oxidative stress, which seem to be involved in the development of preeclampsia, "Ihe aim of this study was to determine the effects of synbiotic supplementation in comparison with placebo on systolic & diastolic blood pressure and pregnancy duration, as primary outcomes. Also, secondary outcomes included proteinuria, serum creatinine level, the incidence of severe PE, the use ofantihypertensive drugs, the rate of natural delivery, incidence of serious complications, platelet count, and serum levels of liver enzymes (ALT and AST), bilirubin, and LDH.
Materials and Methods: This study was a randomized, controlled, phase III, triple-blinded clinical trial conducted on 128 pregnant women with mild PE and a gestational age ofover 24 weeks referred to the high-risk pregnancy clinic of the A1-Zahra Hospital of Tabriz, Iran. The participants were randomly assigned to the intervention and control groups, and those in the intervention group received one oral synbiotic capsule (the concentration of 109 CFU) daily until delivery. The participants of the control group received placebo during the same pericni. Based on gestational age at the time ofdiagnosis, PE was catego-rized as early (< 34 weeks) or late (234 weeks). Data were obtained using appropriate questionnaires, and serum markers were measured by biochemical methods, Finally, SPSS software version 23 was used for statistical analyses. The independent t-test, Chi-square test, trend Chi-square, and Fisher's exæt test were used to compare baseline variables between the study groups. In addition, ANCOVA and Logistic regression adjusted for confounders were employed to compare outcomes the groups at post-intervention.
Results: Regarding socio-demographic characteristics, there were no statistically significant differences between the study groups except for the history of taking vitamin D3. After the intervention, the means of systolic blockl pressure (adjusted mean difference: -13.54, 95% CI: -5.01 to -22.07) and diastolic blood pressure (adjusted mean difference: -10.30, 95% CI: 4.70 to -15.90) were significantly lower in the synbiotic-supplemented group than in the placebo group. Compared to the placebo group, the incidence of severe PE (p< 0.001 ), proteinuria and mean serum creatinine level sig. nificantly declined in the synbiotic-supplemented group after the intervention.
Conclusion: Based on our results, synbiotic supplementation had beneficial effects on some pregnancy outcomes, including hypertension, incidence of severe PE, proteinuria, and serum creatinine level. It is required to conduct more studies with larger sample sizes to investigate the effects of higher doses and longer intervention periods to confirm the potential benefits of synbiotic supplementation in high-risk pregnancies.
Keywords: Probiotic; Synbiotic; Preeclampsia; Pregnancy hypertension; Pregnancy outcomes
Introduction
Preeclampsia (PE) is characterized by hypertension (systolic blood pressure 140 mmHg or diastolic blood pressure 290 mmHg) and proteinuria during pregnancy and may associated with renal failure, thrombocytopenia, hepatic dysfunction, and pulmonary edema, This condition occurs in around 3-8% of pregnancies [1,2] and exerts short-term and long-term adverse effects on mothers and fetuses [3]. PE is the main reason for admission to the prenatal and postpartum care unit due to high-risk pregnancies and the main cause of iatrogenic premature birth, intrauterine growth restriction. and intrauterine fetal death [4,5].
Although low doses of aspirin [6] and some supple. ments such as vitamin E, C have been suggested to prevent PE In high-risk mothers, mainly those with a history of PE [7], there is no reliable and immediate preventive and therapeutic measure for PE. Currently, the definitive treatment includes giving birth and removing the placenta as soon as possible [8]- For PE that occurs early during pregnancy, specialists often have an obliga- tion to terminate pregnancy to prevent maternal morbidity, but this may come at the cost of severe neonatal morbidities such as disabilities, cerebral palsy, intracranial hemorrhage, premature retinopathy, chronic pulmonary disease, and death, especially in those born earlier than week 33rd [9]
The exact underlying mechanism Of PE is still unknown. It has been hypothesized that an increase in the adipose tissue, which is a rich source Of pro-inflammatory cytokines, can trigger a systemic inflammatory reaction, leading to imbalanced growth and placental angiogenesis, and, finally, PE [10]. Endothelial dysfunction and oxidative stress are among important factors contributing to PE pathogenesis via promoting the systemic overproduction of proinflammatory mediators [11,12].
In patients with PE, intestinal microbiota shows detect. able changes from the second trimester toward the third. It has been demonstrated that the dysbiosis (the lack of interaction) of intestinal microbiota during the third trimester of pregnan- cy nurtures inflammation in PE patients, and this inflammatory axis can link intestinal microbiota to PE development [13]. In the animal models of hypertension, unbalanced intestinal microbio- ta has been suggested to play a causative role in the development ofPE [3].
Probiotics and prebiotics are the main parts of synbiotic supplements [14]. The former consists of alive microorganisms that can have beneficial health effects for the host when they are consumed in sufficient amounts [15], Prebiotics refer to indigestible food carbohydrates that facilitate the growth and activity of probiotics [16]. Probiotics, via eradicating pathogenic bacteria and modulating pathophysiological processes involved in inflammation, can improve the health of the digestive system,enhance kidney function, adjust blood pressure, prevent diabe tes, and upgrade overall health in humans [17], Probiotics have been reported to promote their anti-inflammatory effects by modulating the expression of the genes involved in inflammation and blood pressure regulation 1181, reducing the expression of lipopolysaccharide (LPS) on gram-negative bacteria [19,20], and suppressing inflammatory processes in human placental tropho- blast cells [20.21].
Clinical data show that probiotics can be a potential therapeutic option for inflammatory conditions, including PE. Studies on this topic in pregnant women are infrequent and have mostly addressed the relationship between probiotics and pregnancy outcomes [22] or retrospectively evaluated the preventive and protective effects of these supplements on gestational hyper. tension and PE [23]. According to our literature review, no clinical trial has been conducted on the potential therapeutic benefits of probiotics or synbiotics in patients with PE. So, this study was designed to investigate the applicability of oral synbiotic supplementation for treating mild PE and preventing its complications. This is of utmost importance as timely management of this cone dition can improve perinatal, including maternal and fetal, outcomes.
Methods
Study Design and SettingThis was a randomized, controlled, triple-blinded, phase III clinical trial approved under the ethics code of IR.TBZMEDREC. 1398.556 by the Tabriz University of Medical Sciences and registered at the Iranian Registry for Clinical Trials (IRCT20110606006709N20).
Probiotic supplements have been reported to be safe and have no adverse maternal or fetal outcomes [24,25]. In this study, the research population included pregnant mothers with mild PE at a gestational age of 24 weeks or more referred to the high-risk pregnancy clinic of Al-Zahra Hospital (a referral center covering a region with a relatively high incidence of PE), Tabriz, Iran.
OutcomesPrimary outcomes included systolic and diastolic blood pressure and the duration of pregnancy. Secondary outcomes included the incidence of severe PE, proteinuria, serum creatinine level, the use of antihypertensive medications, natural delivery rate, serious complications of PE (cerebral infarction, renal failure, liver failure, HELLP syndrome, disseminated intravascular coagulation, and pulmonary edema), platelet count, and serum levels of liver enzymes (ALT, AST), bilirubin, and LDH.
Inclusion criteria comprised singleton pregnancy, gestational age of 24 weeks or higher, diagnosis of mild PE, and stable maternal and neonatal conditions allowing for waiting management according to the discretion of obstetricians and gynecologists. Exclusion criteria were as follows: diagnosis of cardiovascular diseases, renal or liver failure, chronic and severe hypertension, allergy to probiotics, taking antibiotics in the past two weeks, acute gastrointestinal problems, the use of glucocorticoids and immunosuppressants (except for the cases for whom corticosteroids were prescribed to accelerate fetal lung maturation), and the occurrence of maternal or fetal adverse outcomes (related or unrelated to PE) requiring immediate delivery.
Sample SizeUsing G*POWER (version 3.1.2) software and considering a study power of 80%, α= 0.05, and two-tailed testing, the sample size was determined as n= 39 per group based on gestational age at the time of delivery (m1 =262.5, m2= 267.5, sd1=sd2= 7.35), as n= 34 based on systolic blood pressure (m1 =164, m2 =147.6, sd1=sd2= 25), as n= 21 based on diastolic blood pressure (m1 =107, m2 =96.3, sd1=sd2=12) [26], and as n= 64 based on the duration from the time of PE diagnosis to delivery (m1 =8.3, m2 =10.3, sd1=sd2= 3.95) [27]. Finally, regarding a 10% drop-out, the sample size was considered n= 128 (per group n= 64).
Sample Recruitment and Clinical ProceduresEligible pregnant women were included in the study using the available sampling method, After assessment for eligibility criteria, the subjects were adequately explained about the objectives, protocols, disadvantages, and advantages of entering the study After obtaining written informed consent, a basic demographic information form was completed for each participant by the researcher. One synbiotic capsule (LactoCare, cont. 109 CFU, Zist Takhmir Co.) containing high amounts of probiotics (lactobacilli, bifidobacterial, and streptococci) along with fruc-to-oligosaccharide prebiotics (to support the growth and activity of probiotics) was daily prescribed for the participants of the intervention group.
According to the national protocol, routine management for mild PE includes admission to the hospital, close monitoring of the mother and the fetus, and decision.making based on the gestational age. Primary care includes the administration of antihypertensive drugs such as methyl-dopa, screening the severity of the disease and the signs and symptoms of exacerbation (e.g., headache, visual impairment, epigastric pain, and sudden weight gain of about 1.5 kg or more per week), measuring the height of the uterus, determining the gestational age, daily weighing and resting, prescribing a high-protein high-calorie diet, and measuring blood pressure at the seated position every four hours. Urine protein was measured upon admission, and in the case of random proteinuria (+1 or more pronounced) or a protein to creatinine ratio of 0.3 or more, 24-hour urine same ples were collected. If proteinuria was detected in 24-hour urine samples, no subsequent assessments were performed, and serum creatinine level was regarded as sufficient for monitoring renal function. Cell blocxl counting, including platelet count, was performed, and serum levels of creatinine. liver enzymes (ALT, AST), bilirubin, and LDH were measured. These tests were repeated twice or thrice weekly depending on the condition of the mother and the severity of hypertension.
Fetal health assessment included daily hearing of heart sounds, monitoring fetal movements, an initial ultrasound and then once every three weeks to check fetal growth, and fetal health monitoring using the biophysical profile (i.e., AFI and NST). The frequency of performing these tests depended on gestational age, the severity of PE, the severity of intrauterine growth impairment, amniotic fluid volume, and fetal vascular changes in Doppler ultrasound. If gestational age was less than 37 weeks, these parameters were checked regularly, and primary care was performed until delivery. If gestational age was 37 weeks or higher, pregnancy was terminated.
During routine care, patients whose blood pressure and proteinuria were under control were discharged and monitored in an outpatient basis according to a specialist's discretion. These women, depending on their conditions, visited the clinic once or twice a week to check their blood pressure and other parameters. During these visits, the participants delivered empty envelopes and received the next package containing either synbiotic capsules or placebo. This task continued until delivery. During the study, a few mothers were discharged from the hospital.
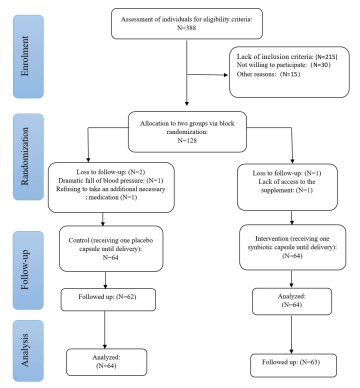
Randomization and BlindingThe participants were randomly assigned to either synbiotic supplementation or placebo group using Random Allocation Software (RAS) and the block randomization method (blocksins of four and six with the allocation ratio of 1:1). Envelopes were prepared in the same number of subjects, and each envelope was assigned with a number from 1 to 128. Then the envelopes, which had the same shape, were sealed. Someone who was not involved in the research generated a random sequence of numtkrs and allocated them to the envelopes. Each envelope contained 14 capsules of either synbiotic (109 CFU) or placebo. The first envelope was given to the first eligible person, and this process continued until the sample size was met. The participants were advised to consume one capsule daily until the day of delivery The control group received placebo capsules manufactured by the same company and in the same packaging, shape, color, and smell. The researcher, patient, and data analyst were blinded to the allocations. During group assignments, stratification was performed based on gestational age at the time of PE diagnosis (i.e., early or late PE) (Figure 1).
Follow-upDuring the treatment period, the patients were followed up either in at the hospital or via phone calls to monitor consuming the capsules and their possible side effects, All participants were instructed to avoid consuming products containing probiotics until delivery A few patients whose blood pressure was under control were discharged from the hospital during the study. These participants were provided with an adequate number of the capsules and were advised to visit either the clinic or a nearby pital for daily monitoring ofblood pressure. Patients were referred to the clinic in the case of elevation of blood pressure. Blood pressure was also analyzed on the day of delivery. A phone number was provided to these patients after discharge so that they could contact the researcher if they had any question or problem.
Data Collection ToolsThese tools included a checklist for assessing eligibility criteria, a demographic information questionnaire, a checklist for documenting the daily consumption of medications and recording their side effects, a questionnaire for recording pregnancy, delivery, and neonatal-related information, a form for gather. ing the results of laboratory tests, and a data sheet for recording blood pressure, The validity of these tools was approved using the content validity method based on the opinions of 10 faculty members.
Statistical Analysis
The data were entered into SPSS (version 23) software. The normality of quantitative variables in each group and sub. group was analyzæd using the Kolmogorov-Smirnov test. The data were described using frequency, percentage, and mean (standard deviation). The independent t-test, Chi-square, trend Chi-square, and Fisher's exact test were used to compare demographic variables between the study groups, After adjustment for baseline values and vitamin D consumption (as a confounding variable), ANCOVA was employed to compare the means of quantitative variables between the study groups. Logistic regression was used to compare variables with binary (categorical) out. comes between the groups, adjusted for confounding variables. For all tests, the a level was considered 0.05, and confidence in. terval as 95%. All calculations were performed based on the approach of intention to treat analysis (ITT). Randomization was supposed to largely omit the effects of confounding variables on the study outcomes.
Results
ParticipantsParticipants were enrolled in the study from February 2020 to January 2021 (Figure 1). Out of 128 eligible women with mild PE, two patients (one in the intervention group and one in the control group) discontinued receiving the supplement. Also, another participant in the intervention group withdrew due to hypotension. There was no loss to follow-up, and since data analysis was based on the ITT approach, the data of all 128 mothers were analyzed at the end of the intervention period. Other participants (97.65%) consumed all the capsules provided to them during the intervention period.
Participants’ Baseline CharacteristicsThe mean (standard deviation) age of the participants was 28.9 (4.8) years in the synbiotic.supplemented group and 27.9 (4.5) years in the control group. The means (SDs) of ges- tational age at the time of entering the study were 208.68 (27.8) and 21339 (223) days in the synbiotic and placebo groups, re. spectively. The average systolic blood pressure was 134,07 (9.08) mmHg in the synbiotic group and 134.67 (8.81) mmHg in the control group. Also, the means of diastolic blood pressure were 83.12 (7.37) and 83.34 (5.10) in the synbiotic and control groups, respectively. A significant intergroup difference was observed regarding the consumption of vitamin D3 during pregnancy (p< 0.001). There was no significant difference between the two groups in terms of other socio-demographic features (Table 1)
Primary OutcomesIhe mean (standard deviation: SD) of systolic blood pressure after the intervention (the day of delivery) was 138.15 (25.85) mmHg in the synbiotic group and 153.64 (22.86) mmHg in the control group, showing a significantly lower value in the former group (P— 0.002, adjusted mean difference (aMD)= -13.54, 95% CI: -5.01 to -22.07) (Table 2).
The mean (SD) of diastolic blood pressure after the intervention (the day of delivery) was 83.23 (18.12) mmHg in the synbiotic group and 95.01 (12.45) mmHg in the control group, showing a significantly lower value in the former group (P< 0.001. aMD- -10.30, -4.70 to -15.90) (Table 2).
Secondary OutcomesThe frequency of progression to severe PE was significantly (about five times) lower in the synbiotic-supplemented group than in the placebo group (P< adjusted odds ratio 5.01, 2.04-12.29). Other outcomes such as premature rupture of membranes, type of delivery, serious complications of PE, and the use of antihypertensive drugs had no significant intergroup differences at post-intervention (Table 2).
Regarding the serum levels of PE-related indicators after the intervention, the mean (SD) of serum creatinine level (mg/dL) was 0.86 (0.01) in the placebo group and 0.79 (0.01) in the synbiotic group, showing a statistically significant difference (P- 0.005, aMD- -0.06, Cl- -0.11 to -0.02). Moreover, the means of random proteinuria (mg) after the intervention was significantly lower in the synbiotic group compared to the place bo group (P- aMD- -0.47, Cl- -0.92 to 0.01) (Table 3). In other outcomes such as the length of pregnancy, premature rupture of membranes, type of delivery, serious complications caused by the disease, use of antihypertensive drugs, as well as blood factors such as platelet count (PLT) and serum levels of lactate dehydrogenase (LDH), alanine aminotransferase (ALT), and aspartate aminotransferase (AST) revealed no statistically significant differences between the study groups after the intervention (Table 3).
The mothers' adherence to use the supplements during the intervention period was satisfactory (compliance rates of 97% and 96% in the intervention and control groups, respectively). None of the participants of the intervention or placebo group reported noticeable side effects.
Discussion
In this study, synbiotic capsules (count: 109 CFU) were used as a daily treatment for pregnant women with mild PE from the time of entering the study until delivery. According to our literature review, this was the first study investigating the effects of synbiotic (containing the probiotic strains of Lactobacillus case, L acidophilus, L. rhamnosus, L. bulgaricus, Bifidobacteri- um breve, B. longum, Streptococcus thermophiles, and prebiotic plus FructcAigoSaccharides (FOS) supplements) on PE-related maternal outcomes and blood indicators.
In general, in this study, favorable results were obtained from the management of PE. In the present study, primary out. comes included systolic and diastolic blood pressure and length of pregnancy. The synbiotic supplement had a significant pos. itive effect on systolic and diastolic blood pressure, but it did not affect the duration of pregnancy Among the secondary out. comes, the occurrence of severe PE, proteinuria, and creatinine significantly improved, but other secondary outcomes such as premature rupture of membranes, type of delivery, serious com. plications caused by the disease, use of antihypertensive drugs, as well as blood factors such as Platelet, lactate dehydrogenase, alanine aminotransferase, and aspartate aminotransferase were unaffected. The use of these supplements during pregnancy has been confirmed without any side effects in mothers and children [28].
Intestinal dysbiosis can be a causative factor for hypertension. Probiotics may restore the balance of intestinal microbiota and increase the production of the metabolites involved in blood pressure regulation, suggesting them as safe and reliable treatments for improving maternal outcomes in pregnant women with PE [29,301. However, the exact mechanisms of action of probiotics are still largely unknown. Meanwhile, probiotic yo- gurt has been noted as a promising dietary supplement during pregnancy [31].
Several mechanisms have been suggested to be involved in the blood pressure lowering effects of probiotics, such as reducing systemic inflammation, [11,12] and oxidative stress 1321, stabilizing the renin-angiotensin system and, subsequently, blood pressure 1331, lowering total cholesterol and low-density lipoprotein (LDL) 130341, decreasing blood sugar. and modulating insulin resistance [35]. Some hypotheses highlight a role for neuroinflammation, which has been noted to play an important role in the pathogenesis of hypertension in humans and animal models. Alterations in intestinal microbiota affect brain homeo- stasis and neuroinflammation through the microbiota-intes- tine-brain axis. The relative frequency of numerous short-chain fatty acid (SCFA)-producing bacteria has been reported to de- cline in animal models of hypertension. Intestinal bacteria can ferment fibers, leading to the production of SCFAs [36], which subsequently can modulate blood pressure via either directly promoting vasodilation or inducing the plasminogen activator inhibitor.l (PAL 1) [37]. In hypertensive patients, dietary calci- um absorption suppresses calcium-induced renin and extracel- lular calcium uptake, thereby reducing BP [38]. Probiotics in. crease dietary calcium absorption in the intestine via SCFAs and lactic acid, which lower intestinal pH and increase the solubility and absorption of calcium ions [39],
New treatment options for hypertension in the form of probiotics and prebiotics have been known to be useful [40]. In several studies, the consumption of these dietary fibers has been associated with the reduction of cardiovascular diseases and blood pressure [41,42]. Gomez-Arango et al. (2016), in a study on overweight and obese women at week 16th of pregnancy, re. ported that the frequency of butyrate.producing bacteria in the gut microbiome inversely correlated with systolic and diastolic blood pressure and PAL-I inflammatory marker [43]. Butyrate is produced from dietary fibers by the bacteria present in intestinal lumen. Dietary supplements containing probiotics and prebi- otics (synbiotics) may change the composition of intestinal mi. crobiome, which can offer a novel way to help maintain normal blood pressure and mitigate inflammation during pregnancy, improving maternal and neonatal outcomes [44].
Ample pieces of evidence, mostly based on studies on animal models of hypertension, have confirmed a link between hypertension and intestinal microbiota. For example, Ganesh et al. (2018) showed that intestinal dysbiosis played a causative role in the development of hypertension in mouse models of obstructive sleep apnea (OSA), while probiotics and prebiotics could prevent OSA-induced hypertension; however, this effect was not observed in mice with normal blood pressure (36). In a systematic review, Ejtahed et al. (2020), who analyzed five me. ta-analysis studies including 2703 males and females in the age range of 12-75 years, reported that probiotic foods and supple- ments (3 to 24 weeks, comprising multiple species, doses above 1011 CFU) were effective in controlling blood pressure in adults with hypertension (BP2130/85 mmHg). These beneficial effects on pressure could be related to the additive or synergistic effects of several high-dose probiotic species [45]. Also, Tanidaetal. reported that the long.term consumption of probiotics (L Gasseri plus L Fermentum or L Coryniformis) reduced endo- thelial dysfunction, oxidative stress, and vascular inflammation in mice [46].
In a recent study, Hajifaraji et al. (2017) assessed the effects of probiotic capsules (containing L. acidophilus LA-5, Bifidobacterium BB- 12, S. thermophilus STY-31, and L delbrueckii bulgaricus LBY.27) at the dose of > 4x109 CUF on systolic and diastolic blood pressure in pregnant women suffering from gestational diabetes mellitus (GDM). Ihe results of the recent study showed that the probiotic supplement prevented blood pressure elevation during pregnancy, but this effectiveness was only evident after six to eight weeks of consumption, suggesting that the beneficial effects of probiotics may be achievable upon long term use. Accordingly, the consumption of this supplement for eight weeks reduced systolic blood pressure up to 8.7 mmHg and diastolic blood pressure up to 10.61 mm Hg [22]. Likewise, in an. other study by Nabhani et al. (2018), a synbiotic supplement was able to reduce systolic pressure by 9.7 mmHg and diastolic blood pressure by 4.8 mmHg [47]. The variabilities observed in blood pressure changes in different studies can be related to the doses of the supplements, duration of consumption, and the populations studied. Although the results of the mentioned studies align with the results of our research, none of them have been performed as a treatment for PE.
Unlike previous studies, in a systematic study, probiotic supplementation did not show an effect on pregnancy outcomes such as blood pressure in pregnant mothers with gestational diabetes 1481. In line with that, in the results of another study, the effect of probiotics on pregnancy outcomes including blood pressure in women with GDM was not shown to be significant [49]. Several factors may contribute to the conflicting results. A key difference between our study and other studies was that the underlying disease in our participants differed from the diseases assessed in similar reports. Our participants suffered from mild PE; however, in the above-mentioned studies, the participants were pregnant women with GDM. Another factor that may ex. plain the differences observed between these results can be dif. ferent types of the probiotics used, as well as variable durations of consumption. It seems that probiotic-containing supplements and foods can have better health effects when they are used in the long-term, which can be due to the gradual corrective effects of probiotics on intestinal microbiota,
Evidence shows that excessive inflammatory responses may play a key role in PE development [50]. Oxidative stress-induced endothelial dysfunction and systemic inflammation are also important determinants of PE [ 12,511. Increased oxidative stress during pregnancy has been reported to be asqxiated with several adverse outcomes, including PE [32], low birth weight [52], preterm delivery [53], and thrombcxytopenia [54]. Probiotics can improve PE by reducing systemic inflammation [55,56] , and oxidative stress [57]. In the present study, probiotics were able to improve kidney factors such as creatinine, proteinuria, and blood pressure in mothers with mild PE. Since these factors are considered as important diagnostic and management factors in the development of PE. it can be said that severe PE was prevented with this mechanism.
In the prospective cohort study of Nordqvist et al. (2018), the timing of probiotic milk consumption during pregnancy and its effects on the incidence of PE and preterm birth were investigated. Consumption of probiotic milk in late pregnancy significantly reduced the risk of PE [231. Also, in another cohort study by Brantsæter et al. (2011), a protective relationship between the consumption of milk products containing probiotics and PE, especially severe PE, has been shown due to inflammatory changes, this relationship is more pronounced in severe PE that the results indicated dose-dependent protection [58].
Although the biological explanation for the association of these dietary components with PE is still unclear, it may be related to the modification of immunological, oxidative and inflammatory responses related to pregnancy [59].
In the systematic review study by Lindsay et al. (2013), the results showed that the use of probiotics in pregnancy can significantly reduce fasting glucose, the incidence of GDM, PE, and Cu-reactive protein levels [191. Contrary to the mentioned studies, in the clinical trial of Lindsay et al. (2014), which investigated the effect of probiotics in obese mothers, there was no significant difference between the studied groups in terms of the incidence of PE [601. Changes in gut microbiota composition based on weight status have also been reported among pregnant women 161,621. Contradictory differences are probably related to confounding factors, such as obesity, that affect gut microbiota [63] and could explain the differences in results between studies.
We can say that the main difference between the previous studies and our study is that our study was conducted as a controlled clinical trial for the treatment of PE in humans, and as a result, its effect on the outcomes related to PE was investigated. but in previous studies. pregnancy outcomes have been investigated following the preventive intervention or in mothers with GDM or obesity or on animals. In relation to the secondary primary outcome of this study (length of pregnancy), no study was found in which the consumption Of probiotics significantly increases the length of pregnancy, and these results have also been shown in recent systematic review studies 148,641, Many reasons that are related to PE and high blood pressure can reduce the duration of pregnancy, which may be that the consumption of these supplements in a limited time was not enough and could not have a positive and significant effect on the duration of pregnancy. As mentioned, the lack of significant impact on other outcomes of the current study and the existence of contradictions between the outcomes of this study and other studies can be due to the conditions of the participants. In our study, the participants are pregnant mothers suffering from PE which is an acute and serious condition of the disease, who did not have enough time to take supplements for a long time. While all participants in other studies were people without PE- And this can be a good reason for the use of these supplements as a preven- tive measure, especially in high-risk people, in order to reduce the complications of this disorder,
The strengths of this study include the use of the most powerful randomized clinical trial method, the necessary training about the study, emphasis on voluntary participation in the study, providing sufficient opportunity for decision-making, consultation with a personal gynecologist and spouse. Also, the participants were followed up by phone to minimize sample loss.
The sample recruitment center, the A1-Zahra Hospital of Tabriz, is a teaching hospital with a high referral rate for high-risk pregnancies from different geographical regions and social groups at different ages and with various socioeconomic status. In this center, PE is managed based on national protocols. All these factors can increase the generalizability of our results.
One of the limitations of the present study was the nature of the disease and its management, limiting the opportunity for adequate consumption of supplements by some participants. Since the supplement was consumed from the time of PE diag. nosis until delivery, and considering that PE (especially late PE) is a stormy condition and can quickly escalate and prompt delivery, some participants (e.g., mothers with late-onset PE) could not take enough supplements. On the other hand, the effects of probiotics reach the ideal state gradually 1221, which can the reason for the lack of significant effects on some pregnancy outcomes, It is recommended to assess the effects of this intervention on mothers with early mild PE in future studies.
Conclusion
According to the results of this study, synbiotic supple. mentation could improve PE indicators, such as systolic and diastolic blood pressure, proteinuria, and serum creatinine level, and prevent the development of severe PE. Therefore, this strategy seems to be beneficial in the management of PE by improving maternal and neonatal outcomes. Despite the significant effect of synbiotic on some pregnancy outcomes, such as systolic and diastolic blood pressure, incidence of severe preeclampsia and other outcomes, these supplements did not show an effect on the duration of pregnancy, More studies with larger sample sizes and adequate duration of supplementation are required to confirm the protective effects of synbiotics against the adverse pregnancy outcomes of PE.
Acknowledgments
Hereby, we express our gratitude to the personnel of AL-Zahra Hospital of Tabriz who assisted us in conducting this research. We thank all the participants and Tabriz University of Medical Sciences for financially supporting this study, as well as the authorities of the School of Nursing and Midwifery of the university and all colleagues who helped us.
Ethical Approval and Participant Consent
Written informed consent was obtained from participants, and the study protocol was approved by the Ethics Committee of Tabriz University of Medical Sciences (ethics code: IR.TBZMED.REC.1398.556).
Availability of Data and Materials
The datasets used and/or analyzed in the current study are available from the corresponding author on reasonable request.
Conflict of Interest
The authors declare no conflict of interest.
Funding
This study was financed by Tabriz University of Medical Sciences. The funder provided proper arrangements in place to initiate the study and monitored study progress, but had no role in conducting the study and did not contribute to manuscript preparation. The study protocol was peer-reviewed at the funding institute. Contact information: +98 4134796770
Access to Data and Results
The research group will convey the results to participants, healthcare and public health professional, and other relevant groups through publication.
- Leeman L, Fontaine P (2008) Hypertensive disorders of pregnancy. American family physician 78: 93-100.
- Duley L (2009) editor The global impact of pre-eclampsia and eclampsia, Seminars in perinatology Elsevier.
- Adnan S, Nelson JW, Ajami NJ, Venna VR, Petrosino JF et al. (2017) Alterations in the gut microbiota can elicit hyperten- Sion in rats. Physiological genomics 49:96-104.
- Bilano VL, Ota E, Ganchimeg T, Mori R, Souza JP (2014) Risk factors of pre-eclampsia/eclampsia and its adverse outcomes in low-and middle-income countries: a WHO secondary analysis. PloS one 9: e91198.
- Kuchake VG, Kolhe SG, Dighore PN, Patil S (2010) Maternal and neonatal outcomes in preeclampsia syndrome. International Journal of Pharmaceutical Sciences and Research 1: 74.
- Atallah A, Lecarpentier E, Gofflnet F, Doret-Dion M, Gaucherand P et al (2017) Aspirin for prevention of preeclampsia. Drugs 77:1819-31.
- Cardoso PM, Surve S (2016) The effect of vitamin E and vitamin C on the prevention of preeclampsia and newborn outcome; a case—control study, The Journal of Obstetrics and Gynecology of India 66: 271-8.
- Bezerra Maia e Holanda Moura S, Marques Lopes L, Murthi P, da Silva Costa F (2012) Prevention of preeclampsia. Journal of pregnancy.
- Cluver CA, Walker sp, Mol GB, Hall DR et al. (2015) Double blind, randomised, placebo.controlled trial to evaluate the efficacy of esomeprazole to treat early onset pre-ec. lampsia (PIE Trial): a study protocol- BMJ open 5:
- Olson KN, Redman LM, Sones JL (2019) Obesity "complements" preeclampsia. Physiological genomics 51 : 73-6.
- Morken NH, Vogel I, Kallen K, Skjærven R, Lang- hoff-Roos J et al. (2008) Reference population for international comparisons and time trend surveillance of preterm delivery proportions in three countries. BMC women's health 8: 1-8.
- Goswami D, Tannetta D, Magee L, Fuchisawa A, Redman C et al. (2006) Excess syncytiotrophoblast microparticle shedding is a feature of early-onset pre-eclampsia, but not normotensive intrauterine growth restriction. Placenta 27: 56-61.
- Wang J, Shi ZH, Yang J, Wei Y, Wang XY et al. (2020) Gut microbiota dysbiosis in preeclampsia patients in the second and third trimesters. Chinese medical journal 133: 1057.
- De Vrese M, Schrezenmeir (2008) Probiotics, prebiotics, and synbiotics. Food biotechnology 1-66.
- Behnsen J, Deriu E, Sassone-Corsi M, Raffatellu M (2013) Probiotics: properties, examples, and specific applications. Cold Spring Harbor perspectives in medicine 3: ao010074.
- Fuentes Zaragoza E, Sánchez Zapata E, Sendra E, Sayas E, Navarro C et al. (2011) Resistant starch as prebiotic: A review. Starch Stärke 63: 406-15.
- Lye HS, Kuan cy, Ewe JA, Fung WY, Liong MT (2009) The improvement Of hypertension by probiotics: effects on cholesterol, diabetes, renin, and phytoestrogens. International journal of molecular sciences 10:3755-75.
- van Baarlen P, Troost F, van der Meer C, Hooiveld G, Boekschoten M et al. (2011) Human mucosal in vivo transcriptome responses to three lactobacilli indicate how probiotics may modulate human cellular pathways. Proceedings of the National Academy of Sciences 108: 4562-9.
- Lindsay KL, Walsh CA, Brennan L McAuliffe FM (2013) Probiotics in pregnancy and maternal outcomes: a systematic review. The Journal of Maternal-Fetal & Neonatal Medicine 26:772-8.
- Yeganegi M, Watson CS, Martins A, Kim SO, Re id G et al. (2009) Effect of Lactobacillus rhamnosus GR-I supernatant and fetal sex on lipopolysaccharide-induced cytokine and prostaglandin-regulating enzymes in human placental trophoblast cells: implications for treatment of bacterial vaginosis and prevention of preterm labor. American journal of obstetrics and gynecology 2(M): 532. el-e8.
- Bloise E, Torricelli M, Novembri R, Borges L, Carrarelli P et al. (2010) Heat.killed Lactobacillus rhamnosus GG modulates urocortin and cytokine release in primary trophoblast cells. Placenta 31:867-72.
- Hajifaraji M, Jahanjou F, Abbasalizadeh F, Aghamo. hammadzadeh N, MESGARI AM (2017) Effect of probiotic supplementation on blood pressure of females with gestational diatktes mellitus: a randomized double blind controlled clinical trial.
- Elias J, Bozzo P, Einarson A (2011) Are probiotics safe for use during pregnancy and lactation? Canadian Family Physi- cian 57:299-301.
- Lee J, Han J, Choi J, Ahn H, Lee S et ale (2012) Pregnancy outcome after exposure to the probiotic Lactobacillus in early pregnancy. Journal of Obstetrics and Gynecology 32:227-9.
- Chappll LC, PT, Briley AL, Kelly FJ, Lee R et ( 1999) Effect Of antioxidants on the occurrence Of pre-eclampsia in women at increased risk: a randomised trial. Ihe Lancet 354: 810-6.
- Cluver CA, Hannan NJ, van Papendorp E, Hiscock R, Beard S et al. (2018) Esomeprazole to treat women with preterm preeclampsia: a randomized placebo controlled trial. American journal of obstetrics and gynecology. 219:388. el -e 17.
- Luoto R, Laitinen K, Nermes M, Isolauri E (2010) impact of maternal probiotic-supplemented dietary counselling on pregnancy outcome and prenatal and postnatal growth: a double.blind, placebo-controlled study. British journal of nutrition 103: 1792-9.
- Broomfield H, Harris M, Goldie J (2022) Could Probiotic Supplements Be an Effective Intervention to Reduce Hy- pertension? A Systematic Literature Review. Online Journal of Complementary & Alternative Medicine, 7: 1-17.
- He J, Zhang F, Han Y (2017) Effect of probiotics on lipid profiles and blood pressure in patients with type 2 diabetes: a meta-analysis of RCTs. Medicine 96.
- He A, Chin J, Lomiguen CM (2020) Benefits Of probiotic yogurt consumption on maternal health and pregnancy out comes: a systematic review. Cureus 12.
- Walsh SW (2009) Plasma from preeclamptic women stimulates transendothelial migration of neutrophils. Reproductive sciences 16: 320-5.
- Ong L, Shah NP (2008) Release and identification of angiotensin-converting enzyme-inhibitory peptides as influenced by ripening temperatures and probiotic adjuncts in Cheddar cheeses. LWT-Fockå .%ience and Technology. 41: 1555-66.
- Ong Shah NP Release and identification of angiotensin-converting enzyme-inhibitory peptides as influenced by ripening temperatures and probiotic adjuncts in Cheddar cheeses. LWT-Food Science and Technology. 41: 1555-66.
- Guo Z, LiuX, ZhangQ, Shen Z, Tian F et al. (2011) Influence of consumption Of probiotics on the plasma lipid profile: a meta-analysis Of randomised controlled trials. Nutrition, Metabolism and Cardiovascular Diseases 21:844-50.
- Tabuchi M, Ozaki M, Tamura A, Yamada N, Ishida T et al. (2003) Antidiabetic effect of Lactobacillus GG in streptozotocin-induced diabetic rats. Bioscience, biotechnology, and biochemistry 67: 1421-44
- Ganesh BP, Nelson JW, Eskew JR, Ganesan A, Ajami NJ et al. (2018) Prebiotics, probiotics, and acetate supplementation prevent hypertension in a model of obstructive sleep apnea. Hypertension 72: 1141-50.
- Mortensen F, Jorgensen B, Christiansen H, Sloth-Nielsen J, Wolff B et al. (2000) Short-chain fatty acid enemas stimulate plasminogen activator inhibitor-I after abdominal aortic graft surgery: a double-blinded, placebo-controlled study, Thrombosis research 98: 361-6.
- Resnick LM (1999) The role Of dietary calcium in hypertension: a hierarchal overview. American Journal of Hyper- tension 12: 12.
- Parvaneh K, Jamaluddin R, Karimi G, Erfani R (2014) Effect Of probiotics supplementation on bone mineral content and bone mass density. The Scientific World Journal.
- Muralitharan RR, Jama HA, Xie L, Peh A (2020) Microbial peer pressure: the role of the gut microbiota in hypertension and its complications. Hypertension 76:1674-87.
- Whelton SP, Hyre AD, Pedersen B, Yi Y, Whelton PK et al. (2005) Effect of dietary fiber intake on blood pressure: a meta-analysis of randomized, controlled clinical trials. LWW.
- Wang X, Ouyang Y, Liu J, Zhu M, Zhao G et al. (2014) Fruit and vegetable consumption and mortality from all causes, cardiovascular disease, and cancer: systematic review and dose-response meta-analysis of prospective cohort studies. Bmj 349.
- Gomez-Arango LF, Barrett HL, McIntyre HD, Callaway LK, Morrison M (2016) Increased systolic and diastolic blood pressure is associated with altered gut microbiota composition and butyrate production in early pregnancy. Hypertension 68: 974-81.
- Vital M, Howe AC, Tiedje JM (2014) Revealing the bacterial butyrate synthesis pathways by analyzing (meta) genomic data. MBio 5: e00889-14.
- Ejtahed HS, Ardeshirlarijani E, Tabatabaei.Malazy O, Hoseini-Tavassol Z Hasani-Ranjbar S et al. (2020) Effect of probiotic foods and supplements on blood pressure: a systematic review of meta.analyses studies of controlled trials. Journal of Diabetes & Metabolic Disorders 19:617-23.
- Nabhani Z, Hezaveh SJG, Razmpoosh E, Asghari-Ja-farabadi M, Gargari BP (2018) The effects of synbiotic supple. mentation on insulin resistance/sensitivity, lipid profile and total antioxidant capacity in women with gestational diabetes mellitus: a randomized double blind placebo controlled clinical trial. diabetes research and clinical practice 138: 149-57.Tanida M, Yamano T, Maeda K, Okumura N, Fukushima Y (2005) Effects of intraduodenal injection of Lactobacillus johnsonii Lal on renal sympathetic nerve activity and blood pressure in urethane-anesthetized rats. Neuroscience letters 389: 109-14.
- Movaghar R, Farshbaf.Khalili A, MirzaRezaei ME, Shahnazi M (2022) The Effect of Probiotics or Synbiotics on the Hypertensive Disorders of Pregnant Women with Gestational Diabetes: A Systematic Review and Meta.analysis. Journal of Caring Sciences 11:94.
- Callaway LK, McIntyre HO, Barrett Foxcroft K, Tremellen A et al. (2019) Probiotics for the prevention of gesta tional diabetes mellitus in overweight and obese women: findings from the SPRING double-blind randomized controlled trial. Diabetes Care 42:364-71.
- Dusse LM, Rios DR, Pinheiro MB, Cooper AJ, Lwaleed BA (2011) Preeclampsia: relationship between coagulation, f1-brinolysis and inflammation, Clinica Chimica Acta 412: 17-21.
- Morken NH, Vogel I, Kallen K, Skjærven R, Lang hoff.Roos J et ale Reference population for international comparisons and time trend surveillance of preterm delivery proportions in three countries. BMC women's health 8:16.
- Gupta P, Narang M, Banerjee B, Basu S Oxidative stress in term small for gestational age neonates born to undernourished mothers: a case control study. BMC pediatrics 4:14.
- Baydas G, Karatas F, Gursu MF, Bozkurt HA, Ilhan N et al. (2002) Antioxidant vitamin levels in term and preterm infants and their relation to maternal vitamin status. Archives of medical research 33: 276-80.
- El Sayed AA ( 2017) Preeclamp sia: A review of the pathogenesis and possible management strategies based on its pathophysiological derangements. Taiwanese Journal of Obstetrics and Gynecology 56:593-8.
- El Sayed AA (2017) Preeclampsia: A review of the pathogenesis and possible management strategies based on its pathophysiological derangements. Taiwanese Journal of Obstetrics and Gynecology 56: 593-8.
- Richards JL. Yap YA, McLeod KH, Mackay CR, Marifio E (2016) Dietary and the gut microbiota: an alternative approach to control inflammatory and autoimmune diseases. Clinical & translational immunology 5: e82.
- Patel B, Kumar P, Banerjee R, Basu M, Pal A et al. (2016) Lactobacillus acidophilus attenuates Aeromonas hydrophila in. duced cytotoxicity in catla thymus macrophages by nundulating oxidative stress and inflammation. Molecular immunology 75: 69-83.
- Kullisaar T, Songisepp E, Mikelsaar M, Zilmer K, Vihalemm T et ale Antioxidative probiotic fermented goats' milk decreases oxidative stress-mediated atherogenicity in human subjects, British Journal of Nutrition 90:449-56,
- Brantsæter AL, Myhre R, Haugen M, MykingS, V et al. (2011) Intake of probiotic food and risk of preeclampsia in primiparous women: The Norwegian Mother and Child cohort Study. American journal of epidemiology 174:807-15.
- North C, Venter C, Jerling J The effects ofdietary fibre on C.reactive protein, an inflammation marker predicting cardiovascular disease. European journal of clinical nutrition 63: 921-33.
- Lindsay KI.. Kennelly M, Culliton M, Smith T, Maguire OC et al. (2014) Probiotics in obese pregnancy do not reduce maternal fasting glucose: a double-blind, placebo-controlled, randomized trial ( Probiotics in Pregnancy Study). The American journal ofclinical nutrition 99: 1432-9.
- Collado MC, Isolauri E, Laitinen K, Salminen S Distinct composition of gut microbiota during pregnancy in overweight and normaLweight women. The American journal of clinical nutrition 88:894-9.
- Santacruz A, Collado MC, Garcia-Valdes L, Segura M, Martin-Lagos J et al. (2010) Gut microbiota composition is associated with body weight, weight gain and biochemical parameters in pregnant women. British Journal of Nutrition 104: 83-92.
- Greiner T, Båckhed F (2011) Effects of the gut microbiota on obesity and glucose homeostasis. Trends in Endocrinology & Metabolism 22: 117-23.
- Zhang J, Ma S, Wu S, Guo C, Long S etal (2019) Effects of probiotic supplement in pregnant women with gestational diabetes mellitus: a systematic review and meta.analysis of ran. domized controlled trials. Journal of diatktes research.

Tables at a glance
Figures at a glance