Anti-Reverse Transcriptase (P66) and Integrase (P31) Antibodies as Markers to Differentiate Recent and Early Chronic HIV Infection
Received Date: November 02, 2025 Accepted Date: November 26, 2025 Published Date: November 29, 2025
doi:10.17303/jaid.2025.12.105
Citation: AliReza Khabiria, Kayhan Azadmaneshb (2025) Anti-Reverse Transcriptase (P66) and Integrase (P31) Antibodies as Markers to Differentiate Recent and Early Chronic Hiv Infection. J HIV AIDS Infect Dis 12: 1-10
Abstract:
Accurate staging of acute and early HIV infection is critical for patient management and public health. Fiebig stage VI is heterogeneous, and no clear endpoint distinguishes recent from early chronic infection. We evaluated antibodies against two HIV-1 Pol proteins integrase (P31) and reverse transcriptase (P66) using high-sensitivity indirect and sandwich ELISA assays in 385 HIV-positive serum samples. Anti-P31 antibodies were detected in 383 samples (99.4%) and anti-P66 in 368 samples (96%), with early or low-avidity responses identified only by sandwich ELISA. Based on these results, Fiebig stage VI was subdivided into VI-a (P31-positive only) and VI-b (P31- and P66-positive), allowing improved discrimination between recent and early chronic infection. This refined staging approach may enhance early diagnosis, guide treatment decisions, and improve epidemiologic monitoring of HIV transmission. The failure to diagnose acute HIV infection represents an important public health problem. The current stratification of acute and early HIV infection is based on a staging algorithm of Fiebig and colleagues. Information is limited on sensitive immunoassay testing would allow further characterization of samples that groups on Fiebig stage VI for better characterization stage VI as representing recent versus early chronic of HIV infection, we evaluated antibodies to two major HIV-1 Pol products, P31 and P66, by high sensitive sandwich ELISA method in 385 serum of individuals infected with HIV. we demonstrate the utility of the ELISA as a sensitive method would allow improved stages for Fiebig V–VI stages and define seven stages for representing recent versus early chronic of HIV infection. So that final stage VI divided into two subgroups as stage VI-a and VI-b that in stage VI-a, ELISA positive for one of the Pol antigens (p31) and in stage VI-b full WB reactivity including a p31 band and ELISA positive for Pol (P31, P66) antigens. Regarding the result and considering that most of three main coding regions of HIV genome, gag, pol and env proteins are immunogenic, It would be define seven stages for representing recent versus early chronic of HIV infection.
Keywords: Recent HIV Infection, Early Chronic HIV Infection, Anti-Integrase (P31) Antibody, Anti-Reverse Transcriptase (P66) Antibody, Sandwich ELISA
Introduction
Acute HIV infection is typically defined as the time from virus entry to completion of seroconversion. Early-stage HIV infection is less well defined but generally refers to the interval between seroconversion and the establishment of the virus load (VL) set point, which usually occurs 6–12 months after infection. The magnitude of the VL set point is prognostic for disease progression [1]. Early recognition and diagnosis of acute HIV infection can provide important benefits on the individual level, because the potential for early initiation of treatment may allow for preserved immune system control of the virus, and on the public health level, because the risk of transmission may be decreased. The decision to initiate therapy must involve careful consideration of potential risks and benefits on a case-by-case basis [2, 3]. The failure to diagnose acute HIV infection represents an important public health problem. Persons with primary infection may be up to 10 times more likely to transmit HIV per sex act than are individuals with established infection [4] and secondary transmission from recently infected persons likely contributes to a significant proportion of overall HIV transmission [5-7]. The term “primary HIV infection” refers to the overall time period from HIV exposure to establishment of the VL set point. Based on the outcome of serological testing for HIV, primary HIV has been divided into two distinct phases, namely acute HIV infection and early HIV infection. Acute HIV infection is defined as the time from initial exposure to the HIV virus, to the first detection of anti-HIV antibodies, which typically occurs three to four weeks later. Early HIV infection refers to the time period immediately after the appearance of anti-HIV antibodies, until the VL set-point is achieved, which typically occurs approximately five to six months post-infection [8-11].
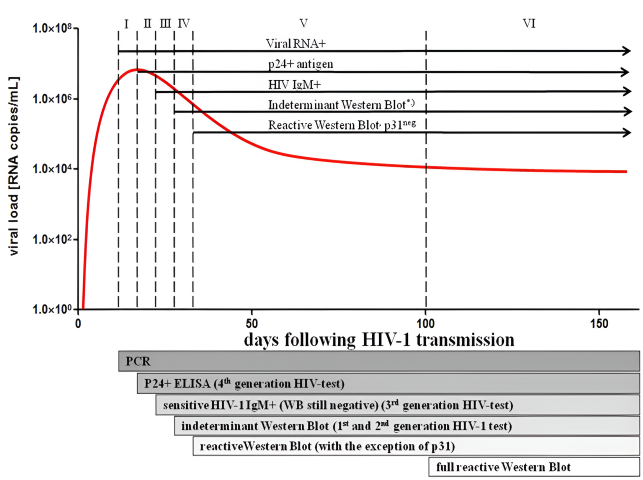
Antibody reactivity against the different virus antigens tested is crucial for staging acute and early infections. The current stratification of acute and early HIV infection is based on a staging algorithm published by Fiebig and colleagues in 2003 [12] [Figure.1]. Acute and early infection was classified with stage I: HIV RNA positive only; stage II: RNA and p24 antigen tests positive but antibody EIA non-reactive and WB negative; stage III/IV: RNA, p24 antigen and Abs positive but WB negative (III) or indeterminate (IV); stage V: reactive WB pattern, with no p31 (integrase) reactivity, and stage VI full WB reactivity including a p31 band. Stages I to V were considered as acute HIV infection and stage VI − as early chronic infection [13]. Fiebig stages are used to classify the progress of HIV infection, particularly in its early stages, which is relevant to cure because numerous studies strongly suggest that eradication is easier the earlier it is undertaken. The projected duration of each of stages I–IV is relatively brief, lasting on average only 3–5 days, whereas stage V (positive Western blot without p31 band) is estimated to last 69.5 days on average [14]. Patients can be categorized into Fiebig stages I–VI, which are based on a sequential gain in positive HIV clinical diagnostic assays (viral RNA measured by PCR, p24 and p31 viral antigens measured by ELISA , HIV specific antibody detected by ELISA and HIV specific antibodies detected by western blot). Patients progress from acute infection through to the early chronic stage of infection at the end of Fiebig stage V, approximately 100 days following infection, as the plasma VL begins to plateau [15]. Fiebig et al [13] explained that the incorporation of sensitive/less sensitive EIA testing would allow further characterization of stage VI samples as representing recent versus early chronic infection and no endpoint was defined for the final stage VI, characterized by a full Western blot pattern. Other study also shown that infection occurred within versus beyond approximately 6 months from antibody seroconversion by an IgM sensitive EIA [16].
In terms of diagnostic tests, HIV infection is identified through assays that target viral antigens, such as p24, p17, and p31, as well as the envelope glycoproteins gp120 and gp41. Although strong antibody responses are generated to the gp120 and gp41 envelope glycoproteins and the core/matrix proteins p24 and p17, encoded by the env and gag genes [17], Notably, the p31 and p66 proteins, which are encoded by the HIV pol gene, have been shown to be highly reactive in serum samples from HIV-positive individuals, with p66 showing a higher seropositivity rate compared to other antigens, such as gp41 [18, 19]. These proteins may be particularly useful for distinguishing between different stages of infection. Recent studies have suggested that the pol antibodies (specifically against p31 and p66) may serve as important predictors for seroconversion and could be used to better characterize stage VI. While the Fiebig staging system is effective in tracking early HIV infection, further refinement is needed, particularly in distinguishing recent HIV infection from early chronic infection. Current testing methods, such as Western blot (WB) and recombinant immunoblot assay (RIBA), though accurate, have limitations in sensitivity and accuracy, particularly in the early stages of infection [19-21].
Comparing between RIBA and WB revealed that RIBA had similar specificity but lower indeterminate results to WB. Since Due to WB and/or RIBA is a test based on indirect immunoassay and recognizes the IgG class not only these tests are low sensitivity but have low accuracy [22, 23].
This study aims to evaluate the reactivity of antibodies against two HIV pol proteins (p31 and p66) using a highly sensitive sandwich ELISA method. We will compare these antibody responses in serum samples from individuals at different stages of HIV infection, with a focus on distinguishing between Fiebig stages V and VI. Our goal is to refine the characterization of stage VI by defining two subcategories (VI-a and VI-b) based on the presence of antibodies against p31 and p66, respectively. This approach may improve the diagnostic accuracy of HIV infection stages and provide a more precise endpoint for Fiebig stage VI.
Materials and Methods
Study Subjects
Three hundred eighty-five HIV seropositive and four hundred eighty-nine HIV seronegative serum samples were obtained from Iranian individuals who were referred to Behavioral Diseases Consultation Centers in Tehran and Alborz. The HIV-positive status of all seropositive individuals was confirmed using a commercial 4th generation ELISA kit and Western blot tests at a reference laboratory.
Cloning, Expression, and Purification of HIV Pol Protein
We previously described the production, purification, and immunoreactivity evaluation of recombinant HIV P31 [24-25]. For cloning, expression, and purification of the p66 reverse transcriptase/RNase H protein, a total of 22 HIV-1 reverse transcriptase (RT) coding sequences from the predominant circulating strain CRF35AD in Iran were retrieved from the Los Alamos HIV sequence database (HIV Sequence Database, Los Alamos National Laboratory). A consensus amino acid sequence was derived from these records, reverse-translated, and codon-optimized for E. coli expression.
Labeling of HIV-P31 and P66
The purified antigens (P31 and/or P66) were labeled with horseradish peroxidase (HRP) following the procedure outlined by Wilson and Nakane [26]. Commercial HRP powder (Sigma, Germany) was dissolved in 100 mM sodium acetate buffer (pH 4.4) to a final concentration of 5 mg/ml. Freshly prepared sodium periodate (NaIO4) was added to the antigen solution to a final concentration of 20 mM, and the mixture was incubated for 20 minutes. The oxidized HRP was separated from NaIO4 by dialysis against 0.1 M carbonate-bicarbonate buffer (pH 9.5). Following the addition of sodium borohydride (NaB3H4), the mixture was incubated at 4°C for 2 hours. The labeled P31 and/or P66 were separated from unreacted material by gel filtration. The final HRP-conjugated antigens were mixed with an equal volume of glycerin and stored at -20°C.
Indirect ELISA For Igg Antibodies to HIV-P31 and/or P66
The 96-well Maxisorp microtiter plates (Nunc, Roskilde, Denmark) were coated with 5 μg/ml of recombinant P31 and/or P66 in coating buffer (0.1 M carbonate-bicarbonate buffer, pH 9.6) and incubated overnight at room temperature. The plates were then washed four times with PBS containing 0.05% Tween-20 (PBS-T) and blocked with 1% BSA in PBS for 1 hour at room temperature. HIV-positive serum (1:100 dilutions) was incubated in the antigen-coated wells for 1 hour at 37°C. After washing, 100 μl of anti-human-IgG-HRP conjugate (Sigma Aldrich, St. Louis, USA) was added to each well and incubated for another hour at 37°C. After a final wash, 100 μl of TMB substrate was added, and the plate was incubated at room temperature for 15 minutes. The enzymatic reaction was stopped by the addition of 50 μl of 2 M H2SO4, and the optical density (OD) was measured at 450 nm.
Double Antigen Sandwich ELISA for Total Antibodies to HIV-P31 and/or P66
Microtiter plates were coated with recombinant P31 and/or P66 antigens for the detection of total anti-HIV antibodies. Serum samples from HIV-positive individuals were diluted 1:10, and 100 μl of the diluted serum was added to the antigen-coated wells and incubated for 1 hour at 37°C. After washing, 100 μl of HRP-conjugated P31 and/or P66, prepared in sample diluent, was added to each well and incubated for an additional 1 hour at 37°C. Plates were washed again as described above, and 100 μl of TMB substrate was added to each well and incubated for 15 minutes at room temperature. The reaction was stopped by adding 50 μl of 2 M H₂SO₄, and the optical density (OD) was measured at 450 nm.
Cut-Off Value Measure
We used arbitrary methods for cut-off value of both indirect and sandwich ELISA methods. In indirect ELISA, the microtiter plate coated with recombinant P31 and /or P66 were incubated with diluted of 489 HIV negative serum samples. The 16-well of plate was used for diluted pool serum of eight HIV seropositive as positive control. After washes, anti-human-IgG-HRP conjugate was added to each well and optical densities were measured at 450 nm as described above. In Sandwich ELISA cut-off value was measured as indirect ELISA, except the plate coated with recombinant antigen(s) were incubated with diluted of 489 HIV negative serum samples and P31-HRP and / or P66-HRP diluted were used as conjugate.
Statistical Analysis
Linear regression and analytical sensitivity were used to analyze standard curve data. Specificity, sensitivity, reproducibility, and correlation coefficients were calculated using SPSS (version 25; Inc., Chicago). The CV was calculated for between-run and within-run in Microsoft Excel (2016).
Results
Cut-off Value
To establish criteria for the identification by ELISA of sera containing antibody to P31 and/ or P66, the variation in colorimetric reactivity of serum derived from seronegative persons were assessed. For Cut-off value, serum samples from 489 seronegative, which were nonreactive by commercial 4th generation HIV ELISA Kit, were evaluated in the coated plates with P31 and / or P66 antigens in both indirect and sandwich. In indirect ELISA the average signal (O.D) for 489 seronegative non-infected sera was 0.112 with a range of 0.042–0.194 and a standard deviation of 0.032 and in sandwich method the average signal (O.D) was 0. 092 with a range of 0.032–0.124 and a standard deviation of 0.024. In both Methods the average signal (O.D) for eight HIV infected sera were greater than 3.00 that were considered equal to 3.0. We used arbitrary methods to set the cutoff value as the mean value of negative sera + 3SD. In this method Cut-off value 0.208 and 0.164 defined for Indirect and sandwich methods respectively. The average signal for each triplicate was determined and the signal to cutoff (S/Co) ratio was calculated. Samples with their S/Co ratio above one were considered as positive and those with S/Co bellow one, regarded as negative.
Screening Of HIV P31 Seropositive Sera
As shown in Table 1, 383 of the 385 samples (99.4%) had an S/Co ratio greater than 1.0 in both assays and were therefore considered positive for anti-P31 antibodies. Among the 383 anti-P31–positive sera, 355 samples showed strong reactivity with average O.D values greater than 3.00 in both ELISA formats. The remaining 28 positive samples demonstrated lower O.D values, ranging from 0.24 to 1.94 in the indirect ELISA and from 0.53 to 2.54 in the sandwich ELISA. These lower signals likely represent early or low-avidity antibody responses. Although both assays used plates coated with recombinant P31 antigen, the immunoglobulin classes detected differed between the two methods. The indirect ELISA, which uses anti-human IgG-HRP, detects only IgG antibodies. In contrast, the sandwich ELISA captures all antibody classes and subclasses, as P31 antigens coated on the plate and HRP-labeled P31 conjugate bind antibodies independently of isotype.
Screening of HIV-Positive Sera for Anti-P66 Antibodies
As shown in Table 1, 368 of the 385 HIV-positive samples (96%) were seropositive for anti-P66 antibodies. Three samples were positive only by the sandwich ELISA, and 17 samples (3.1%) were negative in both methods. Among these 17 seronegative samples, two were confirmed HIV-positive by both a commercial fourth-generation ELISA kit, yet showed no detectable antibodies to either P31 or P66. Of the P66-positive samples, 351 exhibited strong reactivity with average O.D. values greater than 2.5 in both ELISA methods. The remaining 17 samples showed discordant reactivity: 14 were positive in the indirect ELISA with O.D. values ranging from 0.36 to 1.36, while all 17 were positive in the sandwich ELISA with O.D values between 0.23 and 2.35. The three samples that were positive only by the sandwich assay likely represent very early antibody responses, detectable only when all antibody classes are captured.
Discussion
Information regarding the use of highly sensitive immunoassays for detailed characterization of Fiebig stage VI remains limited. This study sought to determine whether ELISA targeting P31 and P66 offers greater sensitivity than Western Blot (WB) for distinguishing Fiebig stages V and VI, and to clarify the role of these markers in differentiating recent from early chronic HIV infection. We also aimed to refine the endpoint of stage VI based on the first appearance of anti-P31 antibodies by WB. Historically, estimates of acute and recent HIV infection have relied on WB or RIBA banding patterns, often without confirmation using more sensitive assays. Several reports indicate that absence of the p31, p66, and p51 bands is increasingly common among individuals with suspected recent infection, supporting the need for more precise virologic markers. Comparisons between RIBA and WB show that although their specificities are similar, RIBA yields fewer indeterminate results and provides clearer information on the timing of antibody emergence. These findings suggest that P66, like P31, may serve as a useful marker for differentiating Fiebig stages, as characteristic indeterminate WB profiles appear at stage IV, progress to full Env reactivity at stage V, and finally show P31 positivity at stage VI [12]. The order in which WB bands appear is closely linked to the stage of infection, and antibodies directed against pol proteins, particularly P31 and P66, may serve as indicators of seroconversion. Prior studies have identified P31 as a potential early infection marker [28–30], and our earlier work demonstrated its utility as a diagnostic antigen [25, 26]. Consistent with this, most individuals in the present study produced antibodies against P31. Because integrase (IN/P31), protease, and reverse transcriptase (RT/P66) are co-translated products of the HIV pol gene, early immunogenicity of these proteins is biologically plausible and P66 has been reported to produce higher seropositivity rates than several gag or regulatory proteins and is comparable to env gp41 [17]. Given the known structural and functional links between RT and IN, the correlation between anti-P31 and anti-P66 responses observed here is expected. While the indirect and sandwich ELISA formats did not yield identical results, sandwich assays capture all antibody classes and subclasses, whereas indirect assays detect IgG only. In this study, 95.3% of P31-positive samples were also P66-positive, and only 3.9% lacked P66 reactivity in both assays. A small subset of samples showed discordant results, suggesting early stages of antibody maturation or the development of IgM or non-IgG subclasses against P66. Overall, the anti-P66 immunoassay proved both sensitive and specific, offering a practical tool for further characterization of Fiebig stage VI samples. When combined with P31 detection, these assays enhance differentiation between Fiebig stages V and VI and help identify the transition from recent to early chronic infection. Based on our results, we propose modified definitions for stages V–VI (Table 2).
Stages I–IV progress rapidly over 3–5 days, while stage V lasts approximately 70 days [14]. Progression from stage V through VI-a to VI-b appears to reflect sequential maturation of antibodies to P31 and P66. Because the catalytic sites of pol proteins are highly conserved across HIV subtypes, antibodies against these epitopes are likely consistent across diverse strains. This raises the possibility that pol-based assays, such as anti-P66, may help detect emerging variants not readily captured by standard antibody tests [31–34].
The combined use of anti-P31 and anti-P66 assays offers improved resolution of late acute infection, particularly where WB results are indeterminate or delayed. These findings support the refinement of Fiebig staging and highlight the clinical importance of pol-based antibody detection for more accurate identification of recent HIV infection.
- Lyles RH, Muñoz A, Yamashita TE, Bazmi H, Detels R, et al. (2000) Natural history of human immunodeficiency virus type 1 viremia after seroconversion and proximal to aids in a large cohort of homosexual men. Journal of Infection Disease 181: 872–80.
- Stekler J, Collier AC (2004) Primary HIV infection. Current HIV/AIDS Reports 1: 68-73.
- Xiridou M, Geskus R, de Wit J, Coutinho R, Kretzschmar M (2004) Primary HIV infection as source of HIV transmission within steady and casual partnerships among homosexual men. AIDS 18: 1311–20.
- Wawer MJ, Gray RH, Sewankambo NK, Serwadda D, Li X, Laeyendecker O, et al. (2005) Rates of HIV-1 transmission per coital act, by stage of HIV-1 infection, in Rakai, Uganda. Journal of Infection Disease 191: 1403–09.
- Yerly S, Vora S, Rizzardi P, Chave JP, Vernazza PL, et al. (2001) Swiss HIV Cohort Study. Acute HIV infection: impact on the spread of HIV and transmission of drug resistance. AIDS 15: 2287–92.
- Pao D, Fisher M, Hué S, Dean G, Murphy G, et al. (2005) Transmission of HIV-1 during primary infection: relationship to sexual risk and sexually transmitted infections. AIDS 19: 85–90.
- Brenner BG, Roger M, Routy JP, Moisi D, Ntemgwa M, et al. (2007) Quebec Primary HIV Infection Study Group. High rates of forward transmission events after acute/early HIV-1 infection. Journal of Infection Disease 195: 951–9.
- Jacquez JA, Koopman JS, Simon CP, et al. (1994) Role of the primary infection in epidemics of HIV infection in gay cohorts. Journal of Acquired Immune Deficiency Syndromes 7: 1169–84.
- Koopman JS, Jacquez JA, Welch GW, Simon CP, Foxman B, et al. (1997) The role of early HIV infection in the spread of HIV through populations. Journal of Acquired Immune Deficiency Syndromes and Human Retrovirology 14: 249–58.
- Mayaphi SH, Martin DJ, Quinn TC, Laeyendecker O, Olorunju SA, et al. (2016) Detection of Acute and Early HIV-1 Infections in an HIV Hyper-Endemic Area with Limited Resources. PLoS One 11: 1-14.
- Kassutto S, Rosenberg ES (2004) Primary HIV Type 1 Infection. Clinical Infectious Diseases 38: 1447–53.
- Fiebig EW, Wright DJ, Rawal BD, Garrett PE, Schumacher RT, Peddada L, Heldebrant C, et al. (2003) Dynamics of HIV viremia and antibody seroconversion in plasma donors: implications for diagnosis and staging of primary HIV infection. AIDS 17: 1871–79.
- McMichael AJ, Borrow P, Tomaras GD, Goonetilleke N, Haynes BF (2010) The immune response during acute HIV-1 infection: clues for vaccine development. Nature Reviews Immunology 10: 11–23
- Ananworanich J, Sacdalan CP, Pinyakorn S, Chomont N, Souza M, et al. (2016) Virological and immunological characteristics of HIV-infected individuals at the earliest stage of infection. Journal of Virus Eradication 2: 43-8.
- Lewis DA (2012) Detection and management of acute HIV infections in patients with sexually transmitted infections: a window of opportunity for HIV prevention within South Africa? The Southern African journal of epidemiology & infection 27: 149-55.
- Janssen RS, Satten GA, Stramer SL, Rawal BD, O'Brien TR, et al. (1998) New testing strategy to detect early HIV-1 infection for use in incidence estimates and for clinical and prevention purposes. JAMA 280: 42–8.
- Filice G, Soldini L, Orsolini P, Razzini E, Gulminetti R,et al. (1991) Sensitivity and specificity of anti-HIV ELISA employing recombinant and synthetic viral antigenic peptides. Microbiological 14: 185–94.
- Hill M, Tachedjian G, Mak J (2005) The packaging and maturation of the HIV-1 Pol proteins. Current HIV Research 3: 73–85.
- Kong W-H, Liu P, Tang L, Zhu Z-R, Xiao P, Zhan J-B, Wang X, Zhou W and Liu M-Q (2019) Estimation of the Seroconversion Duration of HIV-1 Antibodies in Individuals With Recent Infection in China. Front. Microbiol. 10:1322.), Sudha T, Lakshmi V, Teja VD. Western blot profile in HIV infection. Indian J Dermatol Venereol Leprol. 2006; 72: 357–60.
- Liu P, Tang L, Kong W-H, Zhu Z-R, Xiao P, et al. (2020) Anti-HIV-1 antibodies based confirmatory results in Wuhan, China, 2012-2018. PLoS ONE 15: e0238282,
- Liu MQ, Zhu ZR, Kong WH, Tang L, Peng JS, Wang X, et al. (2016) High rate of missed HIV infections in individuals with indeterminate or negative HIV western blots based on current HIV testing algorithm in China. Journal of medical virology. 88: 1462–6.
- Duri K, Muller F, Gumbo FZ, Kurewa NE, Rusakaniko S, et al. (2011) Human Immunodeficiency Virus (HIV) types Western blot (WB) band profiles as potential surrogate markers of HIV disease progression and predictors of vertical transmission in a cohort of infected but antiretroviral therapy naïve pregnant women in Harare, Zimbabwe. BMC Infect Dis. 11: 7.
- Tuaillon E, Sanosyan A, Pisoni A, Liscouet J, Makinson A, Perre PV (2017) Staging of recent HIV-1 infection using Geenius rapid confirmatory assay compared to INNO-LIA, New Lav and Blot 2.2 assays. Journal of clinical virology: the official publication of the Pan American Society for Clinical Virology. 95: 47–51.
- Hashinaka K, Hashida S, Nishikata I, Adachi A, Oka S, et al. (2000) Recombinant p51 as Antigen in an Immune Complex Transfer Enzyme Immunoassay of Immunoglobulin G Antibody to Human Immunodeficiency Virus Type 1. Clinical and Diagnostic Laboratory Immunology 7: 967–976.
- Rikhtegaran Tehrani Z, Azadmanesh K, Mostafavi E, Gharibzadeh S, et al. (2015) Development of an integrase-based ELISA for specific diagnosis of individuals infected with HIV. Journal of Virological Methods 215: 61-6.
- Rikhtegaran Tehrani Z, Azadmanesh K, Mostafavi E, Gharibzadeh S, et al. (2018) High avidity anti-integrase antibodies discriminate recent and non-recent HIV infection: Implications for HIV incidence assay. Journal of Virological Methods. 253: 5-10.
- Wilson MB, Nakane PK (1976) the covalent coupling of proteins to periodate-oxidized sephadex: a new approach to immunoadsorbent preparation. J Immunol Methods. 12: 171-81.
- Sudha T, Lakshmi V, Teja VD (2006) Western blot profile in HIV infection. Ind. J. Dermatol. Venereol. Leprol. 72: 357–60.
- Garland FC, Garland C F, Gorham ED, Brodine SK (1996) Western blot banding patterns of HIV rapid progressors in the U.S. Navy seropositive cohort: implications for vaccine development. Navy retroviral working group. Ann. Epidemiol. 6: 341–37.
- Linley L, Ethridge SF, Oraka E, Owen SM, Wesolowski LG, Wroblewski K, et al. (2013). Evaluation of supplemental testing with the multispot HIV-1/HIV-2 rapid test and APTIMA HIV-1 RNA qualitative assay to resolve specimens with indeterminate or negative HIV-1 Western blots. J. Clin. Virol. 58: e108–12.
- Li G, De Clercq E (2016) HIV genome-wide protein associations: a review of 30 years of research. Microbiology and Molecular Biology. 80: 679–731.
- Loveday C, Tedder RS (1993) Enzyme-linked immunosorbent assays for the measurement of human immunodeficiency virus, type 1 reverse transcriptase antigen and antibodies. Journal of Virological Methods 41: 181–92.
- Advani M, Imagawa DT, Lee MH, Sano K, Morales F, et al. (1989) Cross-sectional study of reverse transcriptase inhibiting antibody as a marker of acquired immune defoeieniy syndrome. Journal of Clinical Microbiology 27: 1453-5.
- Chatterjee R, Rinaldo CR Jr, Gupta P (1987) Immunogenicity of human immunodeficiency virus (HIV) reverse transcriptase: detection of high levels of antibodies to HIV reverse transcriptase in sera of homosexual men. Journal of Clinical Immunology 7: 218-24.

Tables at a glance
Figures at a glance