The Impact of Mesenchymal Stem Cells on the Rejection of Skin Allografts in Murine Models
Received Date: February 17, 2025 Accepted Date: March 17, 2025 Published Date: March 20, 2025
doi:10.17303/jber.2025.9.102
Citation: Alireza Khalili, Shima Kamalijoo, Atousa Nazari, Paria fazalali, Mahshid Sadeghi, et al. (2025) The Impact of Mesenchymal Stem Cells on the Rejection of Skin Allografts in Murine Models. J Biomed Eng Res 9: 1-16
Abstract
Recent advancements in protocols utilizing stem cell therapies have significantly contributed to the understanding of long-term objectives in transplantation. Over the years, considerable progress has been achieved in employing stem cells to enhance transplant success rates, mitigate the incidence and severity of graft rejection, modulate regulatory cell types, accelerate the regeneration of damaged tissues, and induce immune tolerance and immunosuppression. The application of mesenchymal stem cells (MSCs) in the field of transplantation has undoubtedly yielded positive outcomes. The primary objective of this study was to isolate and purify mesenchymal stem cells from murine models and to assess the administration of MSCs in reducing the incidence and severity of skin allograft rejection in mice. The subjects were divided into three distinct groups, each receiving a single systemic injection of MSCs at various time points relative to the allotransplantation of C57BL/6 mouse skin grafts onto Balb/C recipients. Our findings indicate that autologous MSCs can prolong the duration until skin graft rejection occurs, with optimal results observed when MSCs are administered post-transplantation. This effect is likely attributable to the migration of MSCs to the graft site, driven by local inflammatory responses already present in the recipient's body due to the transplantation procedure. Consequently, administering cells after transplantation appears to yield more favorable outcomes. However, further investigation is necessary to explore the numerous variables that may influence this hypothesis. Conversely, MSCs derived from the donor can also enhance graft survival, despite being allogeneic to the recipient's immune system and potentially activating immune responses. Thus, mesenchymal stem cells, whether syngeneic to the host or allogeneic to the donor, are effective in prolonging graft survival; however, a single dosage is insufficient to induce specific immune tolerance.
Keywords: Mesenchymal Stem Cells (MSCs), Allograft, Immune Tolerance, Systemic Injection, Allogenic Source, Autologous Cells.
Introduction
Organ transplantation is often the only viable treatment for end-stage organ failure. The primary objective of this procedure is to enhance the quality of life and survival rates for patients suffering from various critical conditions affecting the kidneys, liver, heart, and other organs. However, alongside the growing demand for transplant candidates and the limited availability of transplantable organs, a significant challenge remains: preventing the rejection of the transplanted tissue or organ. The principal barrier to a successful transplant is all immunity, which refers to the immune response of the transplant recipient against the foreign tissue. The immune system's primary function is to safeguard the body from potentially harmful substances. It identifies these substances as foreign and initiates a response to eliminate them. This immune response also occurs when the transplanted tissue or organ is introduced into the body, as the immune system recognizes the cells from a different individual as foreign and a potential threat. Consequently, the immune system's reaction to the transplant can lead to its rejection. Currently, the predominant strategy for managing immune responses and overcoming the recipient's immune barrier involves the use of immunosuppressive drugs to suppress the immune system. The use of these drugs, due to their non-specificity and low specificity, results in a general weakening of the patient's immune system and an increased risk of developing various diseases, such as infections and malignant cancers. Currently, inducing tolerance in the patient's body to foreign transplant tissue and enhancing the patient's specific tolerance to donor cells is considered a promising avenue in regenerative medicine. As we have observed, to function effectively and avoid rejection of genetically different donor tissue, organ recipients must rely on non-specific immunosuppressive methods for life. This reliance can lead to numerous complications, including increased vulnerability to infections and a heightened risk of developing diseases such as cancer, which arise from the compromised activity of the immune system. Conversely, modulating the immune system or inducing specific tolerance towards the donor, rather than suppressing it, can help protect the immune system from becoming weakened against other harmful factors. This approach allows the immune system to maintain its healthy and natural functions. Mesenchymal cells represent one of the most promising cell populations for cell-based immunotherapy in organ transplantation. These cells can modulate immune responses and prevent acute allograft rejection. Currently, immune cell therapy is one of the methods that has garnered significant attention. The question of whether a patient's own cells or specific donor cells can be utilized for immunotherapy or immune system modulation, and how this process works, remains uncertain. However, many believe that cell therapy has the potential to replace traditional drug treatments. Transplantation, specifically organ transplantation, is one of the most significant medical advancements of the last century. In general, transplantation involves the transfer of cells, tissues, or organs from one part of a living being's body or from a donor to another part of that being's body. This procedure is performed to repair damaged tissues that cannot heal spontaneously or to replace malfunctioning organs with healthy, functional ones, thereby restoring health or reducing the disability of the affected individual. The transfer of organs from one person to another to save a life or improve the quality of life is commonly referred to as organ donation. An organ transplant can be performed using the individual's own organ, a portion of an organ or tissue from another living person, or from brain-dead or heart-dead donors under specific conditions and with specialized equipment and arrangements. An organ transplant is a type of surgery in which a defective or damaged organ is removed and replaced with a new organ. Similarly, grafting is the process of excising tissue from one area and implanting it in another area where it is needed. Reason: Improved clarity, vocabulary, and technical accuracy while maintaining the original meaning.
Allograft transplantation is the most common solution to replace damaged or dysfunctional tissues to date. However, immunosuppression is one of the major issues and problems in this process. Immunosuppression, which is mostly induced by continuous doses and lifelong use of non-specific immunosuppressive steroid drugs, can lead to the risk of various diseases. On the other hand, immune cell therapy as an alternative method can reduce this risk and create immune modulation in the patient. One of the cells that has this property of modulating the immune system is mesenchymal stem cells.
The main issue in this research is to answer questions such as:
1: How are mouse mesenchymal stem cells prepared?
2: What antibodies are expressed negatively and positively on mouse mesenchymal cells?
3: How is a full-thickness skin allograft applied to a mouse?
4: What effect does the injection and application of autologous or allogeneic mesenchymal stem cells have on prolonging the time of graft rejection?
5: Can a single dose of 2 million mesenchymal stem cells alone delay or eliminate graft rejection?
The main goal of this study and research is to prevent acute skin graft rejection in patients with various defects and burns or skin loss due to various types of accidents or at least to postpone the time of graft rejection to reduce the patient's psychological stress and start the spontaneous repair of the tissue. In addition, to achieve the best time to apply cell therapy for transplantation by minimizing or eliminating immunosuppressive drugs and establishing the initial stages of a method for using mesenchymal stem cells in subsequent studies on transplantation of other organs are among its other goals.
Materials and Method
A 1-6-week-old BalbC mouse is killed by cervical dislocation. The body is then placed in 73% alcohol, so that all parts of the body, including the tail, are completely submerged in alcohol. After two minutes, the mouse is sprayed with alcohol and transferred to a hood. The body is placed on the surgical table so that the abdomen of the animal is exposed. An incision is made from the beginning of the hind legs where they connect to the lower abdomen, and the skin is opened along the legs to the ankle bone. This prevents the animal's hair from coming into contact with the internal organs, which can cause contamination. The hind leg is then separated from the abdomen and ankle and collected in a Petri dish containing PBS and 1% penicillin-streptomycin antibiotic. This procedure is repeated for the other leg. The rest of the body is removed from the hood. The animal's legs are removed one by one, and the muscles and tissues attached to them are gently separated with scissors, and as much of the bone as possible is cleaned using sterile gauze and fingertips. After the knee joint area, it is divided into two parts with scissors so that the femur and tibia bones remain intact. It is placed in a 15 mL falcon containing 5 mL PBS and 1% penicillin-streptomycin antibiotic. The falcon is completely closed, sprayed with alcohol, sealed with paraffin, and transferred to the culture room for the rest of the process. Under a culture hood that has been sterilized at 100%, the falcon door is opened and the bones are transferred from it to a Petri dish containing 73% alcohol. After one minute, the bones are removed from the alcohol and transferred to PBS and 1% penicillin-streptomycin antibiotic.
Using an insulin syringe containing approximately 1 mL of αMEM culture medium and antibiotics, the bone marrow is flushed out. To flush, the tip of the syringe is inserted into one end of the bone and its contents are emptied with pressure until the red marrow comes out the other side and pours into the falcon. To completely remove all the bone marrow, it is turned over and this is repeated from the other end until the bone is completely white. This is repeated for the other femur and tibia bones and all the red marrow of the bones is collected into the falcon. The falcon containing the red marrow is then centrifuged at 363 g for 5 minutes at room temperature. The supernatant is discarded and the cell mass at the end of the falcon is resuspended in the culture medium. That is, 1 ml of complete mesenchymal cell media, which was explained in the first section, is added to the cell mass and the entire contents are gently pipetted several times (several times it is drawn into the pipette and emptied). Then, after counting using Trypan Blue 5 staining, which will be explained later, the entire cell suspension is cultured in a 25 × 25 T flask. From each mouse, about 33-136 cells are obtained, a large number of which are blood progenitor cells and non-mesenchymal cells. After several medium changes, many of them are eliminated and only cells adhering to the plastic surface of the flask, the majority of which are mesenchymal stem cells, remain. Therefore, they are all seeded in a small flask. For culture in a 25 × 1-6 T flask First, bone marrow cells suspended in 2-5 ml of the required amount of media are pipetted into the flask and transferred to the flask. Then the Falcon is washed with the remaining media until no cells remain in the tube. Then the culture medium is added to the flask. The flask is closed and transferred from the hood into the incubator with a controlled atmosphere of the culture medium, temperature 37 oC and 1% CO2. After 3 days of the initial culture of bone marrow cells, it is time for the first change of the culture medium. After the first 72 hours, the adherent cells have settled to the bottom of the dish and adhered to it, and the suspended cells remain in the supernatant. Under the microscope, the adherent cells are fixed at the bottom of the dish, while other suspended or dead cells are moving in the medium, which can be clearly seen with a slight shake of the flask. Following sterile precautions, the flask is removed from the incubator and transferred to a sterilized culture hood. The lid is removed and the flask is placed so that all of its medium is collected in one corner. Then, using a pipettor, all of the medium is drawn from the flask into a 5-ml pipette and discarded. Again, using a separate pipette, 5 or 6 ml of fresh culture medium is added to it. The flask lid is tightly screwed on and examined briefly under the microscope. Then it is cleaned with 73% alcohol and transferred back into the incubator. From now on, this operation is performed every three to four days until the cells cover at least 73% of the bottom of the container and reach the so-called confluence 6. And the old medium is replaced with new nutrient medium.
The host mouse is anesthetized by breathing isoflurane-impregnated gas in a closed chamber. The skin surface of the neck and lumbar region is decontaminated and depilated, similar to that of the host. To expedite the surgical procedure, depilation is performed before the donor skin is harvested. Then, when the graft is removed from the donor, the host mouse is anesthetized using an intraperitoneal injection of a pre-prepared ketamine-xylazine anesthetic solution (133 mg per ten grams of body weight). The animal's legs and abdominal area are cleaned with alcohol, and after sterilizing the surgical area using Betadine surgical scrub and alcohol, the animal is transferred under a surgical hood. The animal's legs are taped to the bed using paper tape and the animal is fixed on the surgical bed in such a way that the animal's lumbar region is facing up. Bupivacaine or another suitable anesthetic solution is immediately sprayed at the surgical site. The animal's eyes are lubricated with sterile ophthalmic vitamin A or another suitable lubricant to prevent dryness of the eyes, which are open during anesthesia and can lead to blindness in the long term. At this stage, as with graft removal from the donor, in the host animal, using The surgical site is marked on the animal's back in a circle of the same size or, for better results, one millimeter smaller. This is because the animal's skin in the surgical area stretches slightly after the incision, and the surgical site becomes larger; it is recommended that the graft harvesting site from the donor be larger than that of the host. Before starting the incision, we make sure that the anesthesia is deep and the animal is ready for surgery by pinching the animal's toes. If the animal is completely unconscious, it will not react to this, otherwise it will pull its leg and it is necessary to wait a little for the animal to be completely unconscious. After ensuring that the anesthesia is deep, the animal's skin is grasped in one-fold using forceps and raised. Then, a small incision is made in the area with a blade and bupivacaine anesthetic solution is poured dropwise into the area and the marked area is cut with scissors. The anesthetic drop spray is repeated after the skin is harvested at the surgical site. After harvesting, the skin graft that was harvested from the donor is removed from the solution and carefully placed in the correct orientation. Once correctly positioned, the graft is carefully sutured to the skin of the surgical site. For best results, care should be taken to avoid as much overlap as possible between the host skin and the graft.
Because the creation of folds or the placement of the skin and graft under and on top of each other prevents proper blood supply to the graft and leads to faster tissue necrosis. Depending on the need, between six and ten interrupted single sutures are applied to the skin and graft in pairs facing each other to completely cover all interstitial seams. During suturing, to prevent the site from drying out and to reduce the host's loss of body water, normal saline is slowly sprayed dropwise into the site using a 5 ml syringe without a needle. Care should be taken that excessive use of this may cause embolism at the site. After suturing is completed, the surgical site is first covered with a small incision of Vaseline gauze, then sterile gauze is cut to the appropriate size and the animal is bandaged. Finally, the dressing is fixed in place using an adhesive bandage. The animal is removed from the hood and placed on a soft surface, such as a clean towel or gauze, in a cage until it has fully recovered from anesthesia. A forty-watt infrared lamp is placed at the end of the cage to warm the animal. This helps to improve and speed up the recovery process of the animal.
Result and Discussion
In this section, the results of this study, including the morphological and phenotypic identification of mesenchymal stem cells extracted from the bone marrow of Balb/C mice, as well as the results of skin allograft transplantation with mesenchymal stem cells injected on different days of transplantation, are presented, and their consistency or reasons for inconsistency with published studies by previous researchers are examined. Research conducted on mesenchymal stem cells has made them suitable candidates for delaying transplant rejection or possibly inducing tolerance and immune tolerance.
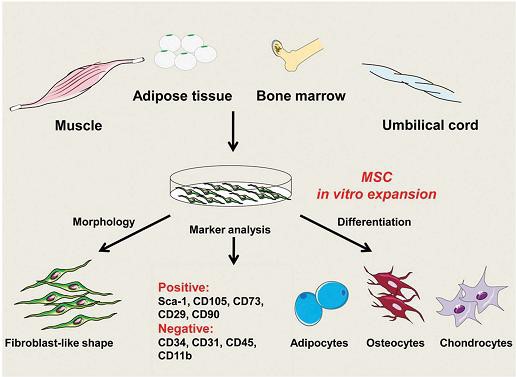
To identify mesenchymal stem cells, mesenchymal cells were harvested from six to eight-week-old Balb/c mouse bone marrow cells and their repeated passages were performed, and their morphology was examined by inverted light microscopy. Balb/c mouse mesenchymal cells showed a common small spindle-shaped fibroblast-like pattern. At higher magnification, their large single nucleus was partially visible. The spindle-like fibroblast-like morphology of mesenchymal stem cells was one of the primary features for their identification. It is common in almost all studies on them. And it is a constant feature for their identification. However, this alone cannot be sufficient to prove that the cells are mesenchymal. Therefore, the next step for identifying mesenchymal cells is the expression or lack of expression of specific markers on them.
Determining the phenotype of cells
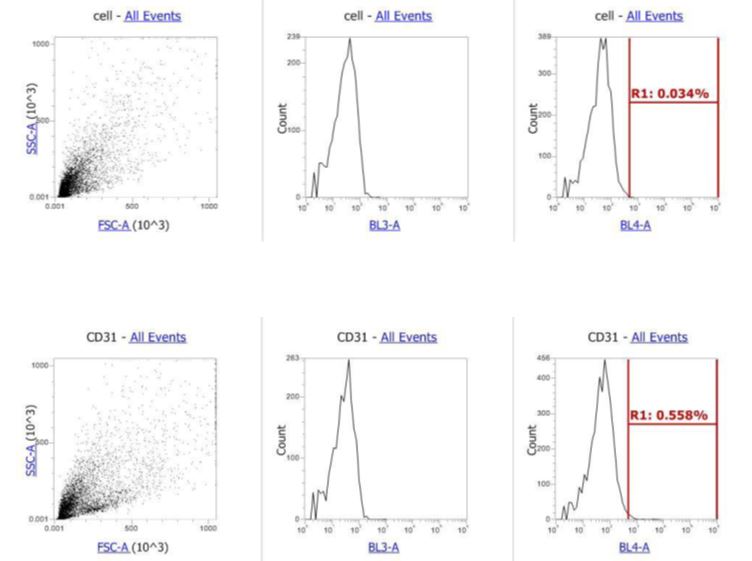
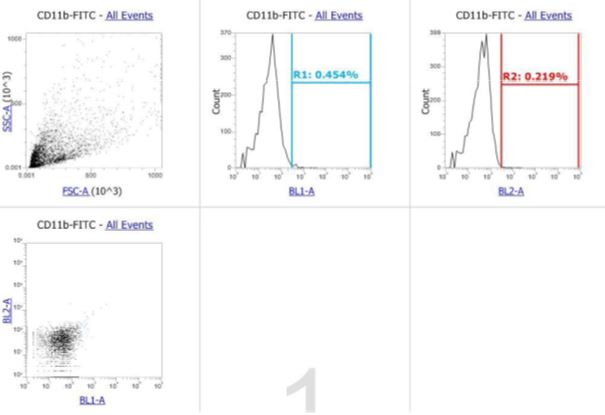
At the beginning of the work, to ensure the purity of the mesenchymal stem cells, flow cytometry was performed on specific markers. The results showed that CD105 antibody was expressed in about 32% and CD44 in about 67% of the cells, while the expression level of the anti-analyse was less than 1% (Figure 4). The results, as expected, indicated that markers -3 and -4 were not expressed in less than 3.5% of these cells (Figure 4). Monocytes and macrophages are present in this cell population and positive expression of mesenchymal markers. Based on these results, mouse Mesenchymal stem cells at passage 4 express mesenchymal markers, but the level of this expression is lower than the desired level. The expression of the CD105 marker, as mentioned earlier, was 32%. This is much lower than the normal average for mesenchymal cells. However, this level was in line with the findings of Per Anderson et al., who reported a 43% expression level of this marker in early passages of mouse mesenchymal cells, especially adipose-derived stem cells. Further in this paper, titled “Flow cytometric absence of the CD105 marker among 33 cells defines a novel subset of mesenchymal stem cells,” they claimed that this level would again decrease during in vitro culture and higher passages.
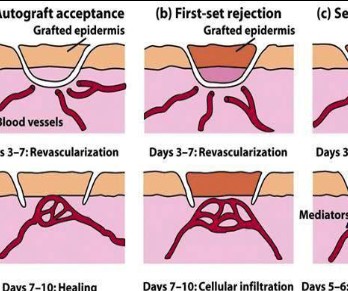
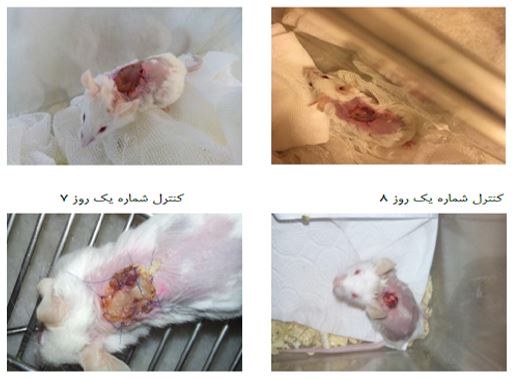
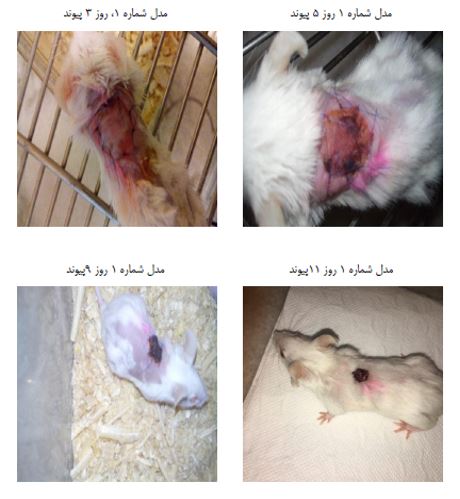
Skin allografts in mice are usually rejected after one week, and sometimes between seven and nine days. A control model consisting of Balb/c mice with allogeneic grafts from C54bl/6 was rejected with a single dose of PBS on day 7 without any signs of recovery, and was lifted off the site without any pressure on day 9.
Model number one, which included Balb/C mice with C57bl/6 skin allografts, received 233 microliters of autologous mesenchymal stem cells in 333 × 136 PBS via the tail vein two days before allograft transplantation. And from the fifth day after transplantation, the animal's skin was still attached to the site. However, signs of darkening of the allograft were visible. The complete graft trace in this model was completely visible on the ninth day, and darkening, wrinkling, and dryness were visible in the skin. The skin was completely lifted from the site and the animal's hair had grown around it, and the allograft was completely removed from the site without any pressure.
The observation that control group two received the same dose of its own cells on the day of skin allograft surgery yet took longer to completely reject the graft than model group two undermines the hypothesis that autograft cells, which are not recognized as foreign by the body and are less likely to be lysed by natural killer cells, may serve as a more effective source for modulating the immune response.
Although the results of Manjir et al. indicated that allogeneic mesenchymal stem cells (MSCs) induce a greater increase in immune-activated cells, including natural killer cells, compared to syngeneic MSCs, leading to a heightened immune response and potential host immune rejection, this study suggests that prolonged allograft survival may be more reliant on the presence of donor cells. Specifically, the model that received donor cells—namely, injected cells, syngeneic cells, and allografts—demonstrated greater survival rates compared to those with syngeneic and autologous cells in the host. This implies that donor-specific antibodies may play a crucial role in counteracting the host immune response, thereby enhancing graft survival. These findings align with previous studies that have shown that donor-specific transfusions can extend allograft survival and promote wound healing.
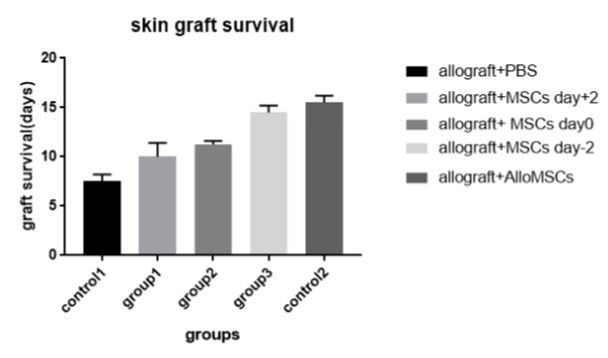
However, in a similar study by La Rocca et al. [74], despite the concordance of the results, it was suggested that autologous and host-homologous cells do not cause significant changes in the average survival time of skin grafts. The average survival of allografts with and without allogeneic cells would be 12.5 and 12 days, respectively. Although the findings of the study conducted with 15 transplants were completely rejected (Figure - Autologous mesenchymal stem cell injection had completely rejected 7 transplants on average within 11 days, the difference - 4-12) and this rate was relatively more noticeable compared to the control without receiving cells, which was relatively more noticeable on days 5. In general, injection of mesenchymal stem cells after transplantation increases graft survival. This may also be due to the difference in the strain of mice tested. With all this, mesenchymal stem cells in a single dose, at any time interval from transplantation, could not create permanent tolerance and immunity for transplant acceptance. And these cells, despite all the modulatory properties they have on immune responses, are not effective alone and in a single dose to overcome transplant rejection.
Limitations of the study, namely that no long-term graft survival assessment was performed and single-dose MSC was administered
Conclusion
Allograft transplantation is currently the only way to replace many organs. The main problem for a successful transplantation is the xenogeneity of the transplanted tissue or organ. Therefore, finding ways to induce donor-specific tolerance is one of the ultimate goals of this treatment. Mesenchymal stem cells, due to their easy harvesting and cultivation in vitro, in addition to their ability to differentiate into multiple lineages and their immune modulatory potential in vivo and in vitro, have become a subject of interest for preclinical and clinical research in a wide range of defects and diseases. In addition, the presence of these cells in most embryonic and adult tissues and the ability of these cells to differentiate into specialized differentiated cell types have made them a suitable target for bioengineering and tissue engineering applications. In this study, an attempt was made to harvest, isolate, and purify this cell population, namely mesenchymal stem cells, to induce immune tolerance and investigate their effects on the time of skin allograft rejection in laboratory mice. Mouse bone marrow stem cells from passage four onwards contain a very high percentage of mesenchymal cells, and therefore, to achieve an almost pure population of mesenchymal cells, these cells must undergo successive passages. After that, these cells were injected into the host receiving the skin allograft, and daily monitoring yielded the following results:
1. The prolonged time required for allograft rejection in models that received a single dose of mesenchymal stem cells compared to the control sample confirms that mesenchymal stem cells have immune system control capabilities.
2. In addition, the longer time to complete rejection of the allograft in the control model 2, which received allogeneic donor cells, may reflect the fact that not only are these allogeneic cells not easily recognized and eliminated by the host immune system in the body, but their presence in the host body following transplantation when they are syngeneic with the allograft donor rather than the host, may be more effective in reducing immune responses. The better quality of the skin allograft in control 2 compared to other models, especially model 2, which received simultaneous transplantation and cell infusion, again suggests that these cells of allograft donor origin will lead to better outcomes. The approximate simultaneous and even delayed graft rejection of one to two days in the model that received cells two days after transplantation, compared to the model that received autologous cells at the same time, could be due to the fact that the presence of immune and inflammatory signals and responses can help the engraftment and migration of mesenchymal stem cells to the injured site.
- Frenette PS, Pinho s, et al. (2013) Mesenchymal stem cell: keystone of the hematopoietic stem cell niche and a stepping-stone for regenerative medicine. Annual Review of Immunology, 31: 285-316.
- Lin JY, Liao SK (2014) new perspectives in tolerance induction for vascularized composite allotransplantation. Surgery: current research, 4: 196-201.
- Sarhane KA, Ibrahim Z, et al. (2012) minimization of immunosuppression and tolerance induction in recontructive transplantation. Current Surgery Reports, 1: 40-6.
- Karaoz E, B Akpinar, (2013) Stem Cells: Current Challenges and New Directions, filling the gaps in relationship between cancer and stem cells Stem Cell Biology and Regenerative Medicine, 33: 225-51.
- Wang Y, Chen x, et al. (2014) Plasticity of Messenchymal Stem Cells in immunomodulation: pathological and thrapeutic implications. Nature immunology, 15: 1009-16.
- Shi M, ZW Liu, FS Wang (2011) Immunomodulatory properties and therpeutic application of mesenchymal stem cells. Clinical and experimental Immunology, 164: 1-8.
- Qinjun Z, R Hongying, H Zhongchao (2016) Mesenchymal stem cells: Immunomodulatory capability and clinical potential in immune diseases. Journal of Cellular Immunology, 20: 3-20.
- Mundra V, IC Gerling, RI Mahato (2013) Mesenchymal Stem Cell-Based Therapy. Mol pharm, 10: 77-89.
- Ma S, Xie N, et al. (2014) Immunobiology of mesenchymal stem cells. Cell Death and Differentiation, 21: 216-25.
- Silverstein DM (2013) Inflammation after renal transplantation: Role in the development of graft dysfunction and potential therapies. Journal and Organ Dysfunction, 5: 233-41.
- Gao F, Chiu SM, et al. (2016) Mesenchymal stem cells and immunomodulation: current status and future prospectives. Cell Death and Disease, 7: E2062.
- Paul B, Valapour M, et al. (2004) Ethics of organ transplantation, in center of bioethics. university of Minnwsota's center of bio ethics: university of Minnwsota.
- www.transplant.ir/history/iran-transplantation-history, in transplantation. 2011, iranian society of organ transplantation.
- Cooper DKC (2012) A brief history of cross-species organ transplantation. proc (bayl univ med center), 25: 49-57.
- CJE W, D JH, (2012) Organ transplantation: historical perspective and current practice. British Journal of Anaesthesia, 108: 29-42.
- Powel A (2011) A transplant makes history, Joseph Murray's 1954 kidney opperation ushered in a new medical era, in harvard gazzet. harvard university news: harvard university.
- Moticka EJ. A Historical Perspective on Evidence-Based Immunology.1th eddition, Elsevier
- Zachary AA, MS Leffell (2013) Transplantation Immunology,Methods and Protocols. Methods in Molecular Biology. 1034.
- Schold JD, Srinivas TR, et al. (2011) The relative risk of overall graft loss and acute rejection among African American renal transplant recipients is attenuated with advancing age. Clinical Transplantation, 25: 721-30.
- Lin CM, RG Gill (2015) Direct and indirect allograft recognition: pathways dictating graft rejection mechanisms. Nov 14. Current Opinion in Organ Transplant.
- Halleck F, Friedersdorff F, et al. (2013) New perspectives of immunosuppression. Transplantation proceedings, 45: 1224-31.
- Willicombe M, P Brookes, E Santos-Nunez (2011) Outcome of patients with preformed donor-specific antibodies following alemtuzumab induction and tacrolimus monotherapy. American Journal of Transplant, 11: 470-7.
- Clarkson MR, S MH (2005) T-cell costimulatory pathways in allograft rejection and tolerance. . Transplantation, 80: 555-63.
- Puttarajappa C, R Shapiro, P Henkie Tan (2012) Antibody-Mediated Rejection in Kidney Transplantation: A Review. Journal of Transplantation, 2012: 9.
- Nicole M, Valenzuela, EF Reed (2013) Antibodies in Transplantation: The Effects of HLA and Non-HLA Antibody Binding and Mechanisms of Injury. Methods in Molecular Biology, 1034: e26.
- de Weerd A, Vonk A, et al. (2014) Late antibody-mediated rejection after ABO-incompatible kidney transplantation during Gram-negative sepsis. BMC Nephrology, 15: e7.
- Baldwin WM, A Valujskikh, RL Fairchild (2014) Mechanisms of antibody-mediated acute and chronic rejection of kidney allografts. Current Opinion in Organ Transplant.
- Ma J, Wang JH, et al. (1995) In vivo treatment with anti-ICAM-1 and anti-LFA-1 antibodies inhibits contact sensitization-induced migration of epidermal Langerhans cells to regional lymph nodes Cell Immunology, 158: 389-99.
- Guangwen R, Zhao X, et al. (2016) Inflammatory Cytokine-Induced Intercellular Adhesion Molecule-1 and Vascular Cell Adhesion Molecule-1 in Mesenchymal Stem Cells Are Critical for Immunosupression. journal of immunology, 184: 2321-28.
- Legendre C (2013) JAK3 inhibition: what potential for the future? Transplant nResearch, 2: 6.
- Fehrer C, G Lepperdinger (2005) Mesenchymal stem cell aging. Experimental Gerontology, 40: 926-30.
- Friedenstein AJ, J Gorskaja, N Kulagina (1976) Fibroblast precursors in normal and irradiated mouse hematopoietic organsExperimental Hematology, 4: 267.
- Friedenstein AJ, IS Piatetzky, PKV (1966) Osteogenesis in transplants of bone marrow cell J Embryol Exp Morphology, 16: 381e90.
- Martin GR (1981) Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. PNAS, 78: 7634.
- Evans MJ, MH Kaufman (1981) Establishment in culture of pluripotential cells from mouse embryos. Nature, 292: 154-6.
- Thomson JA et al. (1998) Embryonic stem cell lines derived from human blastocysts. Science, 282: 1145-7.
- Pittenger Mf, Mackay AM, et al. (1999) Multilineage potential of adult human mesenchymal stem cells. Science, 284: 143e7.
- Xu J, Fan WJ, et al. (2009) Neural ganglioside GD2 identifies a subpopulation of MSC in human umbilical cord. Cell Physiology and Biochemistery, 23: 4-6.
- Zhao Q, Ren H, et al. (2009) Differentiation of human umbilical cord mesenchymal stromal cells into low immunogenic hepatocyte-like cells Cytotherapy, 11: 414e26.
- Dominici M, Le Blanc K et al. (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statementCytotherapy, 8: 315e7.
- Labovsky V, Hofer L, et al. (2010) Cardiomyogenic differentiation of human bone marrow mesenchymal cells: Role of cardiac extract from neonatal rat cardiomyocytes. Differentiation of stem cells, 79: 93 e101.
- Zhou S, Wang J et al. (2013) Mechanical stretchupregulates SDF-1alpha in skin tissue and induces migration of circulating bone marrow-derived stem cells into the expanded skin. StemCells, 31: 2703e13.
- Shi Y, Hu G, et al. (2010) Mesenchymal stem cells: a new strategyfor immunosuppression and tissue repair. Cell Res, 20: 510-8.
- Noack S, Seiffart V, et al. (2014) Periostin secreted by mesenchymal stem cells supports tendon formation in an ectopic mouse model. Stem Cells Development, 23: 1844e57.
- Aggarwal S, MF Pittenger (2005) Human mesenchymal stem cells modulateallogeneic immune cell responses. Blood, 105: 1815e22.
- Giacca M, S Zacchigna (2012) VEGF gene therapy: therapeutic angiogenesis in the clinic andbeyond. Gene Therapy, 19: 622-9.
- Ren G, Zhao X, et al. (2010) cytokine-induced intercellular adhesion molecule-1 and vascular cell adhesion molecule-1in mesenchymal stem cells are critical for immunosuppression. Journal of Immunology, 184: 2321-8.
- Svobodova E, Krulova M, et al. (2012) The role of mouse mesenchymal stem cells in differentiation of naive T-cells into anti-inflammatory regulatory T-cell or proinflammatory helper T-cell 17 population. Stem Cells Development, 21: 901-10.
- Krampera M, Cosmi L, et al. (2006) Andreini A et al. Role for interferon-gamma in the immunomodulatory activity of human bone marrow mesenchymal stem cells Stem Cells, 24: 386-98.
- Li W, Ren G, et al. (2012) Mesenchymal stem cells: a double-edged sword in regulating immune responses Cell Death and Differentiation, 19: 1505-13.
- Strioga M, Viswanathan S, et al. (2012) Same or not the same? Comparison of adipose tissue-derived versus bone marrow-derived mesenchymal stem and stromal cells. Stem Cells Developments, 21: 2724-52.
- Leto Barone A, Khalifian S, et al. (2013) Immunomodulatory Effects of Adipose-Derived Stem Cells: Fact or Fiction? stem cell research and therapy, 2157-7633.
- Djouad F, Plence P, et al. (2009) Immunosuppressive effect of mesenchymal stem cells favors tumor growth in allogeneic animals. Blood, 102: 3837e 44.
- Wood KJ, A Bushell, ND Jones, (2011) Immunologic unresponsiveness to alloantigen in vivo: a role for regulatory T cells. Immunol Rev. 119-32, 241: 119-32.
- Yan Z, Zhuansun Y, et al. (2014) Imunomodulation of mesenchymal stromal cells on regulatory T cells and its possible mechanism. Experimental Cell Research, 324: 65e74.
- Singer N, AL Caplan (2011) Mesenchymal stem cells: mechanisms of inflammation. Annual Review of Pathology, 6: 457-78.
- Ghannam S, Pène J, et al. (2010) Mesenchymal stem cells inhibit human Th17 cell differentiation and function and induce a T regulatory cell phenotype. Journal of Immunology, 185: 302-12.
- Selmani Z, Naji A, et al. Human leukocyte antigen-G5 secretion by human mesenchymal stem cells is required to suppress T lymphocyte and natural killerfunction and to induce CD4+CD25highFOXP3+ regulatory T cells, Stem Cells, 2008. 26: p. 212e22.
- Yen BL, Yen ML, et al. (2013) Multipotent human mesenchymal stromal cells mediate expansion of myeloid-derived suppressor cells via hepatocyte growth factor/c-Met and STAT3. Stem Cell Report, 1: 139e51.
- Uccelli A, L Moretta, V Pistoia (2006) Immunoregulatory function of mesenchymal stem cells. European Journal of Immunology, 36: 2566.
- Ungerer C, Quade-Lyssy P, et al. (2014) Galectin-9 is a suppressor of T and B cells and predicts the immune modulatory potential of mesenchymal stromal cell preparations. Stem Cells Development, 23: 755e66.
- Bar-Meir E, D Mendes, E Winkler (2006) Skin Substitutes. Israel Medical Association, 8: 188-91.
- Garmel (2012) An introduction to clinical emergency medicine, S.V. Mahadevan and G. M., Editors. 2012, Cambridge University Press: Cambridge. 216-9.
- http://www.nku.edu/~dempseyd/SKIN.html
- Andreassi A, Bilenchi R, et al. (2005) Classification and pathophysiology of skin grafts. Clinics in Dermatology, 23: 332-7.
- Brown D, G Borschel, ChangIn EY (2004) Grafts MMoP Surgery, Editor. 16-21.
- Leung JJ, HB Mus, J Fish (2009) skin graft. UTMJ, 86: 61-4.
- Anjos-Afonso F, D Bonnet (2008) Isolation, Culture, and DifferentiationPotential of Mouse Marrow Stromal Cells, in Current Protocols in Stem Cell Biology. 2008, John Wiley & Sons Inc. p. 2B.3.1-2B.3.11.
- Soleimani M, S Nadri (2009) A protocol for isolation and culture of mesenchymal stem cells from mouse bone marrow Nature protocols, 4: 102-6.
- GIBCO®Mouse (C57BL/6) Mesenchymal Stem Cells GIBCO®, Editor. Revision date: 9 December 2009.
- Mundra V, Gerling IC, RI Mahato (2013) "Mesenchymal Stem Cell-Based Therapy," Mol pharm, 10: 77-89.
- Anderson P, Benabdellah K, et al. (2010) CD105 (Endoglin)-Negative Murine Mesenchymal Stromal Cells Define a New Multipotent Subpopulation with Distinct Differentiation and Immunomodulatory Capacities. Journal of Immunology, 183: 993-1004.
- Al-Qaisy A, S Alwachi, N Yaseen (2014) Isolationand Identification of mouse bone marrow derived mesenchymal stem cells. Iraqi Journal of Cancer and Medical Genetics, 7: 49-55.
- Larocca RA, Moares-Vieira PM et al. (2013) Adipose Tissue-Derived Mesenchymal Stem Cells Increase Skin Allograft Survival and Inhibit Th-17 Immune Response. PLoS ONEjournal.pone, 8: e7696.
- Chen L, Tredget EE, et al. (2009) Analys is of Allogenicity of Mesenchymal Stem Cells in Engraftment and Wound Healing in Mice. PLoS ONE. 4: e7119.
- Rostovskaya M, Anastassiadis A (2012) Differential Expression of Surface Markers in Mouse BoneMarrow Mesenchymal Stromal Cell Subpopulations withDistinct Lineage. 7: e51221.
- Mangir N, Akbal C, et al. (2014) Mesenchymal stem cell therapy in treatment of erectile dysfunction Autologous or allogeneic cell sources? the international journal of urology, 21: 1280-5.
- Markees TG, Phillips NE, et al. (1988) long term survival of skin allografts induced by donor splenocytes and anti-CD154 antibody in thymectomized mice requires CD4+ Tcells, interferon-gamma, and CTLA4. Journal of Clinical Investigation, 101: 2446-55.


Figures at a glance