Typical Presentation of Erdheim-Chester Disease: A Case Report
Received Date: May 06, 2025 Accepted Date: May 20, 2025 Published Date: May 25, 2025
doi: 10.17303/croa.2025.10.102
Citation: Rossella Loiacono (2025) Typical Presentation of Erdheim-Chester Disease: A Case Report. Case Reports: Open Access 10: 1-7
Abstract
Erdheim-Chester disease (ECD) is a rare type of non-Langerhans cell histiocytosis. The clinical presentation may range from an indolent focal disease to a life threatening organ failure. ECD may virtually affect any organ system, most commonly skeleton, kidneys, cardiovascular system, central nervous system. The typical histological findings consist in xanthogranulo- matous or xanthomatous infiltration by foamy histiocytes, with immunohistochemistry positive for CD68 and CD163 and negative for CD1 a.
We present the case of a patient with a history of central insipid and exposure who was hospitalized for dyspnoea associated with pleural effusion, with radiological findings of a typical multi-organ involvement (hairy kidneys, coated aorta, Symmetrical osteosclerosis of the middle-distal third of long bones, pericardial and pleural thickening) with the histological findings confirming the diagnosis of ECD
Keywords: Erdheim-Chester; Histiocytosis; Pleural Thickening; Foamy Histiocytes
Case Report
Erdheim-Chester disease (ECD) is a rare type of non-Langerhans cell histiocytosis. Up to now, about 1500 cases have been reported worldwide [l]. Ihe etiology of the disease is unknown. It mainly affects males between 40 and 60 years of age. It is assumed that ECD origins from a deregulation of the Thl response, given the low of IL-4 and high INF-a, IL-7 and IL-12 [2]. Recently, BRAFV600E mutation has been observed in more than a half of ECD patients, with new implications for therapeutic options [3].
The clinical presentation is variable as it may range from an indolent focal disease to a life threatening organ failure. [4] ECD may virtually affect any organ system. Ihe most frequently involved site is the skeleton (96% Of ECD patients), Often associated With bone pain. A pathognomonic finding is bilateral cortical sclerosis involving the diametaphyseal regions, typically observed on radiographs, associated with an abnormally strong labeling on 990iTc bone scintigraphy. Cardiovascular involvement IS frequent too (75% of cases), mostly presenting as pericardial infiltration, periaortic sheathing ("coated aorta") and myocardial infiltration [51, Central nervous system involvement appears in 51 % of ECD cases, with central diabetes insipidus being the most common finding Pulmonary involvement (43% Of cases) can lead to an interstitial lung disease or pleural effusion. ECD associated involvement of the retroperitoneal space is reported in 68% of the patients, most of which asymptomatic, and infiltration of the perirenal fat produces a typical "hairy kidneys" appearance on CT scan images [7].
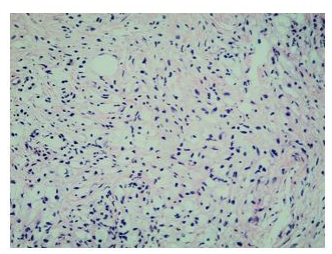
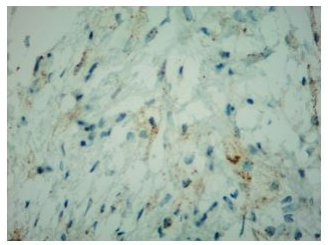
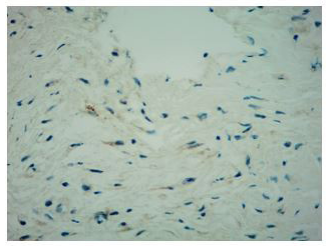
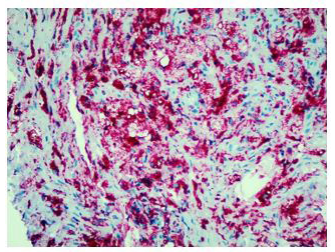
Histologically, xanthogranulomatous or xanthomatous infiltration of the tissue by foamy histiocytes can be detected, with immunohistochemistry (IHC) positive for CD68 and CD163 and negative for CDIa [8].
The prognosis of the disease is poor. At present, the treatment of first choice is interferon alpha [91 (IFN-alpha), with an average 5-year survival of 68%. Infliximab was successfully used in patients with cardiovascular involvement [101 and, in cases of BRAFV€OE mutation, vemurafenib can be used with positive effects on retroperitoneal fibrosis [11].
Case Presentation
We present the case of a 56-year-old man, with work. ing exposure to asbestos for about 30 years. He was affected by hypertension and central insipid diabetes, known since 2012, on therapy with desmopressin.
During 2018 he was hospitalized in the Pneumology Department for left pleural effusion treated with diagnostic- therapeutic thoracoscopy Histological examination of the pleural samples highlighted only inflammatory and reactive aspects.
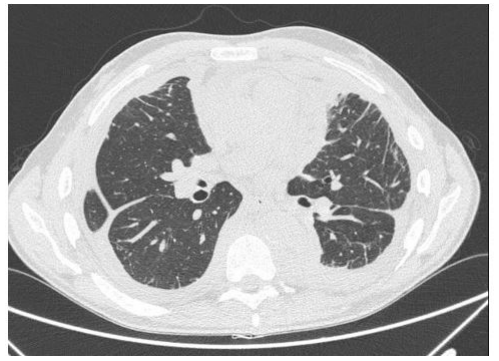
In November 2019 he was hospitalized in the Thoracic Surgery department for bilateral pleural thickening. major on the left, with elevated glucose metabolism assessed by PET-FDG. During hospitalization multiple left pleural biop- sies were performed, documenting a chronic pachypleuritis, in absence of neoplastic cells. Immunohistochemical analysis was positive for calretinin, CK5-6, WT1, EMA, BerEP4, CEAp and HBME1.
In July 2020 he was admitted to our department of In- ternal Medicine for the recent onset of dyspnea after habitual physical activity (climbing a flight of stairs) and weight loss of about 20 kg in the previous 4 months.
At the entrance vital signs were normal and he was apyretic. Physical examination was unremarkable, except for re- duced vesicular breath sounds on the left and the presence Of periorbital xanthelasma.
Blood exams showed neutrophilic leukocytosis (WBC 10.51 x 109,/L, normal value < 10.0 x 109/L, neutrophils 78.0%) mild normocytic anemia (Hb 12.9 g/dL, normal value 13.5- 17. 2 gldl; MCV 77 fl, normal value 80-99 ft.), high platelets (522 x 109,/L, normal value 160-370 x 109/L), RCP 7.64 mg/dL (normal value < 0.50 mg/dL), low total proteins 6.3 g/dL (normal value 6.6-8.3 gldL).
ECG showed sinus rhythm with QTc elongation (492 ms, normal value ms). Pharyngo-tonsillar swab for SARS-CoV.2 infection was negative.
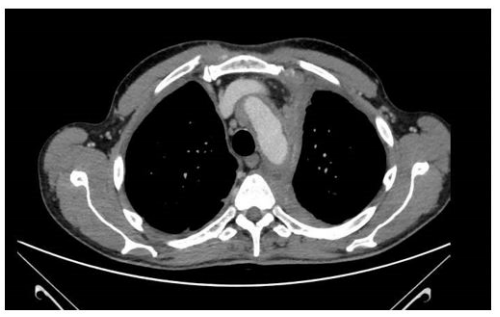
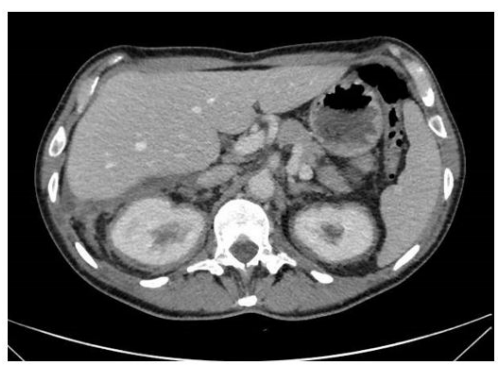
Chest X-ray showed interstitial thickening of lung, ilo-perilary congestion and bilateral pleural effusion major on the left. The findings were investigated with contrast enhanced CT of chest and abdomen, which documented the presence of solid tissue infiltrating both pleurae, mediastinal fatty tissue and pericardium, perirenal fascia and perihepatic area, and surrounding the aortic arch, the proximal tract of the epiaortic vessels.
Case Presentation
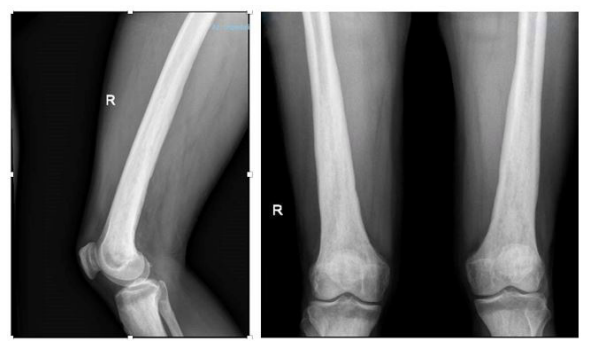
We present the case of a 56-year.old man, with working exposure to asbestos for about 30 years. He was affected by We excluded IgG4-related disease, due to the normal levels of IgG4 on serum and on old histological samples. On suspicion of a multisystem disease, such as Erdheim-Chester disease, we performed bilateral thigh and leg X-rays,reporting symmetrical osteosclerosis of the middle-distal third of both femurs and the middle-distal third of both tibiae.
Further haematochemical tests were performed, documenting increased IL-6 (31.3 pg/mL, normal value < 6.4 pg/mL), slight increase in CD4+/CD8+ ratio (3.73, normal value 1.00- 2.70). Blood levels of TNF alpha, IL-8, IL 12p70, IL-1 Beta, IL-10 were normal.
Finally, percutaneous CT-guided needle biopsy of the perirenal tissue was performed. Histological examination of the sample and IHC documented the presence of fibrous tissue with infiltration of foamy histiocytes containing lipids, with weak positive staining for S-100 and reactive for CD68, consistent with the diagnosis of ECD IHC showed BRAF cytoplasm reactivity of uncertain significance.
Molecular analysis showed BRAF p.Val600Glu (V600E) genetic variant.
In order to evaluate intracranial and orbital involve. ment, gadolinium MRI of the brain was performed, showing short pituitary peduncle. 'Ihe neurohypophysis was not visible and there was no sign of orbital involvement.
Cardiac and mediastinal involvement was investigated by cardiac MRI. which showed elevated Tl myocardial values, suggestive for infiltrative/storage disease. A solid tissue diffusely infiltrated pericardium, especially in the right posterolateral position, and the atrioventricular right sulcus.
Also a bone scintigraphy was performed, revealing hy- perfixation in all long bones (femours, tibias, radius, claviculas), corresponding to the osteostructural osteoaddensing alterations described at X-rays.
The patient was referred to a Rare Diseases Centre (San Raffaele Hospital in Milan) and, due to the presence of BRAFV600E genetic variant, a therapy with Vemurafenib and Anakinra was started, with rapid improvement of dyspnoea.
Discussion
Erdheim-Chester disease is a rare disease. The clinical presentation may range from an indolent focal disease to a life threatening organ failure and it may virtually affect any organ system, therefore the diagnosis is very difficult.
In our patient occupational exposure to asbestos was an important confounding factor. Pleural lesions have guided the main clinical suspicions for years, leading primarily to exclude the diagnosis of mesothelioma.
In fact, the imaging examinations performed during the last hospitalization showed a multiorgan involvement, in. duding coated aorta, hairy kidney. long bone thickening lesions, that lead us to consider a systemic disease. All that findings were consistent with the diagnosis of ECD, which was further corrob- orated by the histological finding of foamy histiocytes containing lipids reactive for CD68.
Conclusion
ECD is a rare disease with multi.organ involvement. lhe aim of this report is to raise awareness of this clinical condition, in order to recognize it, achieve an early diagnosis and start a targeted treatment, improving patients' quality of life.
- Papo M, Emile JF, Maciel TT, Bay P, Baber A, et al. (2019) Erdheim-Chester Disease: a Concise Review. Curr Rheumatol Rep 21: 66.
- Arnaud L, Gorochov G, Charlotte F, Lvovschi V, Parizot C, et al. (2011) Systemic perturbation of cytokine and chemokine networks in Erdheim-Chester disease: a single-center series of 37 patients. Blood 117: 2783-90.
- Badalian-Very G, Vergilio JA, Degar BA, MacConaill LE, Brandner B, et al. (2010) Recurrent BRAF mutations in Langerhans cell histiocytosis. Blood 116: 1919-23.
- Goyal G, Heaney ML, Collin M, Cohen-Aubart F, Vaglio A, et al. (2020) Erdheim-Chester disease: consensus recommendations for evaluation, diagnosis, and treatment in the molecular era. Blood 135: 1929-45.
- Cohen-Aubart F, Emile JF, Carrat F, Helias-Rodzewicz Z, Taly V, et al.(2018) Phenotypes and survival in Erdheim-Chester disease: Results from a 165-patient cohort. Am J Hematol 93: E114-E117.
- Courtillot C, Laugier Robiolle S, Cohen Aubart F, Leban M, Renard-Penna R, et al. (2016) Endocrine Manifestations in a Monocentric Cohort of 64 Patients With Erdheim-Chester Disease. J Clin Endocrinol Metab 101: 305-13.
- Cavalli G, Guglielmi B, Berti A, Campochiaro C, Sabbadini MG, et al. (2013) The multifaceted clinical presentations and manifestations of Erdheim-Chester disease: comprehensive review of the literature and of 10 new cases. Ann Rheum Dis 72: 1691-5.
- Diamond EL, Dagna L, Hyman DM, Cavalli G, Janku F, et al. (2014) Consensus guidelines for the diagnosis and clinical management of Erdheim-Chester disease. Blood 124: 483-92.
- Hervier B, Arnaud L, Charlotte F, Wechsler B, Piette JC, Amoura Z, et al. (2012) Treatment of Erdheim-Chester disease with long-term high-dose interferon-α. Semin Arthritis Rheum 41: 907-13.
- Graziani G, Podestà MA, Cucchiari D, Reggiani F, Ponticelli C (2014) Erdheim-Chester disease: from palliative care to targeted treatment. Clin Kidney J 7: 339-43.
- Haroche J, Cohen-Aubart F, Emile JF, Arnaud L, Maksud P, et al. (2013) Dramatic efficacy ofvemurafenib in both multisystemic and refractory Erdheim-Chester disease and Langerhans cell histiocytosis harboring the BRAF mutation. Blood 121: 1495-5(X).

Figures at a glance