Apatinib Combined with an Anti-PD-1 Agent Promotes Immune Response to Inhibit Tumour Growth in Mice with Ovarian Cancer
Received Date: May 13, 2024 Accepted Date: June 13, 2024 Published Date: June 17, 2024
doi: 10.17303/jcrto.2024.12.301
Citation: Weixuan Luo, Chunyue Liu, Xiaoyan Cheng, Wei Xu, Wenjun Zhou, et al. (2024) Apatinib Combined with an Anti-PD-1 Agent Promotes Immune Response to Inhibit Tumour Growth in Mice with Ovarian Cancer. J Cancer Res Therap Oncol 12: 1-11
Abstract
Objective: Apatinib is a noval antiangiogenic agents, and it has great clinical benefit. Immunotherapy has a low response rate in patients with ovarian cancer, but combination with antiangiogenic therapy might improve the antitumour response. The aim of our study was to assess the expression of programmed cell death 1 ligand 1 (PD-L1) in vivo and in vitro and the potential effect of apatinib in combination with an anti-PD-1 antibody in mice with ovarian cancer.
Methods:We assessed the expression of PD-L1 in vivo and in vitro by real-time quantitative reverse transcription–polymerase chain reaction (qRT‒PCR), flow cytometry and western blotting in different concentrations. We constructed a xenograft model and measured mouse weight and tumour size after treatment. Tumour tissues from the mice were subjected to HE staining, and we detected Ki-67, CD4 and CD8 expression by immunohistochemical staining (IHC).
Results: Apatinib monotherapy increased the expression of PD-L1 by dose-dependent, as determined by qRT‒PCR, flow cytometry, and western blotting in vivo and in vitro. Apatinib combined with an anti-PD-1 antibody had the most pronounced antitumour effect on ovarian cancer among the combination treatment and the two monotherapies. The most obvious changes were a decrease in Ki-67 expression and increases in CD4 and CD8 expression.
Conclusions: Our study showed that apatinib upregulates the expression of PD-L1 to exert antitumour effects. Apatinib in combination with an anti-PD-1 antibody increased the immune response to lead to a remarkable reduction in tumour growth, providing evidence for clinical trials.
Keywords: Apatinib; Anti-PD-1; Ovarian Cancer; Immune response
Introduction
In 2020, ovarian cancer had the highest death rate among gynaecological malignancies, accounting for 207,252 deaths [1]. Because the symptoms of ovarian cancer are untypical, the main treatment for early-stage disease is comprehensive staging surgery; the long-term survival rate of early-stage disease is greater than 80-95% [2,3]. The current standardized treatment for late-stage ovarian cancer is cytoreductive surgery plus platinum-based chemotherapy. The first-line adjuvant systemic therapy is paclitaxel + carboplatin, with or without bevacizumab or PARP inhibitor (PARPi) [4], which can prolong the median progression-free survival (PFS) but cannot prolong overall survival (OS) [5]. Approximately 70% of patients relapse within 2-5 years, and the 5-year survival rate is 40-45%, with a median PFS of 16 months [6]. Eventually, platinum resistance leads to patient death [7]. Therefore, effective and safe drugs are needed to increase the survival rate and prolong the life of patients.
Apatinib is a novel oral tyrosine kinase inhibitor that combines with vascular endothelial growth factor receptor-2 (VEGFR-2) to inhibit tumour proliferation, invasion and metastasis via normalization of tumour vessels [8]. Previous studies revealed that apatinib could prolong OS and median PFS in patients with malignancies including gastroesophageal adenocarcinoma (GEA), non-small cell lung cancer (NSCLC), breast cancer and hepatocellular carcinoma (HCC) [9]. Recently, based on phase II and III clinical trials in gastric cancer, apatinib was approved by the China Food and Drug Administration. The results showed that apatinib had high safety and remarkable therapeutic efficacy in treating patients with advanced gastric cancer [10,11]. Wang [12] and Zhang [13] demonstrated that apatinib alone or in combination with chemotherapeutic agents had promising clinical efficacy and tolerable adverse effects in advanced, recurrent, platinum-resistant ovarian cancer. Furthermore, our present study revealed that apatinib prevents tumour growth in vivo and in vitro [14].
Immune checkpoint inhibitors (ICIs), such as PD-1/PD-L1 axis, play an important role of antitumour. PD-1 is expressed on tumour-specific T cells, and its ligand, PD-L1, is expressed by tumour cells and activates immune escape [15]. PD-1 combined with PD-L1 receptors can escape immune surveillance in tumours and induce T-cell apoptosis. ICIs, which can prolong patient survival, exhibit satisfactory clinical efficacy for treating many malignant tumours. Currently, PD-1/PD-L1 inhibitors are also approved by the Food and Drug Administration for the treatment of many solid tumours [16]. PD-1/PD-L1 inhibitors have been proven to be effective in treating NSCLC and HCC [17]. However, the PD-1/PD-L1 axis plays an important role in ovarian cancer, but only 10-15% of patients achieve a durable response [18].
Previous research revealed that combined therapies are more efficacious and safety than apatinib or PD-1/PD-L1 inhibitors monotherapy in treating HCC, cervical cancer, and colon cancer [19]. Studies have shown that anti-VEGF therapy combined with anti-PD-1/PD-L1 therapy has better treatment outcomes and manageable toxicity, with a better objective response rate (ORR) than anti-PD-1/PD-L1 monotherapy (30% vs. 9%) [6,20].
Currently, there are no clinical trial data on the combination of apatinib with PD-1/PD-L1 inhibitors in ovarian cancer. Our study was designed to assess the immune response of apatinib combined with a PD-1 inhibitor in ovarian cancer.
Methods
ReagentsApatinib was obtained from Hengrui Medicine Co., Ltd. (Jiangsu, China). For the in vitro studies, apatinib was dissolved in 100% dimethyl sulfoxide (DMSO) and diluted to the desired concentration with Dulbecco's modified Eagle's medium (DMEM) (Gibco, USA). For the in vivo studies, apatinib was diluted in phosphate-buffered saline.
The antibodies used in this study were as follows: Anti-PD-1 (αPD-1) mAb was purchased from Bio X Cell (West Lebanon, NH, USA) (BE0273).
The PerCP/Cyanine5.5 anti-human CD274 (B7- H1, PD-L1) antibody was purchased from Biolegend (San Diego, CA, USA) (329738).
Anti-Ki67, anti-CD4, and anti-CD8 antibodies (28074-1-AP, 67786-1-Ig, 9896-1-AP) were purchased from Proteintech (Wuhan, China).
Cell CultureThe human ovarian cancer cell line SKOV3, ES-2, A2780 and murine ovarian serous carcinoma cell line ID8 were purchased from the American Type Culture Collection and cultured in Dulbecco's modified Eagle’s medium (DMEM; Gibco, USA) supplemented with 10% foetal bovine serum (FBS; Gibco, USA), 100 U/ml penicillin and 100 μg/ml streptomycin (penicillin‒streptomycin-glutamine; Gibco, USA) at 37°C in a humidified atmosphere of 5% CO2 in a Forma Steri-Cycle CO2 incubator (Thermo Fisher Scientific, Massachusetts, USA).
Real-Time Quantitative Reverse Transcription–Polymerase Chain ReactionTotal RNA was isolated from SKOV3 cells using TRIzol reagent (CWbiotech, Beijing, China), and RNA was subsequently reverse transcribed with a PrimeScript RT reagent kit (TaKaRa, Otsu, Japan). The acquired cDNAs were used as templates for real-time quantitative reverse transcription–polymerase chain reaction (qRT–PCR) analysis using SYBR Green PCR Master Mix (Bio-Rad, California, USA). The relative RNA expression levels were calculated using the 2-ΔΔCt method, with the target mRNA levels normalized to those of β-actin.
Flow CytometryFlow cytometric analysis was also conducted to determine the PD-L1 expression level in SKOV3 cells treated with 5, 10, 20 and 30 μM apatinib. The cells were initially seeded onto six-well plates at a density of 3 × 105 cells per well. After a 24-h incubation period, the cells were treated with apatinib at concentrations of 5, 10, 20 and 30 μM for an additional 24 h, after which the cells were harvested and washed twice with prechilled PBS. After washing with PBS, the cells were stained with an anti-PD-L1 antibody (San Diego, CA, USA) (329738) at RT for 30 min. The stained cells were analysed using a flow cytometer (FACS Aria Ⅱ; BD Biosciences, San Jose, USA).
Mice and Tumour ModelsSix-week-old female C57BL/6 mice were purchased and housed under standard pathogen-free conditions in the animal centre of the Institute of Analysis and Testing, Beijing Academy of Science and Technology (Beijing Physical and Chemical Analysis and Testing Center). To establish in vivo models, 5×106 ID8 cells were subcutaneously injected into six-week-old C57BL/6 mice. When the tumours reached a mean diameter of 6 mm, the mice were randomly divided into four groups: control (PBS), apatinib (30 mg/kg, every day), anti-PD-1 (5 mg/kg, every 3 days), and apatinib (30 mg/kg, every 3 days) + anti-PD-1 (5 mg/kg, every 3 days). Throughout the treatment, the mice were weighed, and their tumours were measured with a calliper every 3 days. Tumour volume (V) was calculated using the formula V = larger diameter × (smaller diameter)2 /2 mm 3. After 15 days of treatment, when the tumours reached an appropriate size, the mice were euthanized, and the tumours were excised, weighed, and either stored at –80°C or fixed with 4% paraformaldehyde for subsequent examination. All animal experiments were approved by the Laboratory Animal Welfare and Ethics Committee of the Institute of Analysis and Testing, Beijing Academy of Science and Technology (Beijing Physical and Chemical Analysis and Testing Center).
Western BlotAfter SKOV3 cells were treated with 0, 5, 10, 20 or 30 μM of the indicated drug(s), protein was extracted from the SKOV3 cells and xenograft tumour tissue via incubation in lysis buffer (Beyotime). The protein concentrations were quantified via a BCA protein assay kit (TIANGEN BIOTECH, China). The proteins were separated by SDS‒polyacrylamide gel electrophoresis (SDS‒PAGE) and transferred to polyvinylidene fluoride (PVDF) membranes (Millipore, Billerica, MA, USA). After blocking for 1 h with nonfat milk (BD, USA), the membranes were incubated with the primary antibody against PD-L1 (#ab213480, 1:1000; Abcam, Cambridge, UK) at 4°C overnight, after which the membranes were washed 3 times with Tris-buffered saline (TBS). Subsequently, the membranes were incubated with HRP-conjugated secondary antibodies (#A0208, 1:10000; Beyotime) at room temperature for 2 h and washed with TBS buffer 3 times. The protein signals were visualized via an enhanced chemiluminescence (ECL) detection system (Amersham, Bucks, UK).
Immunohistochemical StainingLongitudinal sections (5 μm) of the paraffinembedded tumours were kept at 60°C for 24 h in an oven, deparaffinized with xylene and hydrated with an ethanol gradient (100%–70%). Heat-induced antigen retrieval was performed in citrate buffer in an MLS-3750 autoclave (Sanyo, Japan) for 2 min. After incubation with 3% H2O2 for 10 min, the slides were rinsed with water and incubated with the primary antibodies against Ki67, CD4 and CD8 overnight at 4°C. The next day, the slides were rinsed and incubated with the corresponding secondary antibody (Beijing Zhongshan Golden Bridge Biotechnology Co., Ltd., Beijing, China) for 30 min and then stained with 3,3'-diaminobenzidine (DAB) and haematoxylin. The slides were then examined and photographed using an Axio Observer A1 microscope (Zeiss, Germany).
Haematoxylin-eosin (H&E) StainingHaematoxylin and eosin (HE) staining of ovarian tissues was conducted according to routine protocols. Briefly, after deparaffinization and rehydration, 5-μm longitudinal sections were stained with haematoxylin solution for 5 min, dipped 5 times in 1% acid ethanol (1% HCl in 70% ethanol) and then rinsed in distilled water. Then, the sections were stained with eosin solution for 8 min, dehydrated with graded alcohol and cleared in xylene. The mounted slides were then examined and photographed using an Observer A1 fluorescence microscope (Zeiss, Germany).
Statistical AnalysisThe data are presented as the mean values with standard errors of the means. Statistical analysis was performed with Prism GraphPad 6.0 software (GraphPad Software, Inc., San Diego, CA, USA). Differences between groups were examined by Student’s t test or one-way ANOVA followed by Bonferroni correction or two-way ANOVA with multiple comparisons test for comparison of means. A P value < 0.05 indicated a significant difference, and a P value < 0.01 indicated a statistically significant difference (*P < 0.05, **P < 0.01).
Results
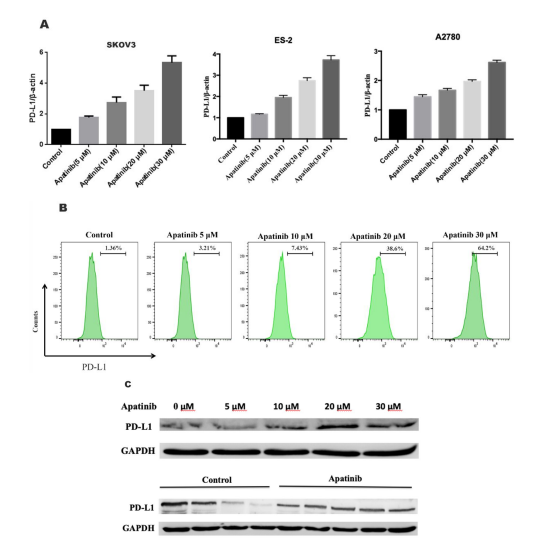
Apatinib Increases the Expression of PD-L1 in Ovarian Cancer Cells and in vivo ModelsTo investigate the effect of apatinib on PD-L1 expression in ovarian cancer cells, we selected the ovarian cancer cell lines SKOV3, ES-2, and A2780 and incubated them with different concentrations of apatinib (0, 5, 10, 20 and 30 μM) for 24 h. qRT‒PCR showed that apatinib upregulated the mRNA expression of PD-L1 in a dose-dependent manner and was most effective at 30 μM (Figure 1A). The qRT‒PCR results showed that the increase in PD-L1 mRNA expression in the SKOV3 ovarian cancer cell line was the most obvious. Therefore, in the following experiments, we selected SKOV3 ovarian cancer cells for study. Cells treated with apatinib were harvested and subjected to flow cytometry and western blot analysis. The PD-L1 protein level in the cells was increased after apatinib treatment (Figure 1B). Western blotting was used to explore the effect of apatinib on the expression of the PD-L1 protein in mouse tumour tissues. The results indicated that the expression of the PD-L1 protein in tumour tissues increased after the administration of apatinib (Figure 1C).
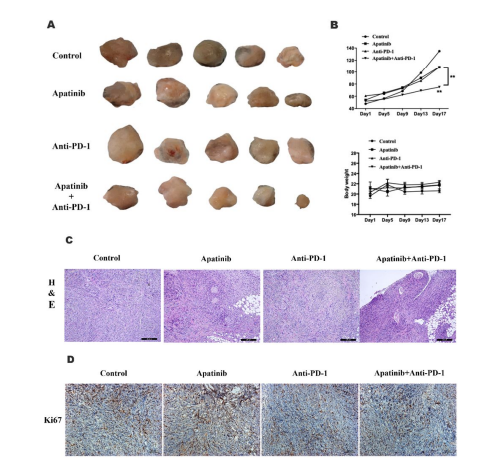
Apatinib Combined with an Anti-PD-1 Antibody Significantly Inhibited Tumour Growth in vivoWe further examined the effect of the combination of apatinib with an anti-PD-1 agent on tumour growth in vivo using a syngeneic mouse model of human ovarian cancer after characterizing its effects on ovarian cancer cells in vitro. As shown in Figure 2, compared with the control treatment, treatment with apatinib alone or the anti-PD-1 antibody alone inhibited ID8 tumour growth. However, the inhibitory effect of apatinib combined with anti-PD-1 therapy was significantly greater (Figure 2A). After 15 days, the tumour volume in the apatinib combined with anti-PD-1 therapy group was significantly smaller than that in the negative control group and the apatinib alone and anti-PD-1 antibody alone groups (P < 0.01). No significant differences in the body weights of the mice were observed between the groups before or after administration (Figure 2B). In addition, H&E staining of tumour tissue sections also indicated that the combination of apatinib and an anti-PD-1 antibody significantly increased the area of tumour necrosis compared with that in the single-drug-treated group (Figure 2C). Immunohistochemical staining revealed a dramatic decrease in Ki67 expression after apatinib and anti-PD-1 cotreatment (Figure 2D). The results mentioned above suggested that the combination of apatinib and an anti-PD-1 antibody significantly inhibited the growth of transplanted tumours in mice.
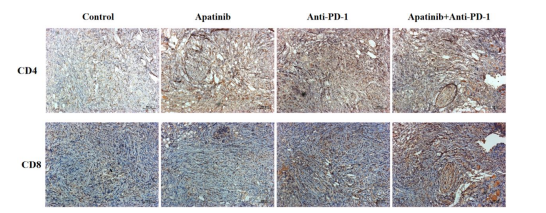
Apatinib Combined with an Anti-PD-1 Antibody Increased the Infiltration of CD4+ T cells and CD8+ T cells in vivoThe combination treatment had a significant antitumour effect on, and we investigated whether the four treatments had an impact on T-cell infiltration. The infiltration of CD4+ and CD8+ T cells in tumour tissue was examined by IHC. As shown in Figure 3, apatinib alone or anti-PD-1 therapy alone promoted the expression of CD4 and CD8. However, the apatinib combined with anti-PD-1 antibody group were further increased, as determined by IHC (Figure 3). The results suggested that the combination treatment increased the infiltration of CD4+ and CD8+ T cells compared with single-drug-treated group in the TME. This enhances the activation of T cells to promote antitumour effects.
Discussion
The mortality rate of ovarian cancer is the highest among gynaecologic malignancies because it is difficult to diagnose in the early-stage and it has a high recurrence rate in the late stage; these factors contribute to the low 5-year survival rate. Anti-angiogenic drug combined with anti-PD-1/anti-PD-L1 antibody inhibit the tumour growth, synergistically. However, there are no clinical trials on the treatment of ovarian cancer by the use of apatinib combined with anti-PD-1 therapy.
Angiogenesis plays a crucial role in malignancy. Tumour cells reduce the expression of VEGF while increasing oxygen consumption and are in a hypoxic state. Through many signalling pathways, VEGF and VEGFR promote the generation of new blood vessels to increase the need for oxygen and nutrients, which leads to tumour growth [21]. Apatinib is a selected targeted VEGFR-2 inhibitor. Previous studies have indicated that apatinib promotes the proliferation of endothelial cells and suppresses the generation of new blood vessels in tumour, which provides theoretical foundation for combined with other therapies [8]. Our study revealed that apatinib upregulates the expression of PD-L1 in a dose-dependent manner, as determined by qRT‒PCR and flow cytometry, and the same results were observed in the mouse model. Similarly, apatinib increased the expression of PD-L1 in gastric cancer and colon cancer [22,23], which may be related to IFN-γ and hypoxia [24]. PD-L1, via IFN-γ and hypoxia, inactivates T cells in tumours to inhibit immune function [25]. In the xenograft models, apatinib group also had anti-tumour effect, and it was related to the upregulation of PD-L1
ICIs are the effective drugs to treat many malignancies. PD-1/PD-L1 axis is hotpot in immunotherapy. In ovarian cancer, 47.9% of tumour cells exhibit high expression of PD-L1 [26]. However, clinical trials of anti-PD-1 monotherapy have also shown limited clinical efficacy in patients with ovarian cancer, with a response rate of approximately 10% [27]. When combined with antiangiogenic agents, such as anti-VEGF inhibitors, ICIs might synergistically inhibit the progression of tumours. Based on the significant efficacy and acceptable toxicity of the combination treatment in other cancers, our study was designed to evaluate the effect of apatinib combined with anti-PD-1 therapy on ovarian cancer in vivo.
In the mouse model, the three experimental groups inhibited tumour growth. However, the combination treatment had the most obvious effect. H&E staining revealed the same results. However, there were no significant differences in mouse survival between treatment groups. On the basis of our findings, we draw conclusion that apatinib combined with an anti-PD-1 antibody has a better clinical effect in treating ovarian cancer but has no impact on survival. Ki-67, a cell cycle-related nuclear protein, is a marker of cell proliferation and tumour occurrence [28]. The degree of Ki-67 downregulation in the combined therapy group was greater than that in the other groups. In many cancers, such as ovarian cancer, high Ki-67 expression is associated with poor prognosis and low OS [29]. Our studies demonstrated that the combination treatment had better effects in terms of the inhibition of tumour proliferation compared with monotherapy
Tumour-infiltrating lymphocytes are the crucial component of the adaptive immune response and constitute the basis of immunotherapy [30]. CD8+ T cells exert antitumour effect, as well as CD4+ T cells, and a high level of infiltration of CD8+ T cells is related to a better prognosis [31]. However, the PD-1/PD-L1 axis decreases the infiltration of CD8+ T cells to induce immune evasion by increasing IFN-γ levels [32]. Moreover, VEGF reduces the level of CD8+ T cells though TOX to inhibit the immune response [33]. Our study showed that the three experimental groups promoted the infiltration of CD4+ and CD8+ T cells into the TME with varying degree, and the infiltration of CD4+ and CD8+ T cells in the combination therapy group was greater than that in the other groups. Similar results have been reported in digestive system malignancies [34]. CD8+ T cells interrupt angiogenesis by secreting IFN-γ [25]. Moreover, CD8+ T cells upregulate the expression of PD-L1 via IFN-γ [35,36]. In addition, similar results were observed in a mouse subcutaneous model [37]. However, the mechanism through which apatinib combined with an anti-PD-1 agent affects the expression of IFN-γ in ovarian cancer is unclear. Overall, our study showed that combination group promoted immune response to decrease the tumour growth compared the other groups.
In summary, apatinib increased the expression of PD-L1 in vitro and vivo, possibly through a mechanism related to IFN-γ. However, additional evidence is needed to prove this. Apatinib combined with an anti-PD-1 agent exerted a better antitumour effect in vivo and promoted the infiltration of T cells. This study provides evidence supporting the use of this strategy in clinical trials of ovarian cancer.
Statements and Declaration
Acknowledgements
We are grateful to Mrs. Xiaoyan Cheng for her excellent assistance with the experiments.
Funding
This study was supported by the Haiyan Foundation of Harbin Medical University Cancer Hospital (04000145) and the Science and Technology Education Research Project of Heilongjiang Province (2023-KYYWF-0229).
Ethics Approval
All animal experiments were approved by the Laboratory Animal Welfare and Ethics Committee of the Institute of Analysis and Testing, Beijing Academy of Science and Technology (Beijing Physical and Chemical Analysis and Testing Center). The research was conducted in accordance with the Declaration of Helsinki.
Competing of Interest
There are no conflicts of interest associated with this work.
Author Contributions
Weixuan Luo and Chunyue Liu contributed equally to this article. Weixuan Luo and Chunyue Liu designed the experiments, performed the experiments, analysed and interpreted the data and wrote the first draft of the manuscript. Xiaoyan Cheng, Wei Xu and Wenjun Zhou performed the experiments and analysed and interpreted the data. Jiaxin Ma, Rui Ren and Meina Tian collected the data. Jing Ding designed the experiments and supervised the study. The first draft of the manuscript was written by Weixuan Luo and Chunyue Liu, and all the authors commented on previous versions of the manuscript. All the authors read and approved the final manuscript.
Date Availability Statement
The data underlying this article are available in the article and in its online supplementary material.
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. (2021) Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA: a cancer journal for clinicians, 71: 209-49.
- Stewart C, Ralyea C, Lockwood S (2019) Ovarian Cancer: An Integrated Review. Seminars in oncology nursing, 35: 151-6.
- Trimbos JB (2017) Surgical treatment of early-stage ovarian cancer. Best practice & research Clinical obstetrics & gynaecology, 41: 60-70.
- Armstrong DK, Alvarez RD, Backes FJ, BakkumGamez JN, Barroilhet L, Behbakht K, et al. (2022) NCCN Guidelines® Insights: Ovarian Cancer, Version 3.2022. Journal of the National Comprehensive Cancer Network : JNCCN, 20: 972-80.
- Morand S, Devanaboyina M, Staats H, Stanbery L, Nemunaitis J (2021) Ovarian Cancer Immunotherapy and Personalized Medicine. International journal of molecular sciences, 22.
- Mei C, Gong W, Wang X, Lv Y, Zhang Y, Wu S, et al. (2023) Anti-angiogenic therapy in ovarian cancer: Current understandings and prospects of precision medicine. Frontiers in pharmacology, 14: 1147717.
- Hartnett EG, Knight J, Radolec M, Buckanovich RJ, Edwards RP, Vlad AM (2020) Immunotherapy Advances for Epithelial Ovarian Cancer. Cancers, 12.
- Zhou K, Zhang JW, Wang QZ, Liu WY, Liu JL, Yao L, et al. (2019) Apatinib, a selective VEGFR2 inhibitor, improves the delivery of chemotherapeutic agents to tumors by normalizing tumor vessels in LoVo colon cancer xenograft mice. Acta pharmacologica Sinica, 40: 556-62.
- Scott LJ (2018) Apatinib: A Review in Advanced Gastric Cancer and Other Advanced Cancers. Drugs, 78: 747-58.
- Geng R, Song L, Li J, Zhao L (2018) The safety of apatinib for the treatment of gastric cancer. Expert opinion on drug safety, 17: 1145-50.
- Roviello G, Ravelli A, Fiaschi AI, Cappelletti MR, Gobbi A, Senti C, et al. (2016) Apatinib for the treatment of gastric cancer. Expert review of gastroenterology & hepatology, 10: 887-92.
- Wang Z, Huang Y, Long L, Zhou L, Huang Y, Gan L, et al. (2021) Apatinib treatment efficiently delays biochemical-only recurrent ovarian cancer progression. Journal of ovarian research, 14: 91.
- Zhang J, Li A, Jiang Q, Zheng F, Zhu H (2019) Efficacy And Safety Of Apatinib Treatment In Platinum-Resistant Recurrent Epithelial Ovarian Cancer: A Real World Study. Drug design, development and therapy, 13: 3913-8.
- Ding J, Cheng XY, Liu S, Ji HY, Lin M, Ma R, et al. (2019) Apatinib exerts anti-tumour effects on ovarian cancer cells. Gynecologic oncology, 153: 165-74.
- Dumitru A, Dobrica EC, Croitoru A, Cretoiu SM, Gaspar BS (2022) Focus on PD-1/PD-L1 as a Therapeutic Target in Ovarian Cancer. International journal of molecular sciences, 23.
- Twomey JD, Zhang B (2021) Cancer Immunotherapy Update: FDA-Approved Checkpoint Inhibitors and Companion Diagnostics. The AAPS journal, 23: 39.
- Sun C, Mezzadra R, Schumacher TN (2018) Regulation and Function of the PD-L1 Checkpoint. Immunity, 48: 434-52.
- Chardin L, Leary A (2021) Immunotherapy in Ovarian Cancer: Thinking Beyond PD-1/PD-L1. Frontiers in oncology, 11: 795547.
- Tian Z, Niu X, Yao W (2021) Efficacy and Response Biomarkers of Apatinib in the Treatment of Malignancies in China: A Review. Frontiers in oncology, 11: 749083.
- Zhu J, Yan L, Wang Q (2021) Efficacy of PD-1/PDL1 inhibitors in ovarian cancer: a single-arm meta-analysis. Journal of ovarian research, 14: 112.
- Choi HJ, Armaiz Pena GN, Pradeep S, Cho MS, Coleman RL, Sood AK (2015) Anti-vascular therapies in ovarian cancer: moving beyond anti-VEGF approaches. Cancer metastasis reviews, 34: 19-40.
- Cai X, Wei B, Li L, Chen X, Liu W, Cui J, et al. (2020) Apatinib enhanced anti-PD-1 therapy for colon cancer in mice via promoting PD-L1 expression. International immunopharmacology, 88: 106858.
- Chen H, Jiang T, Lin F, Guan H, Zheng J, Liu Q, et al. (2021) PD-1 inhibitor combined with apatinib modulate the tumor microenvironment and potentiate anti-tumor effect in mice bearing gastric cancer. International immunopharmacology, 99: 107929.
- Noman MZ, Desantis G, Janji B, Hasmim M, Karray S, Dessen P, et al. (2014) PD-L1 is a novel direct target of HIF-1α, and its blockade under hypoxia enhanced MDSC-mediated T cell activation. The Journal of experimental medicine, 211: 781-90.
- Lee WS, Yang H, Chon HJ, Kim C (2020) Combination of anti-angiogenic therapy and immune checkpoint blockade normalizes vascular-immune crosstalk to potentiate cancer immunity. Experimental & molecular medicine, 52: 1475-85.
- Alwosaibai K, Aalmri S, Mashhour M, Ghandorah S, Alshangiti A, Azam F, et al. (2023) PD-L1 is highly expressed in ovarian cancer and associated with cancer stem cells populations expressing CD44 and other stem cell markers. BMC cancer, 23: 13.
- Varga A, Piha-Paul S, Ott PA, Mehnert JM, Berton-Rigaud D, Morosky A, et al. (2019) Pembrolizumab in patients with programmed death ligand 1-positive advanced ovarian cancer: Analysis of KEYNOTE-028. Gynecologic oncology, 152: 243-50.
- Davey MG, Hynes SO, Kerin MJ, Miller N, Lowery AJ (2021) Ki-67 as a Prognostic Biomarker in Invasive Breast Cancer. Cancers, 13.
- Liu P, Sun YL, Du J, Hou XS, Meng H (2012) CD105/Ki67 coexpression correlates with tumor progression and poor prognosis in epithelial ovarian cancer. International journal of gynecological cancer : official journal of the International Gynecological Cancer Society, 22: 586-92.
- Liu D, Heij LR, Czigany Z, Dahl E, Lang SA, Ulmer TF, et al. (2022) The role of tumor-infiltrating lymphocytes in cholangiocarcinoma. Journal of experimental & clinical cancer research: CR, 41: 127.
- Hamanishi J, Mandai M, Abiko K, Matsumura N, Baba T, Yoshioka Y, et al. (2011) The comprehensive assessment of local immune status of ovarian cancer by the clustering of multiple immune factors. Clinical immunology (Orlando, Fla), 141: 338-47.
- Peng Z, Li M, Li H, Gao Q (2023) PD-1/PD-L1 immune checkpoint blockade in ovarian cancer: Dilemmas and opportunities. Drug discovery today, 28: 103666.
- Wang Q, Gao J, Di W, Wu X (2020) Anti-angiogenesis therapy overcomes the innate resistance to PD-1/PD-L1 blockade in VEGFA-overexpressed mouse tumor models. Cancer immunology, immunotherapy : CII, 69: 1781-99.
- Li H, Huang H, Zhang T, Feng H, Wang S, Zhang Y, et al. (2022) Apatinib: A Novel Antiangiogenic Drug in Monotherapy or Combination Immunotherapy for Digestive System Malignancies. Frontiers in immunology, 13: 937307.
- Abiko K, Hamanishi J, Matsumura N, Mandai M (2023) Dynamic host immunity and PD-L1/PD-1 blockade efficacy: developments after "IFN-γ from lymphocytes induces PD-L1 expression and promotes progression of ovarian cancer". British journal of cancer, 128: 461-7.
- Bagchi S, Yuan R, Engleman EG (2021) Immune Checkpoint Inhibitors for the Treatment of Cancer: Clinical Impact and Mechanisms of Response and Resistance. Annual review of pathology, 16: 223-49.
- Abiko K, Matsumura N, Hamanishi J, Horikawa N, Murakami R, Yamaguchi K, et al. (2015) IFN-γ from lymphocytes induces PD-L1 expression and promotes progression of ovarian cancer. British journal of cancer, 112: 1501-9.

Figures at a glance