Hyperamylasemia as a Potential Indicator of Ovarian Cancer: Case Report
Received Date: July 08, 2025 Accepted Date: July 16, 2025 Published Date: July 25, 2025
doi:10.17303/jcrto.2025.13.201
Citation: Wang Jingzhi, Ye Wang, Yue Niu, Jingyang Wang, Wenting Liu (2025) Case Report: Hyperamylasemia as a Potential Indicator of Ovarian Cancer J Cancer Res Therap Oncol 13: 1-7
Abstract
Rationale: We report a case of ovarian cancer with abnormally elevated blood amylase.
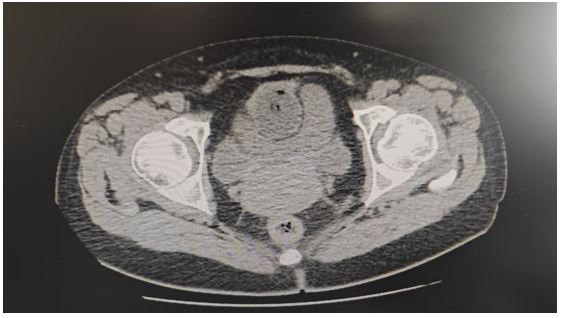
Patient's concerns: A 46-year-old woman was admitted to the hospital due to lower abdominal pain. Her blood amylase was abnormally elevated. Pelvic CT enhancement showed a mass-like mixed, slightly lower and equal density shadow in the left adnexal area. The shape was irregular and the size was about 59×53mm.
Diagnosis: Postoperative pathology suggested ovarian cancer. Postoperative blood amylase returned to normal. Pancreatitis was excluded based on clinical symptoms and imaging examinations.
Intervention: The patient underwent surgery. Because only one side of the fallopian tube and ovary were removed in the emergency surgery, the patient underwent elective surgery again.
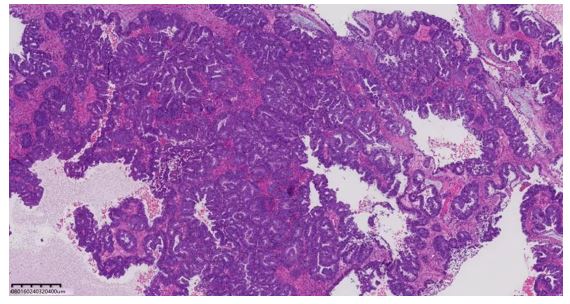
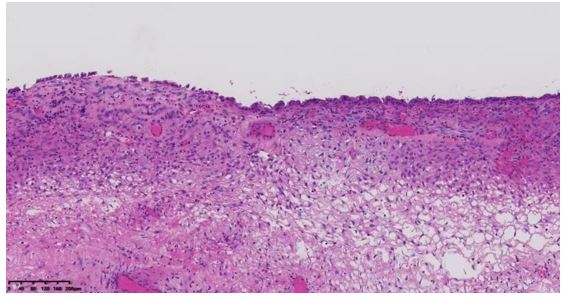
Outcomes: Postoperative pathology suggested: (left side) well-differentiated ovarian endometrioid carcinoma, with endometriosis in the background. (left side) Endometriosis in the fallopian tube serosa
Experience: After excluding digestive system diseases, elevated blood amylase should be considered as a possible ovarian cancer. Provide a new laboratory reference index for the diagnosis of ovarian cancer.
Keywords: Hyperamylasemia; Ovarian Carcinoma; Surgery; Hyperamylasemia; Ovarian Cancer
Abbreviation: CT = computed tomography.
Introduction
Ovarian cancer is still the leading cause of death from malignant tumors in Chinese women. The early symptoms are not obvious, and most of them are in the late stage when they are discovered. At present, the main tumor markers for ovarian cancer are CA-125 CA-199, but the specificity is poor. After consulting the literature, we found several reports of abnormally elevated blood amylase, excluding digestive system diseases and finally determining ovarian cancer. However, reports of abnormally elevated blood amylase and pathological results showing that ovarian endometriosis cysts have transformed into well-differentiated ovarian endometrioid carcinoma are relatively rare. We reported a case of abnormally elevated blood amylase, which was finally diagnosed as ovarian endometriosis cysts transforming into ovarian cancer by pathology.
Case Report
The patient, female, 46 years old, was admitted for "lower abdominal pain for two hours." Half a day prior, the patient had palpated a lower abdominal mass herself, approximately 7 cm in diameter, firm in consistency, accompanied by abdominal distension, but no abdominal pain, tenderness, nausea, vomiting, diarrhea, constipation, or other discomforts. Bowel and urinary habits were normal; the mass persisted after defecation. About two hours before admission, upon applying pressure to the mass, she experienced sudden severe lower abdominal pain with associated nausea but no vomiting. Urination remained normal; a single bowel movement yielded no hematuria, melena, hematochezia, or diarrhea. The pain gradually subsided after about an hour. Emergency examination revealed mild abdominal muscle tension and positive lower abdominal tenderness, with no obvious rebound pain. Bedside abdominal Doppler ultrasonography was inconclusive due to poor filling and limited visualization. Transabdominal Doppler ultrasound showed the uterus was anteverted, normal in morphology, with heterogeneous myometrial echotexture, midline alignment, and an endometrial thickness of 0.93 cm. The right adnexa was visible while the left side was poorly visualized. A cystic area was noted with poor internal sound transmission and numerous fine punctate hypoechoic signals, measuring about 9.9×4.8 cm in total; within the left aspect there was an inhomogeneous hyperechoic mass about 5.4×5.0 cm, displaying blood flow signal with arterial spectral measurement: Vp 46 cm/s, RI 0.68. The abnormal findings suggested possible corpus luteum rupture and/or mass lesion with fluid (hemoperitoneum). No obvious uterine abnormality was noted; repeat ultrasound was recommended once the patient’s condition stabilized.
CT on April 8, 2025, post-indwelling urinary catheter placement, revealed suboptimal filling with balloon and gas shadow inside. Local protrusion of the uterus was observed. The left adnexal area showed a mass-like, heterogeneous, slightly hypodense to isodense shadow, irregular in shape, approximately 59×53 mm. The right adnexal area was full. No marked wall thickening or significant enlargement. Small amount of fluid accumulation. A left adnexal lesion was noted, recommending further clinical correlation; changes post-catheter placement; possible uterine fibroid; minimal effusion. Laboratory results showed CA-125: 88.29 U/ml (reference range 0–35), CA-199: 46.89 U/ml (reference range 0–30), while CEA, AFP, and SCC were all within normal limits. Serum amylase was 477.0 U/L (reference range 35–135), while lipase was 33.2 U/L (reference range 0–60). Liver and renal function tests were normal.
Following admission, the patient immediately underwent laparoscopic exploration. Intraoperatively, approximately 300 mL of dark-brown fluid was found in the pelvic cavity. The uterus was enlarged to the size of a 7-week gestation, smooth-surfaced, with multiple intramural fibroids (2.0–3.0 cm). The left fallopian tube was smooth; a left ovarian cyst was noted, 10×10 cm, with a smooth surface and a 1×1 cm rupture site leaking dark-brown fluid, but no active bleeding. The left cyst was densely adherent to the left fallopian tube, posterior and anterior uterine walls, and posterior leaf of the left broad ligament. Multiple right ovarian cysts (1–2 cm, smooth surface, intact capsule) were densely adherent to the posterior leaf of the right broad ligament; a millet-sized nodule and multiple yellow vesicular endometriotic lesions were found on the right fallopian tube. Both sacrouterine ligaments were contracted, with significant rigidity ("finger-like" on the left), and the left round ligament and adjacent soft tissue were firm, with marked retroperitoneal thickening. Part of the greater omentum was densely adherent to the anterior abdominal wall; yellow vesicular changes (3×3 mm) were noted along the left peritoneum near the round ligament. The bowel, mesentery, omentum, liver, spleen, and diaphragm appeared smooth with no obvious lesions; a flat black dot-like lesion (diameter ~4 mm) was observed on the peritoneum anterior to the liver, suspicious for endometriosis. During dissection, a cauliflower-like solid mass (4×3 cm) was found within the left cyst. As this was an emergency surgery, only the right adnexa was excised intraoperatively, with another elective surgery planned. Postoperative pathology: high-grade endometrioid carcinoma (endometrioid carcinoma, HEC) of the left side, with background evidence of endometriosis. Endometriosis was also observed on the serosa of the left fallopian tube. On postoperative day 1, repeat serum amylase was 75.4 U/L. Cytology from ascites was negative for tumor cells. Due to poor intestinal condition, a second staged surgery was planned.
Discussion
Gynecological acute abdomen is an emergency primarily characterized by abdominal pain, often accompanied by other symptoms. Etiologies include malformations, infections, endometriosis (endometriosis, EMs), gynecologic tumors, surgical complications, or special circumstances, with peri- or postmenopausal cases largely attributed to tumors, trauma, or dysfunctions [1]. Acute abdomen progresses rapidly; failure to promptly and accurately diagnose and treat may result in shock and life-threatening conditions, representing surgical indications. Clinical symptoms, ultrasound, imaging, laparoscopy, and laboratory tests are the fundamental diagnostic approaches for evaluating causes of acute gynecologic abdomen [2]. This patient (46 years old) presented with abdominal pain mainly due to tumor etiology. Comprehensive assessment using ultrasound, CT, laparoscopy, laboratory testing, and postoperative pathology led to the diagnosis of right-sided high-grade endometrioid carcinoma on the background of endometriosis, with endometriosis also involving the left fallopian tube serosa. Ovarian endometrioid carcinoma (OEC) is an epithelial malignancy originating in the ovary with histopathological features similar to endometrioid carcinoma of the endometrium. OEC has a significant association with endometriosis [3,4], consistent with our case. Regarding laboratory tests, CA-125 and other tumor markers are routinely assessed, but serum amylase measurement also warrants clinical attention. An elevation of amylase in serum or body fluids has been associated with various neoplasms of non-pancreatic origin. Among cancer-related studies, amylase-producing tumors predominantly include cases of lung cancer, carcinoma, and multiple myeloma [5,6,7]. Serum amylase is considered a promising tumor marker. Amylase-producing tumors may present similarly to pancreatitis, as hyperamylasemia is common among such malignancies [8]. The predominant tumor amylase phenotype is often of the salivary type [9,10]. The pancreas and salivary glands are physiological sources of amylase. Normally, amylase is present in the digestive tract; its presence in the blood may indicate pancreatic or salivary gland cell injury—such as in pancreatitis, bile duct obstruction, sialadenitis, diabetic ketoacidosis, traumatic brain injury, or abdominal aortic aneurysm—causing release of amylase into the blood and resulting in elevated serum amylase [11]. Literature review reveals Jie et al. reported a case of carcinoma with intestinal involvement and elevated amylase; the diagnosis postoperatively was high-grade serous carcinoma, with amylase levels declining after surgery and chemotherapy [12]. Logie JJ et al. also reported a case of a woman in her 50s with abdominal distension and serum amylase exceeding 30 times the upper normal limit; postoperative pathology revealed high-grade serous carcinoma with metastatic spread. After chemotherapy and surgical treatment, serum amylase and CA-125 significantly decreased [13]. Gao et al. described a 46-year-old female with elevated amylase, ruling out other causes such as pancreatitis, finally diagnosed with advanced serous carcinoma [14]. In addition, Tohya T reported a 45-year-old patient with endometrioid adenocarcinoma associated with hyperamylasemia and liver metastases [15]. O'Riordan T reported a patient with serous surface carcinoma associated with abnormal amylase elevation [16]. Collectively, most reported cases are of serous carcinoma pathology; the current case, however, represents a rare instance of high-grade endometrioid carcinoma secondary to malignant transformation of an endometriotic cyst, accompanied by elevated amylase—an uncommon pathological type in previous reports. This suggests that carcinoma accompanied by elevated amylase may present in diverse pathological types. Several studies strongly suspect that tumor cells themselves produce amylase; however, further studies are needed to validate amylase as a significant tumor marker for endometrioid-type carcinomas. This case serves as supplementary evidence for clinical consideration, highlighting that in cases of acute abdominal pain with elevated serum amylase, malignancy should be considered in addition to acute pancreatitis. Carcinoma is a common gynecologic malignancy. CA-125 and CA-199 are standard tumor markers, but their specificity is limited. In clinical practice, large cystic tumors are frequently encountered, and imaging alone may not reliably differentiate between benign and malignant lesions. A novel marker would assist in determining tumor nature. For massive tumors—especially when presenting as acute abdomen requiring emergency surgery—serum amylase testing may offer further insight into the tumor's character and may inform the feasibility of large-scale screening for carcinoma.
In this case, amylase levels declined markedly or normalized after tumor excision or effective chemotherapy, suggesting a potential role for amylase as a prognostic indicator in carcinoma. Previous studies have demonstrated that amylase serves as a useful tumor marker for follow-up in endometrioid and serous carcinoma. Regardless of treatment—whether surgical [17], radiotherapy [18], or chemotherapy [19]—serum amylase levels rapidly decline, often to within normal limits. Thus, hyperamylasemia is associated with poor clinical outcomes in carcinoma and is considered a prognostic biomarker [20]. However, prior research indicates that CA-125 and galactosyltransferase may have higher accuracy for predicting poor prognosis, and that amylase and alkaline phosphatase isoenzymes are of limited follow-up utility in carcinoma [21]. This contradicts the present findings; therefore, further investigation is required to determine whether amylase can be used as a prognostic reference index.
Acknowledgements
This study was not funded by any fund. Thanks to the Department of Pathology and the Department of Imaging of the Aerospace Center Hospital.
- Lang Jinghe (2022) Gynecologic Acute Abdomen [J]. Chinese Journal of Obstetrics and Gynecology, 57: 3.
- Habek D, Premuzić M, Cerkez Habek, (2006) J. [Syndrome of acute abdomen in gynaecology and obstetrics]. Acta Med Croatica. 60: 227-35.
- Zafrakas M, Grimbizis G, Timologou A, et al. (2014) Endometriosis and ovarian cancer risk: a systematic review of epidemiological studies [J].Front Surg, 1: 14.
- Kim HS, Kim TH, Chung HH, et al. (2014) Risk and prognosis of ovarian cancer in women with endometriosis:a metaanalysis [J] Br J Cancer, 110: 1878-90.
- Tozawa (1993) T. Electrophoretic studies of amylase produced by tumors in human sera. The physico-chemical biology Japanese journal of electrophoresis. 37: 147-53.
- Liu Meng, Chen Guoliang, Zhou Liqing (2022) A Case of Lung Cancer with Elevated Serum Amylase [J]. 43: 36-7.
- Guo Maodong, Ding Jin, Chen Yanping, et al. (2019) Multiple Myeloma with Hyperamylasemia: A Case Report and Literature Review [J]. Chinese General Practice, 22: 1372-6.
- Sichel F, Salaün V, Bar E, et al. (1994) Biological markers and ovarian carcinomas: galactosyltransferase, CA 125, isoenzymes of amylase and alkaline phosphatase. CLIN CHIM ACTA. 227: 87-96.
- Moriyama T, Tozawa T, Matsuno K. (2022) [Tumor producing amylase]. Rinsho Byori. 50: 566-70.
- Moriyama T (2008) Sialyl salivary-type amylase associated with ovarian cancer. Clin Chim Acta. 391: 106-11.
- HAO Lu, HU Liang-hao, LI Zhao-shen (2019) Clinical significance of hyperamylasemia. Academic Journal of Second Military Medical University, 40: 426-9
- Jie Y, Li J, Man CF, Fan Y. (2024) Ovarian cancer with intestinal wall invasion and hyperamylasemia:a case report. Front Oncol. 14: 1299226.
- Logie JJ, Cox M, Sharkey J, Williams A (2015) A multidisciplinary approach to an unusual cause of hyperamylasaemia.BMJCaseRep. bcr2015209780.
- Guo S, Lv H, Yan L, Rong F (2018) Hyperamylasemia may indicate the presence of ovarian carcinoma: A case report. Medicine (Baltimore). 97: e13520.
- Tohya T, Shimajiri S, Onoda C, Yoshimura T (2004) Complete remission of ovarian endometrioid adenocarcinoma associated with hyperamylasemia and liver metastasis treated by paclitaxel and carboplatin chemotherapy: a case report. Int J Gynecol Cancer. 14: 378-80.
- O'Riordan T, Gaffney E, Tormey V, Daly P (1990) Hyperamylasemia associated with progression of a serous surface papillary carcinoma. Gynecol Oncol. 36: 432-4.
- Juang, CM, Yeng, MS, Twu, NF, et al. (2000) Hyperamylasemia associated with endometroid carcinoma of the ovary. Zhonghua Yi Xue Za Zhi (Taipei). 63: 710-3.
- Zakrzewska I, Pietryńczak M (1997) The alterations in the activity of amylase and its salivary isoenzyme in the serum of patients with ovarian carcinoma, submitted to radiotherapy. Rocz Akad Med Bialymst. 42: 229-35.
- Jie Y, Li J, Man CF, et al. (2024) Ovarian cancer with intestinal wall invasion and hyperamylasemia: a case report. Front Oncol. 14: 1299226.
- Mohamed, M, Kruk, P (2016) Abstract 5080: Amylase overexpression promotes ovarian cancer invasion CANCER RES. 76: 5080.
- Sichel F, Salaün V, Bar E, et al. (1994) Biological markers and ovarian carcinomas: galactosyltransferase, CA 125, isoenzymes of amylase and alkaline phosphatase. CLIN CHIM ACTA. 227: 87-96.

Figures at a glance