Rapid Elevation of 25(OH)D Level Reduces C-Reactive Protein Level in COVID -19 Patients with Vitamin D Insufficiency
Received Date: January 15, 2023 Accepted Date: February 15, 2023 Published Date: February 18, 2023
doi: 10.17303/jfn.2023.9.104
Citation: Sintija Sauša, Svjatoslavs Kistkins, Olga Čirkova, Daina Kalniņa, Betija Jurģe (2023) Rapid Elevation of 25(OH)D Level Reduces C-Reactive Protein Level in COVID-19 Patients with Vitamin D Insufficiency.J Food Nutr 9: 1-20
Abstract
Background: Vitamin D insufficiency is a predictor of poor prognosis in COVID-19 patients. Vitamin D supplementation is related to improved clinical outcomes in terms of intensive care unit admission and death, particularly in individuals with moderate-to-severe forms of COVID-19.
Methods and findings: The placebo controlled five-day study was performed on 100 hospitalized COVID-19 patients with vitamin D insufficiency randomized into two groups. Vitamin D in the form of a sublingual sprayable microemulsion was given three times daily (daily dose 12,000 IU) to 51 patients with blood 25(OH)D levels below 30 ng/ml. 48 patients in the control group received a placebo spray in the same daily regimen. ITT test and pre-protocol analysis were used to verify the impact of true 25(OH)D level elevation.
Results: There was a statistically significant increase by 8.7 ±7.6 ng/mlin 25(OH)D level from the baseline level of 15.58 ±6.48 ng/ml in the case group. There was a strong negative correlation between changes in 25(OH)D and CRP levels in control group (p< 0,05) by using pre-protocol analysis. Individuals with moderately severe disease showed negative correlation between changes in 25(OH)D and CRP levels in both ITT and pre-protocol analysis (p< 0.05). Mild and severe cases showed no statistical significance in CRP levels. There were no statistically significant changes in ferritin and IL-6 levels in ITT and pre-protocol analysis.
Conclusion: Pre-protocol analysis revealed that high-dose vitamin D therapy was accompanied by significant decrease in CRP levels in patients with a moderate to severe illness. In contrast, ITT analysis in most cases was not able to detect statistically significant changes in all measured inflammatory markers (CRO, ferritin and IL-6) levels during the short term high-dose vitamin D spray therapy.
Keywords: COVID-19; Vitamin D Insufficiency; Sublingual Sprayable Microemulsion; C-Reactive Protein
Introduction
Vitamin D and prognosisSeveral studies suggest that vitamin D insufficiency may be a predictor of poor prognosis in COVID-19 patients [1-9]. Vitamin D supplementation, as demonstrated by R. Pal et al, is related to improved clinical outcomes in terms of ICU admission and death, particularly in individuals with moderate-to-severe COVID-19 necessitating hospitalization [1, 10]. In the study the term vitamin D insufficiency is used to describe the clinical manifestation of COVID-19 in presence of low blood vitamin D level. The observational study done by Kaufman et al showed that the rate of SARS-CoV-2 positivity is both directly and inversely linked with the amount of circulating 25(OH)D, a connection that holds across latitude, race/ethnicity, sex, and age [8]. Severity of clinical disease and mortality in COVID-19 upturns in case of vitamin D insufficiency. Vitamin D insufficiency patients have a more pronounced inflammatory response, which is accountable for the increase in morbidity and mortality [9].
Low 25(OH)D levels and prognosisThe clinical studies in individuals with a 25(OH)D level of less than 20 ng/ml demonstrated an increased risk of COVID-19 compared to individuals with a 25(OH)D level of 30–34 ng/ml [3-8]. An extensive retrospective cohort study published by Oristrell et al showed that those who received cholecalciferol or calcifediol supplementation and achieved serum 25(OH)D levels of 30 ng/ml had superior COVID-19 treatment results [11].
Vitamin D and contradictory findingsNumerous research is underway to determine the benefits of vitamin D on individuals with COVID-19 [13]. However, researchers produced contradictory findings. After high-dose intramuscular D3 treatment in the early period of COVID-19, no difference in duration of hospital stay, endotracheal intubation, or death rates in critical patients in the ICU has been seen in the study of Güven and Gültekin [14]. Another study had similar findings, showing no link between 25(OH)D levels and COVID-19 severity or death in 341,484 UK Biobank individuals [15]. The Mendelian randomization study by Butler-Laporte et al found no evidence to indicate a relationship between 25(OH)D levels and COVID-19 susceptibility, severity, or hospitalization in a two-sample MR investigation [16]. In the systematic review and meta-analysis done by Basstne and colleagues, where they analyzed 31 peer-reviewed observational studies, found that while the existing data to date, derived primarily from low-quality observational studies, may be interpreted as indicating a tendency toward a link between low blood 25(OH)D levels and COVID-19- related health outcomes, this link was not found to be statistically significant. A possible preventive effect of calcifediol supplementation on COVID-19-related ICU admissions could be found [17].
Vitamin D and CRPAt the same time, it is worth mentioning contradictory findings as well in response to inflammatory markers. Higher levels of CRP during COVID-19 have been found to have greater complications and a more severe case of COVID-19 overall [18]. Several studies, which are in line with our study, have shown an inverse association between vitamin D and CRP [19-20]. One study showed inverse correlation between vitamin D and CRP, but more importantly highlighted that said inverse correlation isstronger in patients who have inflammatory diseases compared to patients who do not have those [20]. Yet, another study shows that there is not a noticeable inverse relation between vitamin D supplementation and inflammatory biomarkers specifically in asymptomatic adults [21]. The study where vitamin D and inflammatory marker CRP were compared in people aged 60 years and older, showed that CRP levels were lower in patients with overall higher 25(OH)D levels. These findings with vitamin D intervention study could help answer the question if vitamin D decreases CRP levels or if CRP decreases 25(OH)D levels [22].
A single-center study studying COVID-19 and inflammatory markers showed that in the first wave of COVID-19 inflammatory markers were higher than in the second wave most likely due to changed guidelines on treating patients [23].
Vitamin D and ferritin and IL-6Vitamin D may affect ferritin and IL-6 levels as it influences iron metabolism and erythropoiesis. Iron is one of the essentials for vitamin D synthesis [24]. There is a probability that vitamin D down regulates mRNA expression of hepcidin levels. It’s believed that antimicrobial hepcidin peptides influence iron absorption and release through activating and suppressing ferroprotein which is a cellular iron exporter [25]. In a systematic review done by Arabi et al that included a total of 14 reports and 1385 individuals with mean baseline of 25(OH)D levels ranging between 10 and 30 ng/ml observed that vitamin D supplementation in 11 of the included studies did not improve ferritin concentrations overall. The study concluded that vitamin D supplements had no significant effect on ferritin levels while having positive effects on transferrin saturation and iron status [26]. However, a prospective observational study by Singh et al that included 343 COVID-19 positive asymptomatic and 59 ICU patients, argued that inflammatory markers such as serum ferritin and IL-6 were significantly elevated in critically ill patients and high levels of 25(OH)D correlate with reduced levels of IL-6[27]. In another cross-sectional study by Teama et al that included 124 patients diagnosed with COVID-19 a negative correlation was observed between vitamin D and ferritin. Study concluded that vitamin D deficiency significantly and negatively correlated with ferritin. This indicates that vitamin D might have a beneficial role on the systemic inflammatory state of COVID-19 [28].
Materials and Methods
The aim of this study was to investigate the impact of the high dose vitamin D supplementation on laboratory markers of systemic inflammation such as CRP, ferritin and IL-6 in hospitalized patients with COVID-19. It was performed at the Pauls Stradiņš Clinical University Hospital (PSCUH) from February till July 2021 (ClinicalTrials.gov Identifier: NCT05502068). The blood samples were analyzed at the Joint Laboratory of the hospital. The study protocol was approved by the local ethics committee. A written consent was obtained a day before intervention began. Patients with confirmed SARS-CoV2 infection (PCR) were randomly divided into two groups: the intervention group received vitamin D in the form of a sublingual sprayable microemulsion (LYL love your life® sunD3 LYLmicro™) of 4,000 IU three times daily after breakfast, lunch and dinner; the control group received the same regiment of placebo spray.
A total of 99 inpatients were selected for the study, 48 patients in the vitamin D group and 51 in the control group. All patients received standard care for COVID-19 and existing comorbidities (diabetes, arterial hypertension, etc) according to hospital approved protocol. The primary outcome was defined as the change in the level of inflammatory markers and the severity of the disease.
Only those patients whose 25(OH)D level was below 30 ng/ml were included in the study. Patients with mental health problems, eGFR ≤30 ml/min, 25(OH)D level ≥ 30 ng/ml, pregnant women and any other illness or condition that the researcher deemed may interfere with the results were excluded. All individual patient data obtained during the study was coded and was available only to the study participants. The duration of the pharmaceutical intervention was 5 days.
PopulationDisease severity was defined by the blood oxygen saturation level. Mild clinical manifestation was characterized by SpO2≥94%, moderate 90%≤SpO2< 94% and severe - SpO2< 90%. Age, BMI, GFR, vitamin D and COVID-19 severity were considered as randomization parameters. 25(OH)D, ferritin and IL-6 were analyzed by chemiluminescence immunoassay (Atellica IM 1300 Analyzer, Siemens) while C-reactive protein (N: 0–5 mg/l) was processed by a turbidimetric method using Atellica CH 930 Analyzer, Siemens.
Statistical analysisPatient data were analyzed in two runs: including all patients (Intention-to-Treat (ITT) analysis) and stratifying patients according to the increase in 25(OH)D (pre-protocol analysis for 25(OH)D elevation effect: responders/noresponders).
Shapiro – Wilk (n < 50 in the group) or Kolmogorov- Smirnov (n > 50 in the group) tests were used to evaluate whether the data follows the normal distribution with statistically significant PN.
The difference between groups with a nominal distribution was determined by the chi-squared (χ2) criterion method. A statistically significant difference between groups was considered at Pχ < 0.05. Cramer's V was chosen to determine the relationship between nominal data: above 0.10 - weak relationship, above 0.30 - medium, and above 0.50 - strong relationship.
T-test (statistical confidence PT ) was used to determine differences between group numerical values (mean or median) if data from both groups were normally distributed. The Mann-Whitney test (PMV) was applied if at least one group was not normally distributed. ANOVA test (PF) or Kruskal-Wallis (PKV) test was used if more than two groups were compared.
The relationship between nominal and numerical indicators was determined using the eta (η) indicator: up to 0.2, the relationship was considered very weak; from 0.2 to 0.4 weak; from 0.4 to 0.7 moderately tight; from 0.7 to 0.9 tight; above 0.9 very tight, and 1.00 perfect.
In all tests, the statistically significant difference between groups was considered when P < 0.05. All tests were performed using IBM SPSS Statistics version 25.0.
Results
The mean age in the placebo group was 67.1 ± 13.5 years (Table S1), with an interval of 34 to 88 years. The confidence interval (CI 95%) for the mean was 59.2 to 67.0 years. The mean age in the vitamin D group was 63.1 ± 13.9 years (Table S1), with an interval of 34 to 90 years. Both groups were in normal distribution according to the specific indicator (Table S1, supplementary).
In the case of the B group only responders (true case group), and in the A group only nonresponders (true placebo group) were selected. Inclusion/exclusion was determined by statistically significant changes in 25(OH)D levels according to the coefficient of variation (CV) of the laboratory equipment (CL5I DocumentEPQ5-A3). Those participants whose 25(OH)D level on the sixth day was higher than on the first day were considered to be responders (equation 1). The equation used for inclusion was:
CvitD2 > CvitD1+ CV (responders)(1)
CvitD2< CvitD1+CV (nonresponders)
Where
CvitD2 - level of 25(OH)D on the 6th day
CvitD1 - level of 25(OH)D on the 1st day
CV - coefficient of variation
90.2% of all participants in group B fit the criteria of the true case group (Table 1). Whereas only 34 participants (70.7%) in the placebo group showed no increase in 25(OH)D levels in the blood.
Severity of COVID-19 casesThe distribution of patients in both groups by severity of disease was statistically similar (Table 2). The difference between groups was not statistically significant (p=0.06).
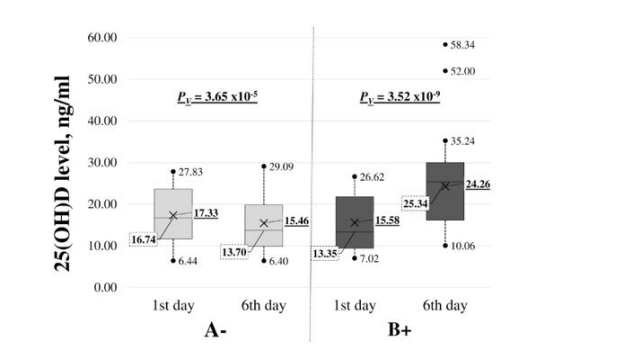
Changes in 25(OH)D levelsThe level of 25(OH)D increased on average by 8.69 ± 7.63 ng/ml in group B, also the median in group B increased by 5.82 ng/ml with an IQR (interquartile range) of 8.37 ng/ml indicates an increase in value. The baseline of 25(OH)D levels in true case group was 15.58 ± 6.48 mg/l (median 13.35 mg/l, IQR 12.56 mg/l), while in true placebo group – 17.33 ± 6.71 mg/l (median 16.74 mg/l, IQR 12.15 mg/l). The change in 25(OH)D levels in group B over 5 days was considered statistically significant PV = 2.72 x10-14 (Table 3 and Figure 1). However, it should be noted that the delta values of 25(OH)D level in 5 patients (9.8%) from group B didn't rise above the coefficient of validation (Table S2 25(OH)D levels in ITT analysis with all patients).
In the placebo group, 25(OH)D level decreased on average by -1.87 ± 1.97 ng/ml. The resulting mean change was also statistically significant.
There were no statistical differences in delta values of 25(OH)D level comparing disease-severity groups (Table 4). However, the largest increase in 25(OH)D level was observed in patients with a severe form of COVID-19. The median value in the severe group was twice as high than in the mild group
Changes in C - reactive protein levelsThe decrease of C-reactive protein (CRP) was observed in both groups. The baseline of CPR levels in true case group was 98.11 ± 74.55 mg/l (median 76.47 mg/l, IQR 104.86 mg/l), while in true placebo group – 63.40 ± 45.45 mg/l (median 52.57 mg/l, IQR 77.78 mg/l). However, the delta indicators were not in a normal distribution, thus the medians were compared (Figure 2).
The mean delta values of CRP level before and after the treatment (Table 5) in the B vial group is significantly higher than in the A vial group: -52.98 ± 82.34 mg/l vs. -18.77 ± 54.17 mg/l. The differences between two groups were statistically significant (p < 0.05). No statistically significant differences were found between the groups when we analyzed CRP changes or deltas by ITT analysis (Table S3).
No statistically significant differences in delta values of the CRP level in mild and severe groups were found (Table 6). However, the decrease in CRP level in a moderate severe group was statistically significant compared to the placebo group (p < 0.05) in ITT analysis (Table S4) and also in pre-protocol analysis (Table 6). In the ITT analysis, the difference between the groups was less significant or smaller than in the pre-protocol analysis.
Therefore, it can be concluded that vitamin D in a moderate-severe COVID-19 patient contributes to the reduction of CRP level.
Changes in ferritin levelsThe response of other inflammatory markers such as ferritin and IL-6 were also investigated. The ferritin level in the placebo group decreased on average, but in the B group, the indicator increased on average (Table 7). It should be noted that there was no normal distribution in group B thus the median values were compared. Despite the rise in average ferritin value, the median delta value in B group indicated a decrease of ferritin level. The differences between true and placebo groups were not statistically significant, and obtained data also showed a very large distribution in blood levels of ferritin. (supplementary material, Tables S5-S6)
The ferritin delta values between the two groups were also not statistically significant applying a nonparametric data analysis method. In addition, there was a high dispersion in both groups.
Changes in IL-6 levelsA decrease of IL-6 was observed in both groups, but the delta value was not in the normal distribution, thus non-parametric data methods were applied. There was no statistically significant difference in delta values between the groups (P> 0.05). The wide range of delta values in both groups, especially in group A (Table 8) may have contributed to such results.
A possible explanation of such distribution could be explained by the different baseline levels, which could again be tested by analysis in different groups of patients with disease severity. However, there was also no statistically significant difference between the severity groups of patients.
It should be noted that there was one patient in each group who showed significantly higher IL-6 levels compared to others (supplementary materials Tables S7- S8). The IL-6 levels in the patients of A and B groups were 1552.10 pg /ml and 1766.50 pg/ml respectively. Due to this, two patients were excluded from analysis.
Discussion
Our data showed that large doses of vitamin D may effectively decrease CRP level in vitamin D deficient COVID-19 patients during just 5-day-intervention compared to those who haven’t received vitamin D. Literature analysis shows that vitamin D decreases synthesis of proinlammatory markers in macrophages by increasing synthesis of anti-inflammatory markers in T-cells (Th1). As a proinflammatory marker IL-6 impacts synthesis of CRP in liver thus, as a result, vitamin D decreases CRP via decrease of IL-6 synthesis in macrophages [55]. In our study we haven’t seen the statistically significant decrease of IL-6. We haven’t also observed a significant decrease of IL-6 synthesis in both groups. The study results don’t exclude the impact of rapid decrease of IL-6 values in Group B at the first day of intervention thus a dynamic evaluation is crucial. For these reason additional studies are necessary to observe the dynamic changes in IL-6 in blood induced by vitamin D. The increase of vitamin D level by average 8 ng/ml in blood was accompanied by decrease of CRP level in patients with moderately severe clinical manifestation of the disease. Exogenous vitamin D toxicity (25(OH)D >150 ng/ml) is a serious condition, but it is relatively rare. Cholecalciferol supplementation is less likely to produce excessively high values of 25(OH)D because it is the gold standard for the equivalence between international units and molecular mass. The conversion of vitamin D3 to calcifediol is related to many factors, including liver function and storage in adipose tissues. The conversion of vitamin D3 to calcifediol is related to many factors, including liver function and vitamin D storage in the adipose tissues. Our intervention was significantly shorter compared to the study [56] where the high-dose cholecalciferol intervention didn't highly accelerate the levels of 25(OH)D during the first week of interventions. Study results showed that a dose of 100 000 IU per 2 weeks increased the mean 25(OH)D levels by 10 ng/ml in the first week. We haven't used calcifediol in this study due to a high risk of vitamin D excess, promoting undesired side effects (such as kidney failure). Halftime of vitamin D3 is 24 h, while calcifediol is 2-3 weeks. In the study we have defined the true case group to investigate the effect of the 25(OH)D level increase on inflammatory markers compared to those, who haven’t received the supplement. B group patients whose 25(OH)D levels did not rise above the coefficient of variation were excluded. It was necessary to analyze the effect of the increase of 25(OH)D levels on inflammatory markers. The true placebo group was defined to exclude an uncontrolled increase of 25(OH)D levels from the food supplements or other exogenous sources of vitamin D in A group.
Based on the results, one might assume that only the moderate severity of COVID-19 has the most positive outcome. However, it is worth mentioning that in most cases mild clinical manifestation of the disease is selflimiting, thus the intervention with vitamin D might not cause a statistically significant improvement. The severe form of COVID-19 is not significantly affected by vitamin D, thus showing that the supplementation with vitamin D3 should be investigated according to other comorbidities and drug interactions.
For instance, a similar randomized placebocontrolled study (SHADE) where participants have been randomized to receive 60,000 IU of cholecalciferol daily for 7 days did not show any effect on IL-6, ferritin, and CRP levels [54]. Comparing two studies we have seen the differences in dosing and patients’ recruitment criteria. During the SHADE study, the level of vitamin D has been increased by ~40 ng/ml on average, compared to the 8.6 ng/ml increase in our study. Such a change of 25(OH)D level has been achieved by 60,000 IU/daily interventions, compared to our intervention (12,000 IU/daily). It is worth mentioning that the included population in the SHADE study didn’t have a significantly high level of CRP to study the impact of vitamin D. Secondly, the population was significantly lower compared to our study. Thirdly, there was no stratification of patients by disease severity compared to the study provided in our research site. Additional survival study, however, is necessary to verify the most appropriate therapy for severe COVID-19 form
The total vitamin D dose in our study was at 72,000 IU which was much lower compared to other highdose interventions [12, 29, 30, 54].
The study with a single oral bolus dosage of 100 000 IU of cholecalciferol did not show any improvement in the outcomes of COVID-19. However, this study shows that a lower D vitamin level at admission time was associated with higher pulmonary involvement and a higher rate of ICU admission. According to this study, the level of 25(OH)D > 25 ng/ml was associated with better outcomes [29]. Suggesting this it may be helpful to provide screening blood tests of vitamin D to select patients with a higher risk and start immediate supplementary treatment with cholecalciferol.
Studies with a single high bolus dose of 200,000 IU of cholecalciferol did not show any effect on mortality, length of hospital stay, hospital discharge, ICU admission, and rates of mechanical ventilation, despite achieving sufficient 25(OH)Dlevels ≥30 ng/mL [12]. However, studies with a larger single dose of 400,000 IU compared to the standard dose of 50,000 IU in older adults within 72 hours after the diagnosis of COVID-19 were associated with reduced overall mortality on day 14. The increase of 25(OH)D levels on day 7 was significantly higher in the high-dose group on average at 38 ng/ml compared to 8.4 ng/ml in the standard-dose group [30]. There are some trials with smaller population groups receiving smaller doses of cholecalciferol for 2 weeks [31, 32].
In another multicenter randomized controlled trial patient with mild to moderate COVID-19 and suboptimal vitamin D were receiving 5000 IU and 1000 IU of oral cholecalciferol daily for 2 weeks. This study reports that a 5000 IU daily oral vitamin D3 supplementation significantly shortens recovery time to symptoms, but shows no significant differences in ICU, mortality events, and days to discharge. Within-group comparisons also showed a significant decrease in IL-6 levels over time in both groups but were not clinically significant in betweengroup comparisons. However, the increase in vitamin D level was statistically significant on average at only 3.6 ng/ml. It is worth mentioning that 47% of patients also received vitamin C supplementation [31].
In a small randomized clinical trial, 22 outpatients with a mild disease course received oral supplementation of 10,000 IU daily of vitamin D3 for 14 days. The administered supplementation was sufficient to increase total 25(OH)D serum levels significantly on day 14 (28.2 ng/mL on average). And the intervention group had fewer symptoms on the 7th and 14th day of follow-up [32]. According to research conducted in Brazil (200,000 IU/5000 g) [12] and in France (80,000 IU/2000 g) [33], using vitamin D3 during COVID-19 hospitalization does not lessen the risk of death. Additional studies are necessary to analyze mortality in patient groups who have received vitamin D according to our protocol and to find out the optimal dosage.
We provide such a kind of placebo-controlled prospective study in order to escape several limitations of other studies e.g., in the case of observational studies the randomization was limited due to clinical judgments of healthcare providers according to patients' clinical condition [10]. In the case of prospective studies, there is a limited number of placebo-controlled trials. There are limitations in statistical analysis in such experiments, where only the intervention has been taken into account escaping the true changes in blood 25(OH)D levels, which are the key factor measuring the changes in inflammatory markers [12, 13, 53].
According to the result of this study vitamin D did not produce any significant changes in ferritin and IL-6 levels. Other studies support current findings. A retrospective study by Feld et al., for example, showed that despite greater ferritin levels being related to all-cause mortality, ferritin could not predict numerous critical outcomes, including death, with any certainty [40]. Some researchers suggested that ferritin levels could be used to predict the severity of COVID-19 [34-52]. On the other hand, a retrospective cohort study that included 158 confirmed COVID-19 patients showed that COVID-19 patients with low serum iron levels were prone to suffer from a severe disease and multiple–organ damage [52]. In our case, IL-6 levels, similarly to ferritin levels, did not show any statistically significant changes. A supporting study by Fernandes et al indicated that a single high dose of 200,000 IU vitamin D had no effect on cytokines, chemokines, and growth factors in patients with moderate to severe COVID-19 [53].
Conclusions
There was a rapid elevation of circulating 25- hydroxyvitamin D levels in hospitalized vitamin D deficient COVID-19 patients treated by high-dose sublingual sprayable vitamin D microemulsion for five days. Preprotocol analysis revealed that high-dose vitamin D therapy was accompanied by significant decrease in CRP levels in patients with a moderate to severe illness. In contrast, ITT analysis was not able to detect statistically significant changes in all measured inflammatory markers (CRO, ferritin and IL-6) levels during the short term high-dose vitamin D spray therapy. Additional studies must be done to verify the correlation between vitamin D intervention and mortality outcomes in COVID-19 patients.
Declarations
Author contributions
All authors contributed and reviewed the manuscript. VP, KG, SS participated in study design, DK, BJ, KI, ZS, AF, LK participated in a clinical trial, TT, REZ, OC, VB, SKa participated in data curation and original draft preparation, VP and KG, IT, SKi, triple checked all data and provided statistical support. All authors read and approved the final manuscript.
Funding
This research was funded by SIA Pharm&Med (LYL love your life® sunD3 LYLmicro™), grant number 2021sunD3LYLmicroTMSARS-CoV-2
Ethical Approval
The study was conducted in accordance with the declaration of Helsinki and approved by the Ethics Committee of Pauls Stradiņš Clinical University Hospital (code of approval: 170621-24L; date of approval: 25/03/2021). ClinicalTrials.gov Identifier: NCT05502068 (date of approval: 16/08/2022)
Consent to participate
Informed consent was obtained from all subjects involved in the study.
Consent for publication
All authors approved the manuscript and gave their consent for publication.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
Acknowledgments
We would like to thank all COVID-19 patients who participated in this clinical study.
Competing interests
The authors declare no competing interests.
- Pal R, Banerjee M, Bhadada SK et al. (2022) Vitamin D supplementation and clinical outcomes in COVID-19: a systematic review and meta-analysis. J Endocrinol Invest 45: 53-68.
- Liu G, Hong T, Yang J (2020) A Single Large Dose of Vitamin D Could Be Used as a Means of Coronavirus Disease 2019 Prevention and Treatment. Drug Des. Devel. Ther 14: 3429-34.
- Ling SF, Broad E, Murphy R, Pappachan JM, PardesiNewton S et al. (2020) Kong, M.-F.; Jude, E.B. High-Dose Cholecalciferol Booster Therapy is Associated with a Reduced Risk of Mortality in Patients with COVID-19: A Cross-Sectional Multi-Centre Observational Study. Nutrients 12: 3799
- Xu Y, Baylink DJ, Chen CS, Reeves ME, Xiao J et al. (2020) The importance of vitamin d metabolism as a potential prophylactic, immunoregulatory and neuroprotective treatment for COVID-19. J Transl Med 18: 322.
- Radujkovic A, Hippchen T, Tiwari-Heckler S, Dreher S, Boxberger M et al. (2020) Merle, U. Vitamin D Deficiency and Outcome of COVID-19 Patients. Nutrients 12: 2757.
- Demir M, Demir F, Aygun H (2021) Vitamin D deficiency is associated with COVID-19 positivity and severity of the disease. J Med Virol 93: 2992-9.
- Sulli A, Gotelli E, Casabella A, Paolino S, Pizzorni C et al. (2023) Alessandri, E.; Grosso, M.; Ferone, D.; Smith, V.; Cutolo, M. Vitamin D and Lung Outcomes in Elderly COVID-19 Patients. Nutrients 13: 717.
- Kaufman HW, Niles JK, Kroll MH, Bi C, Holick MF (2020) SARS-CoV-2 positivity rates associated with circulating 25-hydroxyvitamin D levels. PloS one 15: e0239252.
- Demir AD, Durmaz ZH (2021) A Comparison of Vitamin D Deficiency with Neutrophil Lymphocyte Ratio and CRP Levels in Covid-19 Patients. Med Sci Discov 8: 306-9.
- Alcala-Diaz JF, Limia-Perez L, Gomez-Huelgas R, Martin-Escalante MD, Cortes-Rodriguez B et al. (2021) Calcifediol Treatment and Hospital Mortality Due to COVID-19: A Cohort Study. Nutrients 13: 1760.
- Oristrell J, Oliva JC, Casado E, Subirana I, Domínguez D et al. (2022) Vitamin D supplementation and COVID-19 risk: a population-based, cohort study. J. Endocrinol. Invest 45: 167-79.
- Murai IH, Fernandes AL, Sales LP, Pinto AJ, Goessler KF et al. (2021) Effect of a Single High Dose of Vitamin D3 on Hospital Length of Stay in Patients with Moderate to Severe COVID-19: A Randomized Clinical Trial. JAMA 325: 1053-60.
- da Rocha AP, Atallah AN, Aldrighi JM, Pires A et al. (2021) Dos Santos Puga, M. E.; Pinto, A. Insufficient evidence for vitamin D use in COVID-19: A rapid systematic review. Int. J. Clin. Pract 75: e14649.
- Güven M, Gültekin H (2021) The effect of high-dose parenteral vitamin D3 on COVID-19-related inhospital mortality in critical COVID-19 patients during intensive care unit admission: an observational cohort study. Eur J Clin Nutr 75: 1383-8.
- Hastie CE, Pell JP, Sattar N (2021) Vitamin D and COVID-19 infection and mortality in UK Biobank. Eur. J. Nutr 60: 545-8.
- Butler-Laporte G, Nakanishi T, Mooser V, Morrison DR, Abdullah T et al. (2021) Adeleye, O.; Mamlouk, N.; Kimchi, N.; Afrasiabi, Z.; Rezk, N.; et al. Vitamin D and COVID-19 susceptibility and severity in the COVID-19 Host Genetics Initiative: A Mendelian randomization study. PLoS medicine 18: e1003605.
- Bassatne A, Basbous M, Chakhtoura M, El Zein O, Rahme M et al. (2021) El-Hajj Fuleihan, G. The link between COVID-19 and Vitamin D (VIVID): A systematic review and meta-analysis. Metab. Clin. Exp 119: 154753,
- Sadeghi-Haddad-Zavareh M, Bayani M, Shokri M, Ebrahimpour S, Babazadeh A et al. (2021) C-Reactive Protein as a Prognostic Indicator in COVID-19 Patients. Interdiscip Perspect Infect Dis 5557582.
- Tao RX, Zhou QF, Xu ZW, Hao JH, Huang K et al. (2015) Inverse Correlation between Vitamin D and CReactive Protein in Newborns. Nutrients 7: 9218-28.
- Kruit A, Zanen P (2016) The Association between Vitamin D and C-Reactive Protein Levels in Patients with Inflammatory and Non-Inflammatory Diseases. Clin. Biochem 49: 534-37.
- Amer M, Qayyum R (2012) Relation between Serum 25-Hydroxyvitamin D and C-Reactive Protein in Asymptomatic Adults. Am. J. Cardiol 109: 226-30,
- Aspell N, Laird E, Healy M, Shannon T, Lawlor B et al. (2019) The Prevalence and Determinants of Vitamin D Status in Community-Dwelling Older Adults: Results from the English Longitudinal Study of Ageing (ELSA). Nutrients 11: 1253
- Asghar MS, Yasmin F, Ahsan MN, Alvi H, Taweesedt P et al. (2021) Comparison of First and Second Waves of COVID-19 through Severity Markers in ICU Patients of a Developing Country. J. Community Hosp. Intern. Med. Perspect 11: 576-84.
- Azizi-Soleiman F, Vafa M, Abiri B, Safavi M (2016) Effects of Iron on Vitamin D Metabolism: A Systematic Review. Int J Prev Med 7: 126.
- Zughaier SM, Alvarez JA, Sloan JH, Konrad RJ, Tangpricha V (2014) The Role of Vitamin D in Regulating the Iron-Hepcidin-Ferroportin Axis in Monocytes. J. Clin. Transl. Endocrinol 1: e19-e25.
- Arabi SM, Ranjbar G, Bahrami LS, Vafa M, Norouzy A (2020) The Effect of Vitamin D Supplementation on Hemoglobin Concentration: A Systematic Review and MetaAnalysis. J. Nutr 19.
- Singh RP, Chauhan K, Tripathi A, Chaudhary E (2021) Vitamin D and COVID-19 - Let's Explore the Relationship! Maedica 16: 628-33,
- Teama MAEM, Abdelhakam DA, Elmohamadi MA, Badr FM (2021) Vitamin D Deficiency as a Predictor of Severity in Patients with COVID-19 Infection. Sci Prog 104: 368504211036854,
- Cannata-Andía JB, Díaz-Sottolano A, Fernández P, Palomo-Antequera C, Herrero-Puente P et al. (2022) A Single-Oral Bolus of 100,000 IU of Cholecalciferol at Hospital Admission Did Not Improve Outcomes in the COVID-19 Disease: The COVID-VIT-D—a Randomised Multicentre International Clinical Trial. BMC Med 20.
- Annweiler C, Beaudenon M, Gautier J, Gonsard J, Boucher S et al. (2022) Chapelet, G.; Darsonval, A.; Fougère, B.; Guérin, O.; Houvet, M.; et al. High-Dose versus StandardDose Vitamin D Supplementation in Older Adults with COVID-19 (COVIT-TRIAL): A Multicenter, Open-Label, Randomized Controlled Superiority Trial. PLOS Med 19: e1003999.
- Sabico S, Enani MA, Sheshah E, Aljohani NJ, Aldisi DA et al. (2021) Alotaibi, N. H.; Alshingetti, N.; Alomar, S. Y.; Alnaami, A. M.; Amer, O. E.; et al. Effects of a 2-Week 5000 IU versus 1000 IU Vitamin D3 Supplementation on Recovery of Symptoms in Patients with Mild to Moderate Covid-19: A Randomized Clinical Trial. Nutrients 13: 2170.
- Sánchez-Zuno GA, González-Estevez G, MatuzFlores MG, Macedo-Ojeda G, Hernández-Bello J et al. (2021) Mora-Mora, J. C.; Pérez-Guerrero, E. E.; García-Chagollán, M.; Vega-Magaña, N.; Turrubiates-Hernández, F. J.; et al. Vitamin D Levels in COVID-19 Outpatients from Western Mexico: Clinical Correlation and Effect of Its Supplementation. J. Clin. Med 10.
- Annweiler C, Hanotte B, Grandin de l'Eprevier C, Sabatier JM, Lafaie L et al. (2020) Célarier, T. Vitamin D and survival in COVID-19 patients: A quasi-experimental study. J. Steroid Biochem. Mol. Biol 204: 105771,
- Gómez-Pastora J, Weigand M, Kim J, Wu X, Strayer J et al, (2020) Hyperferritinemia in Critically Ill COVID-19 Patients – Is Ferritin the Product of Inflammation or a Pathogenic Mediator? Clin. Chim. Acta; Int. j. clin. chem. lab. med 509: 249-51,
- Lv Y, Chen L, Liang X, Liu X, Gao M et al. (2020) Wang, Q.; Wei, Q.; Liu, L. Association between Iron Status and the Risk of Adverse Outcomes in COVID-19. Clin Nutr 40: 3462-9,
- Kappert K, Jahić A, Tauber R (2020) Assessment of Serum Ferritin as a Biomarker in COVID-19: Bystander or Participant? Insights by Comparison with Other Infectious and Non-Infectious Diseases. Biomarkers 1-36,
- Lin Z, Long F, Yang Y, Chen X, Xu L et al. (2020) Serum Ferritin as an Independent Risk Factor for Severity in COVID-19 Patients. J. Infect 81: 647-79.
- Dahan S, Segal G, Katz I, Hellou T, Tietel M et al. (2020) Ferritin as a Marker of Severity in COVID-19 Patients: A Fatal Correlation. Isr. Med. Assoc. J 22: 494-500.
- Ahmed S, Ansar Ahmed Z, Siddiqui I, Haroon Rashid N, Mansoor M et al. (2021) Evaluation of Serum Ferritin for Prediction of Severity and Mortality in COVID-19- a Cross Sectional Study. Ann. Med. Surg 63: 102163.
- Feld J, Tremblay D, Thibaud S, Kessler A, Naymagon L (2020) Ferritin Levels in Patients with COVID‐19: A Poor Predictor of Mortality and Hemophagocytic Lymphohistiocytosis. Clin Lab Haematol 42: 773-9.
- Tural Onur S, Altın S, Sokucu SN, Fikri Bİ, Barça T et al. (2020) Could Ferritin Level Be an Indicator of COVID‐19 Disease Mortality? J. Med. Virol 93: 1672-7.
- Taneri PE, Gómez-Ochoa SA, Llanaj E, Raguindin PF, Rojas LZ et al. (2020) Anemia and Iron Metabolism in COVID-19: A Systematic Review and Meta-Analysis. Eur. J. Epidemiol 35: 763-73,
- Bolondi G, Russo E, Gamberini E, Circelli A, Meca MCC et al. (2020) Iron Metabolism and Lymphocyte Characterisation during Covid-19 Infection in ICU Patients: An Observational Cohort Study. World J. Emerg. Surg 15: 41.
- Deng F, Zhang L, Lyu L, Lu Z, Gao D et al. (2021) Increased Levels of Ferritin on Admission Predicts Intensive Care Unit Mortality in Patients with COVID-19. Medicina Clinica (English Ed.) 156: 324-31.
- Banchini F, Cattaneo GM, Capelli P (2021) Serum Ferritin Levels in Inflammation: A Retrospective Comparative Analysis between COVID-19 and Emergency Surgical Non-COVID-19 Patients. World J. Emerg. Surg 16.
- Colafrancesco S, Alessandri C, Conti F, Priori R (2020) COVID-19 Gone Bad: A New Character in the Spectrum of the Hyperferritinemic Syndrome? Autoimmun Rev 19: 102573.
- Ruscitti P, Berardicurti O, Di Benedetto P, Cipriani P, Iagnocco A et al. (2020) Severe COVID-19, Another Piece in the Puzzle of the Hyperferritinemic Syndrome. An Immunomodulatory Perspective to Alleviate the Storm. Front. Immunol 11: 1130.
- Perricone C, Bartoloni E, Bursi R, Cafaro G, Guidelli GM et al. (2020) COVID-19 as Part of the Hyperferritinemic Syndromes: The Role of Iron Depletion Therapy. Immunol. Res 1-12.
- Zhou F, Yu T, Du R, Fan G, Liu Y et al. (2020) Clinical Course and Risk Factors for Mortality of Adult Inpatients with COVID-19 in Wuhan, China: A Retrospective Cohort Study. The Lancet 395: 1054-62,
- Ruscitti P, Giacomelli R (2020) Ferritin and Severe COVID-19, from Clinical Observations to Pathogenic Implications and Therapeutic Perspectives. The Israel Medical Association journal: IMAJ 22: 516-8.
- Wu C, Chen X, Cai Y, Xia J, Zhou X et al. (2020) Risk Factors Associated with Acute Respiratory Distress Syndrome and Death in Patients with Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA 180.
- Kell DB, Pretorius E (2014) Serum Ferritin Is an Important Inflammatory Disease Marker, as It Is Mainly a Leakage Product from Damaged Cells. Metallomics: integrated biometal science 6: 748-73.
- Fernandes AL, Murai IH, Reis BZ, Sales LP, Santos MD et al. (2022) Effect of a Single High Dose of Vitamin D3 on Cytokines, Chemokines, and Growth Factor in Patients with Moderate to Severe COVID-19. Am. J. Clin. Nutr 115: 790-8.
- Rastogi A, Bhansali A, Khare N, Suri V, Yaddanapudi N et al. (2022) Short term, high-dose vitamin D supplementation for COVID-19 disease: a randomised, placebo-controlled, study (SHADE study). Postgraduate medical journal 98: 87-90.
- Calton EK, Keane KN, Newsholme P, Soares MJ (2015) The Impact of Vitamin D Levels on Inflammatory Status: A Systematic Review of Immune Cell Studies. PloS one 10: e0141770.
- Fassio A, Adami G, Rossini M, Giollo A, Caimmi C et al. (2020) Pharmacokinetics of Oral Cholecalciferol in Healthy Subjects with Vitamin D Deficiency: A Randomized Open-Label Study. Nutrients 12: 1553.

Tables at a glance
Figures at a glance