Long Non-Coding RNA H19 Enhanced Osteogenic Differentiation of BMSCs may Induce Antihyperalgesia Through Activating NF-Kappab Signaling Pathway
Received Date: February 08, 2026 Accepted Date: February 19, 2026 Published Date: February 25 2026
doi:10.17303/jgdt.2026.1.104
Citation: Qing Li, Hui Long, Wei Gao, Li Yuan, Zhengguang Wang (2026) Long Non-Coding RNA H19 Enhanced Osteogenic Differentiation of BMSCs may Induce Antihyperalgesia Through Activating NF-Kappab Signaling Pathway. J Gen Disor Thera 1: 104
Abstract
Ostealgia is a serious symptom in bone malignant diseases and often caused by bone loss. Reduction of bone loss or even induction of bone generation is a solution of ostealgia and has lots of benefits to bone cancer patients. Long non-coding RNA H19 might be a potential candidate to regulate BMSC cells osteogenic differentiation. This study was designed to explore how H19 induce osteogenic signaling pathway and bone generation. BMSC cells were cultured and osteogenic induction was performed. Lnc-RNA H19 was either overexpressed or knockdown. Alizarin red was performed to test cell mineralization. Flow cytometry and MTT assay were performed to study cell viability. Real time PCR was performed to examine gene expression level. Mineralization cores were obviously increased after osteogenic induction and H19 overexpressed. Viability of BMSC cells were upregulated by overexpression of H19 but inhibited by knockdown of H19. 3 osteogenic markers, ALP, Runx2 and Col 1 were induced by H19. Moreover, BMP2, NF-κB and Wnt5A signal were activated by H19. In conclusion, Lnc-RNA H19 induced BMSC cells osteogenic differentiation through BMP2, NF-κB and Wnt5A signal by upregulated ALP, Runx2 and Col 1 expression.
Keywords: Lnc-RNA; BMSC Cells; Osteogenesis; Antihyperalgesia; Bone Malignant Diseases
Introduction
Bone malignant diseases always cause bone loss and ostealgia [1]. Both symptoms were highly related with osteoclasts [2]. Osteoclasts activation induces bone resorption and generates acid, which is the major reason of bone cancer pain [3]. Treatment of bone malignance contains many approaches including radiotherapy, surgery, chemotherapy, bisphosphonates etc. [4]. However, the ultimate solution of ostealgia should be to reduce bone loss and promote bone regeneration. In the last two decades, BMSCs were widely applied to acquire bone regeneration.
Bone marrow stromal cells (BMSCs) belongs to pluripotent stem cells and can differentiate into several kinds of mesenchymal cells, such as osteoblasts, chondrocytes and adipocytes [5]. Because of the multiple directional differentiating ability, BMSCs are applied for treatment of various bone diseases [6], including bone regeneration, the most popular application. Besides, BMSCs are also used in pain relief by regulating immune system [7]. In consideration of the multiple differentiation ability, BMSCs were often guided to form osteoblasts other than adipocytes, which has negative effect to bone strength [8]. Epigenetic regulation is an important effector of bone formation, moreover, BMSC gene expression controlled by non-coding RNAs was another way to affect BMSCs differentiation [9].
Non-coding RNAs include micro-RNA, long non-coding RNA, circle-RNA and so on. Amounts of micro-RNAs were proved affect BMSC differentiation by regulating relevant signaling pathways [10]. Lnc-RNAs were also proved involved in regulating several signaling pathways related to bone regeneration. One of the first series lnc-RNAs, lnc-RNA H19, was upregulated by BMP9 stimulation of BMSCs and correlated with BMP9 induced expression of osteogenic genes [11]. Others like lnc-RNA MEG3 was also interacted with SOX2 and BMP4 promoter to increase expression of osteogenic related genes [12]. H19 is related to bone cancer and served as a tumor suppressor [13]; and it is also been reported acted as a regulator of bone regeneration. H19 would function as a miRNA precursor or inhibitor of specific miRNAs [14,15]. However, in some malignant diseases, such as oral squamous cell carcinoma, hepatocellular carcinoma, H19 was upregulated and act as a biomarker [16]. Therefore, the exact role of H19 in bone metastasis is still to be explored. Furthermore, how H19 performed in the signaling pathways which related to BMSCs differentiation is unclear to date.
In the present study, we focused on the classic lnc-RNA H19, to further explore how lnc-RNA H19 induced BMSCs differentiation and increased bone regeneration. We explored several signaling pathways which were closed related to BMSC differentiaton to osteoblasts and found out which one was reacted to H19 alteration. Especially for NF-κB signal, it is reported the popular factor not only induced osteoclast releasing acid to triger cancer pain but also directly involved in BMSC antihyperalgesia. We expect H19 may act as a target to develop new drugs to reduce bone loss or increase bone volume. As bone volume increased, bone cancer pain may be alleviated.
Material and Methods
Cell Line
Bone marrow-derived mesenchymal stem cells (PCS-500-012 ™) were obtained from American Type Culture Collection. Cells were cultured in RPMI1640 medium with 10% FBS in a 37℃, 5% CO2 incubator.
Osteogenic Differentiation and Mineralization Assay
BMSCs were cultured using 6well plates (2.5 × 106 cells/well) for 7 days by applying the osteogenesis induction medium (5 mmol/L β-glycerophosphate and 50 μg/mL ascorbic acid, 100 ng/mL dexamethasone) to make osteoblastic mineralization induction. After that, 2% Alizarin Red‐S (Sigma‐Aldrich) was utilized for staining cells at pH 4.2 in order to make an evaluation of the mineralization of the cell matrix. Photos were taken under an inverted microscope with camera system.
RNA Isolation and qRT-PCR
Total RNA was extracted from cultured cells with Trizol (Invitrogen, USA) and cDNA was generated using the First Strand cDNA Synthesis kit (Thermo, USA). MRNA expression was analyzed using the SYBR Green PCR Master Mix (Toyobo, China) on Real-Time PCR System (BioRad, USA). Primers used for qRT-PCR were as follows: LncRNA-H19 forward, 5’- CCAGGCATTCATCCCGGTCA-3’ and reverse, 5’- TGCCAGCTACACCTCCGTTG-3’. GAPDH forward, 5’-AGCCACATCGCTCAGACAC-3’ and reverse, 5’-GCCCA ATACGACCAAATCC-3’. Data were calculated by a 2-ΔΔCt method by target gene compared with GAPDH firstly and control groups secondly.
Cell Transfection
The pcDNA3.1 was used as control. For overexpression of lnc-RNA H19, the full-length oligonucleotides of H19 into pcDNA3.1 vector (Introvigen, China). Small interference RNAs(siRNAs) targeting H19 and their control siRNA (si-NC) were designed and synthesized by RioBio (China). Sequence for H19 siRNA was 5’-GGACGUGACAAGCAGGACATT-3’; sequence for negative control was 5’- UUCUCCGAACGUGUCACGUTT-3’. BMSC cells were cultured and transfected with nucleotides described above using Lipofectamine 2000 reagent (Themofisher, USA). Cells were incubated for 48 hours before subsequent experiments. The efficiency of H19 overexpression or knockdown were evaluated by qRT-PCR (data not shown).
Proliferation Assay
MTT assay was used to examine the cell proliferation. Cells were seeded into 96-well plates and incubated for 0, 24, 48 hours with or without Lnc-RNA H19 overexpressed, 20μL MTT reagent (beyotime, China) was added into each well and incubated for 4 h at 37 °C in the dark. Then, the supernatant was removed, add 150 μL of DMSO into each well for another 15 min. The optical density (OD) value per well was measured at 570 nm with microplate reader (Themofisher, USA). Each experiment was repeated 3 times.
Flow Cytometry
Cells were harvested and fixed overnight with 70% ethanol at 4 °C, followed by resuspension in 500 μL of PBS with about 106 cells. After addition of 10 μL RNase (10 mg/mL), cells were left for 30 min at 37 °C and stained with 10 μL propidium iodide (1 mg/mL). Cellular DNA content was determined on a flow cytometer Beckton Dickinson (BD) FACScan (BD Biosciences, CA). Quantification of apoptotic cells was performed according to the Annexin-V-PE/7-AAD Apoptosis Detection Kit manufacturer instructions (Beyotime, China). Analyses were performed by a flow cytometer (BD FACScan). Phycoerythrin (PE) -positive and 7-amino-actinomycin D (7-AAD) -negative cells were regarded as apoptotic cells. Each experiment was repeated 3 times. Data was analyzed with flowjo 1.0 software.
Data Analysis
Data were calculated by GraphPad Prism 5.0. All experiments were repeated at least three times, and all data were presented as the mean ± SD. The differences between group were analyzed using ANOVA. Paired Student’s t-test was used for data analysis. Probability value was calculated and less than 0.05 was considered to be statistically significant.
Results
Lnc-RNA H19 was Upregulated in Condition of Osteogenic Differentiation in BMSC Cells
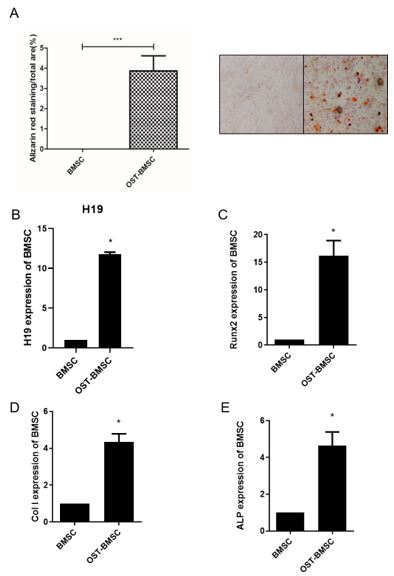
Previous experiments were performed to verified lnc-RNA H19 expression in human BMSC cells (data not show). Human BMSC cells were cultured and induced osteogenic differentiation using Osteogenic induction medium for 7 days. Alizarin Red staining showed mineralization cores formed and the volume was 4% compared almost none with the control group (figure 1A). After Osteogenic differentiation induction, expression level of H19 was elevated and accompanied with ALP, Runx2 and ColI boosted (figure 1 B-E). In detail, H19 was induced about 12 folds, Runx2 about 15 folds, collagen I about 4 folds and ALP about 4 folds, respectively. All these results had significance for P < 0.05.
Lnc-RNA H19 Induced Osteogenic Differentiation and Proliferation of BMSC Cells
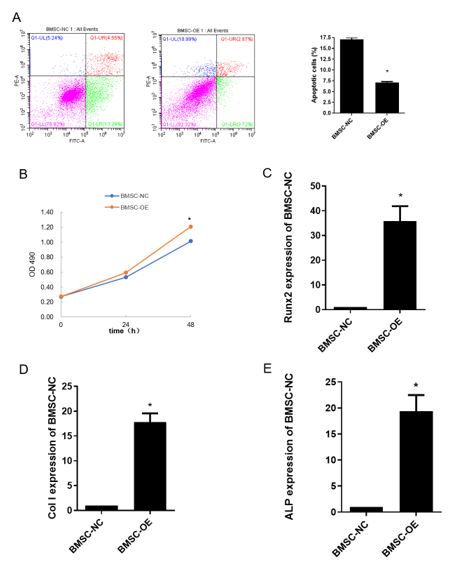
To explore whether lnc-RNA H19 was involved in BMSC cells osteogenic differentiation, we performed transient overexpression of exogenous H19. Overexpression of H19 was confirmed by real time RT-PCR (data not show), and cells were cultured after H19 overexpressed for 7 days. Alizarin Red staining showed that H19 overexpression induced Osteogenic differentiation of BMSC cells just similar as chemical induction (supplemental data). After overexpressed H19 for 48 h, flow cytometry was performed to test BMSC cell viability. The results showed that overexpression of H19 significantly inhibited cell apoptosis by 2 folds compared with control groups (figure 2A). MTT assay was also performed to study cell proliferation rate in condition of H19 overexpression. The results showed that BMSC cells with H19 overexpressed had a higher proliferation rate than control groups (figure 2B). Moreover, after H19 overexpressed, ALP, Runx2 and ColI level were also examined. The results showed that all those molecules were significantly induced (figure 2C-E), and the trend was same as osteogenic induction did with more extensive mRNA expression.
Knockdown of Lnc-RNA H19 Inhibited Activity of BMSC Cells and Osteogenic Ability
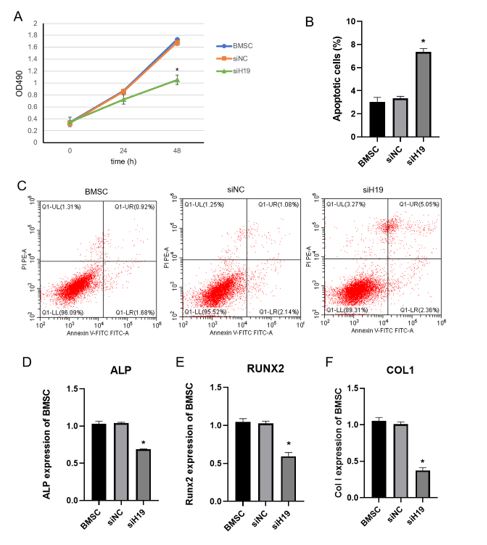
To further study H19 was critical to BMSC osteogenic potential, we knocked down H19 by 48 h in BMSC cells. MTT assay was performed to test cell proliferation rate. The results showed that cell proliferation rate was significantly slowed down by nealy 50% after knockdown of H19 (figure 3A). Flow cytometry results showed that apoptotic cells increased 2 folds by knocking down H19 compared with control groups (figure 3B-C). The osteogenic markers, ALP, Runx2 and ColI were significantly downregulated around 2 folds after knockdown of H19 (figure 3D-F).
Lnc-RNA H19 Activated BMP2, Wnt-5A and NF-κB Signals and Further Induced BMSC Cell Osteogenic Differentiation
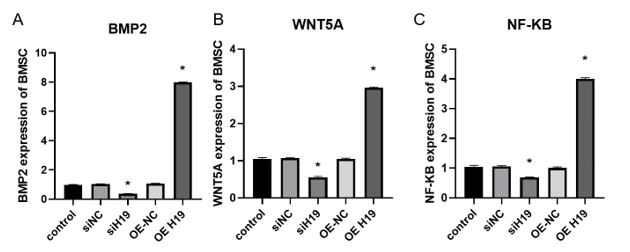
to further explore how lnc-RNA H19 induced BMSC osteogenic differentiation, several osteogenic related signaling pathway were checked after H19 was overexpressed or knockdown. The results showed that BMP2 was upregulated about 8 folds in condition of H19 overexpressed (figure 4A), meantime, Wnt-5A was upregulated about 3 folds (figure 4B) with NF-κB about 4 folds (figure 4C). Knockdown of H19 inhibit the expression of those molecules mentioned above about 70%, 50% or 60%, respectively. These results indicated that H19 could activate multiple osteogenic signals pathway to induce BMSC cells osteogenic differentiation.
Discussion
In the presents study, we observed that lnc-RNA H19 were significantly induced during osteogenic differentiation of BMSC cells in vitro. To confirm whether H19 regulating osteogenic markers or not, we subjected knockdown and overexpression of H19. The results showed H19 mediated RUNX2, ALP and Col1 expression. Furthermore, we proved H19 upregulated those bone generating markers through activating BMP2, Wnt-5A and NF-κB signaling pathways. Finally, overexpression of H19 promoted cell proliferation and inhibited apoptosis of BMSC cells. These results indicated that Lnc RNA H19 performed osteogenic function by activating and maintaining BMSC osteogenic differentiation. Because BMSC cells have multiple directional differentiation ability, to get the right lineage is key point for BMSC application in bone regeneration. Many regulators are involved in BMSC differentiation, including transcription factors, non-coding RNAs, epigenetic modifications etc. For osteogenesis, the key transcription factor is Runt-related transcription factor 2 (Runx2) [17]. Moreover, other factors could upregulate Runx2 and then cooperate with it, such as bone morphogenetic protein-2 (BMP2). BMP2 is proven a key signaling pathway by targeting Runx2 and showed dose-dependent effects on osteogenesis [18]. Furthermore, microRNAs are often played roles in inhibiting osteogenesis. For instance, miR-204 and miR-211 are activated during adipogenesis and inhibited Runx2 expression [19]. MiR-17-5p/miR-106a and miR-30c/miR-30d inhibit BMP signaling by targeting BMP2 and Smad1, which are the key components of the pathway [20]. Lnc-RNAs can also play a role in the regulation of stem cell osteogenesis. The mechanism includes mediating epigenetic changes, affecting transcriptional regulation or interacting with proteins to affect their stability and activity. Lnc-RNA MEG3 overexpression would increase the transcription and secretion of BMP4 by interrupting SOX2, a BMP4 inhibitor, binding on BMP4 promoter to promote osteogenic differentiation of MSCs [21]. Previous studies had reported that H19 upregulated Wnt/β-catenin/Runx2 through inhibiting miR-22 and miR-141 [15]. In the present study, lnc-RNA H19 induced osteogenic differentiation of BMSC cells by upregulating NF-κB, BMP2 and Wnt-5A signaling pathways. All these factors were important markers of bone formation. Despite BMP2 and Runx2, Wnt-5A is another important signaling pathway in bone formation process [22]. Since H19 could activate multiple osteogenic signaling pathways, drugs might be developed targeting to it for treatment of bone defect.
Bone regeneration is not only benefit for bone defect diseases, the ultimate solution of cancer derived bone pain also depends on it. The major factor caused cancer derived bone pain is osteoclast derived acid, inflammation factors and bone resorption. Previous studies indicated that BMSCs osteogenic differentiation could inhibit osteoclast activation and avoid bone resorption [23]. Our results also showed lnc-RNA H19 promoted BMSCs osteogenic differentiation in vitro. It is implied that targeting to lnc-RNA H19 might be benefit to both bone loss disease and cancer derived bone pain. In addition, upregulating of NF-κB was involved in BMSCs induced pain relief [7]. Especially in application of BMSC treatment to antihyperalgesia trial in animal models, p65 was significantly upregulated. Our results also showed overexpression of lnc-RNA H19 induced NF-κB expression in BMSCs. Although NF-κB dual effect of pro- or anti-inflammation, the complexity of exact mechanisms was far to reveal. The present study just showed a limited perspective. However, lnc-RNA H19 still could be a candidate to develop treatment strategy of bone loss and pain. The present study is a preliminary exploration on H19 performing in BMSCs and has a big gap from cultured cell lines to in vivo research. More work is needed to confirm whether H19 mediated osteogenic signal work or not in animal models.
In conclusion, lnc-RNA H19 induced BMSCs osteogenic differentiation in vitro through activate BMP2, Wnt-5A and NF-κB signaling pathway. H19 also induced BMSCs proliferation and avoided apoptosis of BMSCs. However, these results still need to be tested in vivo to further confirm the function of lnc-RNA H19 in BMSCs differentiation process and antihyperalgesia.
Acknowledgement
This study supported by Health Commission of Hunan Province (No: 202202054261)
Author Contribution
Qing Li and Zhengguang Wang set the conception and designed the work; Hui Long, Li Yuan and Qing Li did the experiments; Wei Gao and Qing Li made contribution to analysis and interpretation of data; Qing Li and Zhengguang Wang have drafted the manuscript and substantively revised it. All authors reviewed the manuscript.
Additional Information
The authors declared no competing interests.
Data Availability Statement
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
- Coleman RE (2006) Clinical features of metastatic bone disease and risk of skeletal morbidity. Clin Cancer Res. 12: 6243s-9.
- Mantyh PW (2006) Cancer pain and its impact on diagnosis, survival and quality of life. Nat Rev Neurosci. 7: 797-809.
- Teitelbaum SL (2007) Osteoclasts: what do they do and how do they do it? Am J Pathol. 170: 427-35.
- Mercadante S (1997) Malignant bone pain: pathophysiology and treatment. Pain, 69: 1-18.
- Gao Q, Wang L, Wang S, Huang B, Jing Y, Su J (2021) Bone Marrow Mesenchymal Stromal Cells: Identification, Classification, and Differentiation. Front Cell Dev Biol. 9: 787118.
- Arthur A, and Gronthos S (2020) Clinical Application of Bone Marrow Mesenchymal Stem/Stromal Cells to Repair Skeletal Tissue. Int J Mol Sci. 21.
- Guo W, Imai S, Yang JL, Zou S, Li H, Xu H, Moudgil KD, Dubner R, Wei F, Ren K (2018) NF-KappaB Pathway Is Involved in Bone Marrow Stromal Cell-Produced Pain Relief. Front Integr Neurosci. 12: 49.
- Ambrosi TH, Scialdone A, Graja A, Gohlke S, Jank AM, et al. (2017) Adipocyte Accumulation in the Bone Marrow during Obesity and Aging Impairs Stem Cell-Based Hematopoietic and Bone Regeneration. Cell Stem Cell, 20: 771-84 e776.
- Guo Q, Guo Q, Xiao Y, Li C, Huang Y, Luo X (2020) Regulation of bone marrow mesenchymal stem cell fate by long non-coding RNA. Bone, 141: 115617.
- Vulf M, Khlusov I, Yurova K, Todosenko N, Komar A, et al. (2022) MicroRNA Regulation of Bone Marrow Mesenchymal Stem Cells in the Development of Osteoporosis in Obesity. Front Biosci (Schol Ed). 14: 17.
- Liao J, Yu X, Hu X, Fan J, Wang J, Zhang Z, et al. (2017) lncRNA H19 mediates BMP9-induced osteogenic differentiation of mesenchymal stem cells (MSCs) through Notch signaling. Oncotarget, 8: 53581-601.
- Zhang N, Hu X, He S, Ding W, Wang F, Zhao Y, Huang Z (2019) LncRNA MSC-AS1 promotes osteogenic differentiation and alleviates osteoporosis through sponging microRNA-140-5p to upregulate BMP2. Biochem Biophys Res Commun. 519: 790-6.
- Yoshimizu T, Miroglio A, Ripoche MA, Gabory A, Vernucci M, et al. (2008) The H19 locus acts in vivo as a tumor suppressor. Proc Natl Acad Sci U S A. 105: 12417-22. 10.1073/pnas.0801540105
- Wang Y, Liu W, Liu Y, Cui J, Zhao Z, Cao H, et al. (2018) Long noncoding RNA H19 mediates LCoR to impact the osteogenic and adipogenic differentiation of mBMSCs in mice through sponging miR-188. J Cell Physiol. 233: 7435-46.
- Wu J, Zhao J, Sun L, Pan Y, Wang H, Zhang WB (2018) Long non-coding RNA H19 mediates mechanical tension-induced osteogenesis of bone marrow mesenchymal stem cells via FAK by sponging miR-138. Bone, 108: 62-70.
- Ghafouri-Fard S, Esmaeili M, Taheri M (2020) H19 lncRNA: Roles in tumorigenesis. Biomed Pharmacother. 123: 109774.
- Otto F, Thornell AP, Crompton T, Denzel A, Gilmour KC, Rosewell IR, et al. (1997) Cbfa1, a candidate gene for cleidocranial dysplasia syndrome, is essential for osteoblast differentiation and bone development. Cell, 89: 765-71.
- Javed A, Bae JS, Afzal F, Gutierrez S, Pratap J, Zaidi SK, et al. (2008) Structural coupling of Smad and Runx2 for execution of the BMP2 osteogenic signal. J Biol Chem. 283: 8412-22.
- Huang J, Zhao L, Xing L, Chen D (2010) MicroRNA-204 regulates Runx2 protein expression and mesenchymal progenitor cell differentiation. Stem Cells, 28: 357-64.
- Kang H, Hata A (2015) The role of microRNAs in cell fate determination of mesenchymal stem cells: balancing adipogenesis and osteogenesis. BMB Rep. 48: 319-23.
- Zhuang W, Ge X, Yang S, Huang M, Zhuang W, Chen P, et al. (2015) Upregulation of lncRNA MEG3 Promotes Osteogenic Differentiation of Mesenchymal Stem Cells from Multiple Myeloma Patients by Targeting BMP4 Transcription. Stem Cells, 33: 1985-97.
- Tan Z, Ding N, Lu H, Kessler JA, Kan L (2019) Wnt signaling in physiological and pathological bone formation. Histol Histopathol. 34: 303-12.
- Yang Z, Liu X, Zhao F, Yao M, Lin Z, Yang Z, et al. (2022) Bioactive glass nanoparticles inhibit osteoclast differentiation and osteoporotic bone loss by activating lncRNA NRON expression in the extracellular vesicles derived from bone marrow mesenchymal stem cells. Biomaterials, 283: 121438.
Figures at a glance