Angina with Non-Obstructive Coronary Arteries (ANOCA): Current Concepts in Pathophysiology, Diagnosis, and Management
Received Date: January 25, 2026 Accepted Date: February 09, 2026 Published Date: February 12, 2026
doi:10.17303/jhhi.2026.2.101
Citation: Abdullah SARIHAN, Abdülmelik BİRGÜN, Ömer Burak ÇELİK, Mehmet Murat ŞAHİN, Macit KALÇIK, et al. (2026) Angina with Non-Obstructive Coronary Arteries (ANOCA): Current Concepts in Pathophysiology, Diagnosis, and Management. 2: 1-15
Abstract
Angina with non-obstructive coronary arteries (ANOCA) is increasingly recognized as a prevalent and clinically relevant condition characterized by angina symptoms in the absence of obstructive epicardial coronary artery disease. ANOCA, which was once thought to be a benign finding after "normal" coronary angiography, is now recognized as a heterogeneous syndrome linked to significant symptom burden, poor quality of life, and unfavorable long-term cardiovascular outcomes. According to epidemiological data, obstructive coronary artery disease is not present in up to 40–70% of patients undergoing coronary angiography for stable angina, with a notable predominance among women. While endothelial dysfunction, autonomic dysregulation, and inflammatory processes further contribute to the complex clinical presentation of ANOCA, mounting evidence has identified coronary microvascular dysfunction and coronary vasomotor disorders as important pathophysiological mechanisms underlying the condition. These functional abnormalities cannot be detected by traditional diagnostic pathways that primarily rely on anatomical assessment. Non-invasive ischemia testing and coronary angiography alone frequently fail to explain persistent symptoms in this population. On the other hand, mechanism-based diagnosis and improved phenotypic classification are made possible by invasive coronary functional testing that includes measurements of coronary flow reserve, microvascular resistance, and vasoreactivity. The definition, epidemiology, pathophysiology, diagnostic assessment, and management approaches of ANOCA are all thoroughly covered in this review. Future research priorities, the limitations of the current body of evidence, and current guideline recommendations are also covered. To improve long-term results and advance patient-centered care, ANOCA must be acknowledged as a clinically significant syndrome rather than a benign angiographic finding.
Keywords: Angina with Non-Obstructive Coronary Arteries (ANOCA); Coronary Microvascular Dysfunction; Microvascular Spasm; Myocardial Ischemia; Coronary Angiography
Introduction
For a long time, angina pectoris was mostly seen as a clinical sign of atherosclerotic stenoses in the epicardial coronary arteries [1]. This traditional view put coronary angiography at the center of the diagnostic evaluation of stable angina. Patients who didn't have any significant stenosis were often thought to have "normal" findings or clinically insignificant disease [2]. However, over the past 20 years, more and more evidence has shown that a large number of angina symptoms can happen even when there is no obstructive coronary artery disease. This patient group is clinically diverse but still important for predicting outcomes [3].
The term "angina with non-obstructive coronary arteries" (ANOCA) currently refers to the presence of angina symptoms in patients without obstructive lesions on coronary angiography [4]. ANOCA encompasses a wide range of clinical conditions, including angiographically normal or minimally diseased coronary arteries, as well as chest pain resulting from non-ischemic mechanisms and myocardial ischemia associated with functional abnormalities [5]. In this regard, epicardial coronary anatomy alone cannot account for the pathophysiology of ANOCA, which should be viewed as a multifaceted syndrome.
Epidemiological data indicate that a substantial proportion of patients undergoing coronary angiography for stable angina estimated at nearly 40–70% are found to have no obstructive coronary artery disease [6]. This phenomenon appears to be particularly pronounced in women, pointing toward a clear female predominance of angina with non-obstructive coronary arteries (ANOCA) [7]. In everyday clinical practice, however, the diagnostic evaluation of these patients is often curtailed once obstructive disease is excluded. Symptoms may be dismissed as non-cardiac in origin, or treatment is confined to empirical antianginal therapy without further phenotyping [8]. Over time, this strategy translates into persistent symptoms, impaired quality of life, frequent emergency department presentations and outpatient visits, and a growing burden on healthcare resources.
Accumulating evidence suggests that ANOCA should no longer be regarded as a benign clinical entity. Rather, specific phenotypes particularly those related to coronary microvascular dysfunction (CMD) and coronary vasomotor abnormalities are consistently associated with a higher risk of major adverse cardiovascular events and substantial long-term morbidity [9]. Yet, most conventional diagnostic pathways remain largely centered on anatomical assessment. This emphasis makes the detection of functional coronary disturbances challenging and, in turn, constrains the adoption of targeted, mechanism-based therapeutic approaches [10].
Within this framework, a shift in perspective is needed. ANOCA should not be viewed simply as the angiographic absence of obstructive disease, but as a clinical syndrome that calls for systematic exploration of underlying pathophysiological mechanisms, incorporation of functional diagnostic tools, and implementation of individualized, mechanism-oriented therapies [4]. Accordingly, this review aims to summarize and discuss the contemporary definition of ANOCA, its biological underpinnings, available diagnostic approaches, and current concepts in mechanism-based management, drawing on the existing body of evidence.
Methods / Literature Search Strategy
The literature for this narrative review was identified through comprehensive searches of PubMed/MEDLINE, Scopus, and Web of Science databases. Articles published predominantly over the past two decades were reviewed, with particular focus on studies addressing the epidemiology, clinical characteristics, pathophysiology, diagnostic evaluation, management strategies, and prognostic implications of angina with non-obstructive coronary arteries (ANOCA). Priority was given to international clinical guidelines, expert consensus documents, randomized controlled trials, meta-analyses, and high-quality observational studies that provided mechanistic insights or clinically relevant outcomes. To ensure completeness, reference lists of selected key publications were also manually screened to identify additional relevant articles not captured in the initial database searches.
Definition, Terminology, and Clinical Spectrum
Definition of ANOCA
ANOCA describes a clinical syndrome in which patients present with symptoms compatible with angina pectoris despite the absence of obstructive epicardial coronary artery disease, typically defined as <50% luminal stenosis on invasive or non-invasive coronary imaging [1, 2]. Importantly, this concept goes beyond a simple anatomical description and reflects a broader pathophysiological construct that incorporates functional and dynamic disturbances of the coronary circulation.
ANOCA may coexist with demonstrable myocardial ischemia, but it can also manifest as angina-like chest pain in patients without objective evidence of ischemia [10]. This observation underscores that ANOCA should not be viewed purely as an ischemia-driven syndrome. Instead, it represents a clinical expression of diverse mechanisms that impair the structural and functional integrity of the coronary circulation. Current evidence most commonly implicates coronary microvascular dysfunction, epicardial or microvascular vasospasm, and endothelial dysfunction as key pathophysiological substrates of ANOCA [11].
ANOCA versus INOCA
Although ANOCA and ischemia with non-obstructive coronary arteries (INOCA) are frequently used as interchangeable terms in everyday practice, they describe related but conceptually distinct clinical entities [4]. INOCA refers specifically to patients with objectively documented myocardial ischemia in the setting of non-obstructive coronary arteries, whereas ANOCA captures a wider spectrum of individuals in whom angina symptoms predominate, regardless of whether ischemia can be objectively demonstrated [8].
From this perspective, INOCA can be considered a specific subset within the broader ANOCA spectrum. Myocardial ischemia driven by coronary microvascular dysfunction or vasospastic mechanisms aligns with the definition of INOCA, whereas angina-like chest pain related to altered pain processing, autonomic imbalance, or other non-ischemic pathways is more appropriately categorized as ANOCA [9]. In routine practice, failure to make this distinction may fuel diagnostic ambiguity and, ultimately, lead to suboptimal or misdirected therapeutic decisions. In order to facilitate a clear conceptual distinction between ANOCA and INOCA, the key differences between the two entities are summarized in Table 1.
Overlapping Phenotypes and Diagnostic Challenges
In patients with ANOCA, it is uncommon to identify a single dominant pathophysiological mechanism. More often, overlapping phenotypes reflecting the coexistence of several concurrent processes are encountered [12]. The combination of coronary microvascular dysfunction with epicardial or microvascular vasospasm adds to the heterogeneity of clinical presentations and helps explain the variable and sometimes unpredictable nature of patient-reported symptoms.
Such heterogeneity inherently limits the diagnostic value of strategies that rely exclusively on symptom profiles or conventional non-invasive testing. Although standard coronary angiography yields detailed information about epicardial coronary anatomy, it provides little insight into microvascular function or coronary vasomotor behavior [13]. Accordingly, meaningful phenotyping across the ANOCA spectrum depends on the incorporation of functional diagnostic modalities into modern diagnostic pathways.
Before a diagnosis of ANOCA is established, a range of alternative clinical conditions should be thoughtfully considered and excluded. Non-cardiac causes of chest pain such as musculoskeletal, gastrointestinal, and psychogenic etiologies warrant systematic assessment as an initial step [14]. Moreover, acute coronary syndromes, and in particular myocardial infarction with non-obstructive coronary arteries (MINOCA), constitute a separate clinical entity that requires distinction from ANOCA on the basis of both underlying mechanisms and clinical presentation [15].
In a similar vein, structural heart diseases such as significant valvular pathology, hypertrophic cardiomyopathy, or advanced left ventricular dysfunction should also fall outside the definition of ANOCA. Making this distinction is crucial, not only to ensure consistency across clinical research, but also to guide appropriate therapeutic decision-making in everyday practice [4]. Within this framework, ANOCA encompasses multiple mechanism-based phenotypes, including functional and structural forms of coronary microvascular dysfunction, vasospastic angina, and mixed or overlapping endotypes (Table 2).
Epidemiology and Clinical Presentation
ANOCA is frequently encountered among patients evaluated for stable angina. Indeed, nearly 40–70% of individuals undergoing elective coronary angiography are found to have no obstructive coronary artery disease, underscoring that a large share of angina symptoms arise from mechanisms beyond epicardial coronary stenosis [2].
Sex-specific analyses consistently demonstrate a substantially higher prevalence of ANOCA in women. Even in the presence of stable angina symptoms and, in some cases, objective evidence of ischemia, female patients are more likely to exhibit non-obstructive coronary arteries [16]. This sex-related difference is thought to reflect a combination of biological and pathophysiological factors, including hormonal influences, variations in microvascular architecture, and altered coronary vasomotor responsiveness.
Traditional cardiovascular risk factors are commonly encountered in patients with ANOCA, yet their overall distribution appears less uniform than in individuals with obstructive coronary artery disease. Hypertension, dyslipidemia, and diabetes mellitus are frequently present; however, the broader risk profile of ANOCA remains notably heterogeneous [3]. This pattern implies that conventional risk-based assessment models may not perform optimally in this patient population.
The clinical presentation of ANOCA is highly variable and often diverges from the classical description of stable angina. Many patients report exertional chest pain, chest tightness, dyspnea, or reduced exercise tolerance; yet symptoms may also arise at rest or be triggered by emotional stress [5].
In ANOCA associated with coronary microvascular dysfunction, chest pain frequently displays atypical characteristics and may not satisfy traditional angina criteria. Such presentations increase the likelihood that symptoms are attributed to non-cardiac causes and may contribute to delays in establishing the correct diagnosis [9]. Moreover, symptoms in this subgroup are often prolonged, recurrent, and show variable or incomplete responses to conventional antianginal therapy.
By contrast, ANOCA driven by vasospastic mechanisms most commonly presents with chest pain at rest, often occurring during the night or in the early morning hours and following an episodic pattern. This presentation mirrors the dynamic behavior of epicardial or microvascular coronary vasospasm [12]. Beyond symptom characteristics, patients with ANOCA typically carry a considerable symptom burden that translates into a meaningful reduction in quality of life. Prior studies have reported frequent healthcare utilization, restrictions in everyday physical activity, and higher levels of psychological distress within this population [17]. Collectively, these observations highlight that ANOCA is not only a diagnostic challenge, but also a clinically important and distinctly patient-centered condition.
ANOCA was once regarded as a relatively benign condition; however, this perception has been challenged by accumulating evidence. Specifically, ANOCA phenotypes related to coronary microvascular dysfunction or coronary vasomotor abnormalities have been associated with a higher risk of major adverse cardiovascular events [13]. Therefore, ANOCA should not be viewed solely as a source of symptoms, but rather as a clinical syndrome with important long-term prognostic implications.
Pathophysiology of ANOCA
Coronary Microvascular Dysfunction (CMD)
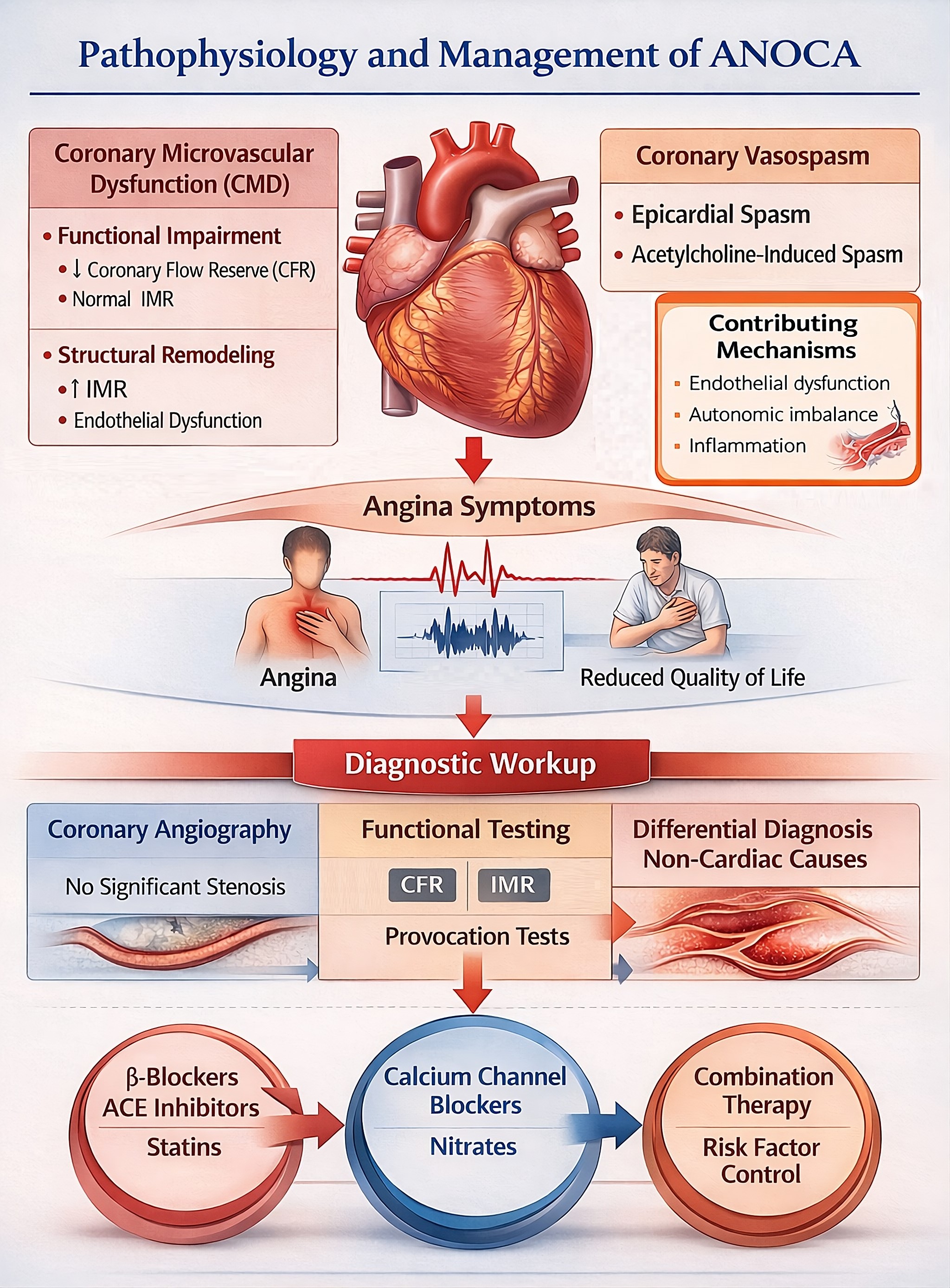
Coronary microvascular dysfunction (CMD) encompasses a constellation of structural and functional abnormalities within the coronary microvascular bed and constitutes a central pathophysiological substrate of ANOCA. Hallmark features include increased microvascular resistance, impaired endothelium-dependent and endothelium-independent vasodilation, and a consequent reduction in coronary flow reserve [9]. Collectively, these disturbances can promote myocardial ischemia, particularly during periods of heightened myocardial oxygen demand. The mechanisms underlying CMD are multifaceted and involve microvascular luminal narrowing, microvascular rarefaction, endothelial dysfunction, and activation of inflammatory pathways. Across a range of clinical contexts, including metabolic syndrome and heart failure, CMD has been linked to heightened myocardial stress and an increased risk of major adverse cardiovascular events (MACE) [9].
Coronary Vasomotor Disorders (Microvascular Spasm)
Coronary vasomotor disorders represent a major pathophysiological component of ANOCA and may involve either epicardial or microvascular spasm. These conditions are defined by an abnormal contractile response of the coronary vasculature, capable of producing transient reductions in coronary blood flow and episodic myocardial ischemia.
In clinical practice, the diagnosis of coronary spasm is most often established through pharmacological provocation testing with agents such as acetylcholine or ergonovine. Reproduction of typical symptoms accompanied by ischemic electrocardiographic or angiographic changes during testing is considered diagnostic of a vasomotor disorder [18].
Pooled data from meta-analyses suggest that epicardial coronary spasm is detected in roughly 40% of patients with ANOCA (range 16–73%), whereas microvascular spasm is identified in approximately one-quarter of cases. Together, these observations further emphasize the marked heterogeneity of ANOCA [19]. Key clinical, ischemic, and diagnostic distinctions between CMD and vasospastic angina are summarized in Table 3.
Endothelial Dysfunction
Endothelial dysfunction serves as a shared pathophysiological substrate across several mechanisms implicated in ANOCA, most notably CMD and coronary vasospasm. Under physiological conditions, endothelial cells regulate vascular tone largely through the production of nitric oxide (NO). When endothelial function is impaired, NO bioavailability is reduced and vasoconstrictor pathways become relatively dominant, leading to abnormal vasomotor responses at both the microvascular and epicardial levels [9]. Importantly, the relevance of endothelial dysfunction extends beyond symptom generation, as it has also been linked to an increased risk of adverse cardiovascular outcomes over the long term.
Autonomic and Neurohumoral Regulation
Alterations in autonomic nervous system balance and neurohumoral regulation represent additional contributors to the pathophysiology of ANOCA. Disturbances in sympathetic and parasympathetic activity can promote fluctuations in vascular tone, thereby predisposing to vasomotor abnormalities such as microvascular or epicardial spasm [20]. These processes may help explain the occurrence of angina symptoms at rest or in association with emotional stress.
Inflammation and Microvascular Structural Changes
Low-grade chronic inflammation may play a contributory role in promoting structural alterations within the coronary microvasculature and in driving endothelial dysfunction, thereby increasing susceptibility to CMD and vasomotor disorders. Chronic inflammatory activity has also been linked to microvascular rarefaction and heightened intramural resistance, leading to further impairment of coronary microvascular function [9].
Diagnostic Evaluation of ANOCA
The diagnostic evaluation of suspected ANOCA begins with a thorough clinical assessment. Nevertheless, approaches that rely predominantly on symptom characterization have important limitations in this population. Patients may exhibit features typical of stable angina, yet they frequently report non-specific complaints such as atypical chest pain, dyspnea, or reduced exercise tolerance. Importantly, symptom patterns do not reliably indicate the underlying pathophysiological mechanism [5]. As a result, dependence on clinical presentation alone may foster diagnostic uncertainty and lead to suboptimal or misdirected therapeutic decisions. In this context, growing evidence supports the adoption of mechanism-based diagnostic strategies for patients with ANOCA [4].
Non-Invasive Testing
Non-invasive ischemia testing is commonly incorporated into the evaluation of patients with suspected ANOCA, yet its overall diagnostic performance remains modest. Modalities such as exercise electrocardiography, stress echocardiography, and nuclear perfusion imaging are well suited for the detection of epicardial coronary stenosis; however, they often lack sensitivity for uncovering microvascular dysfunction or vasomotor abnormalities [1]. Cardiac magnetic resonance (CMR) perfusion imaging has emerged as a promising tool for the assessment of myocardial perfusion heterogeneity and microvascular involvement. Still, no single non-invasive technique can reliably delineate ANOCA subtypes, and normal test results do not preclude the presence of clinically relevant functional coronary abnormalities [9].
Role and Limitations of Coronary Angiography
Coronary angiography remains the reference standard for defining epicardial coronary anatomy. In patients with ANOCA, angiographic findings are often normal or reveal only minimal atherosclerotic change. This scenario may inadvertently prompt early termination of the diagnostic workup and the mislabeling of patients as having “normal” coronary arteries [2]. A key limitation of coronary angiography in the context of ANOCA is its inability to directly interrogate microvascular function or coronary vasomotor behavior. Accordingly, once obstructive coronary artery disease has been excluded, additional functional testing should be pursued [4].
Invasive Coronary Functional Testing
Invasive coronary functional testing occupies a pivotal position in the diagnostic evaluation of ANOCA, as it permits direct interrogation of the underlying pathophysiological mechanisms. Commonly applied assessments include measurements of coronary flow reserve (CFR), the index of microvascular resistance (IMR), and coronary vasoreactivity testing [21]. CFR and IMR offer quantitative insights into microvascular function and are fundamental for establishing the diagnosis of coronary microvascular dysfunction. In addition, provocation testing with acetylcholine enables the detection of both epicardial and microvascular coronary spasm [22]. Prospective investigations have shown that these procedures are safe, feasible, and clinically informative. Importantly, a mechanism-based diagnosis derived from invasive functional assessment supports the implementation of targeted therapies that translate into improved symptom burden and better quality of life [13].
Diagnostic Algorithms and Mechanism-Based Approach
Current concepts emphasize that ANOCA should not be approached as a diagnosis of exclusion based solely on anatomical findings, but rather as a condition that warrants a positive, mechanism-based diagnostic evaluation. After obstructive coronary artery disease has been ruled out, incorporation of invasive functional testing into diagnostic pathways is therefore recommended [4]. This approach allows precise phenotyping of ANOCA, including microvascular dysfunction, vasospastic angina, and mixed or overlapping patterns, and provides the basis for truly individualized management strategies. The respective strengths and limitations of non-invasive and invasive diagnostic modalities in ANOCA are summarized in Table 4.
Management Strategies in ANOCA
The central principle of ANOCA management is not simply to label the condition as “non-obstructive,” but to actively identify the dominant underlying pathophysiological mechanism and institute targeted therapy accordingly. Empirical, purely symptom-driven treatment approaches are frequently inadequate in this population and may leave patients with ongoing symptoms [5]. In this context, contemporary consensus statements advocate differentiating between major ANOCA phenotypes, such as coronary microvascular dysfunction (CMD) and vasospastic angina (VSA), and applying mechanism-oriented treatment strategies aligned with the identified substrate [4].
Treatment of Coronary Microvascular Dysfunction
In patients with coronary microvascular dysfunction, the overarching therapeutic aim is to improve microvascular tone and restore a more favorable balance between myocardial oxygen supply and demand. Beta-blockers are commonly employed as first-line agents, given their ability to lower heart rate and myocardial oxygen consumption, which in turn facilitates symptom relief [23]. Angiotensin-converting enzyme inhibitors and angiotensin receptor blockers are also recommended, largely because of their favorable effects on endothelial function. Statins may further contribute to symptomatic improvement through anti-inflammatory actions and stabilization of endothelial biology [9]. By contrast, nitrates tend to have limited efficacy in CMD and, in some individuals, fail to achieve meaningful symptom control. Accordingly, therapeutic decisions should be individualized and anchored in a mechanism-based treatment strategy [4].
Treatment of Vasospastic Angina
Management of vasospastic angina centers on therapies that enhance coronary vasodilation. Calcium channel blockers are considered first-line agents for preventing both epicardial and microvascular coronary spasm and are highly effective in controlling symptoms [24]. Long-acting nitrates can be added as adjunctive therapy when used in combination with calcium channel blockers. By contrast, beta-blockers particularly non-selective agents should generally be avoided in vasospastic angina, as they may aggravate coronary spasm [18]. Beyond pharmacological treatment, identification and modification of precipitating factors, including smoking, cold exposure, and heightened sympathetic activity, represent an essential component of comprehensive management.
Stratified Therapy Based on Invasive Functional Testing
Stratified medical therapy guided by invasive coronary functional testing has emerged as a major shift in the contemporary management of ANOCA. The clinical value of this approach was established in the CorMicA trial [13]. In this randomized controlled study, treatment strategies tailored according to invasive functional test results were associated with significant reductions in angina burden and substantial improvements in quality of life compared with standard care. Together, these data underscore that mechanism-based therapy is superior not only in addressing the underlying pathophysiology, but also in achieving outcomes that are meaningful to patients. Mechanism-oriented treatment strategies according to the underlying ANOCA endotype are summarized in Table 5.
Long-Term Management and Follow-Up
ANOCA should not be viewed as a benign clinical entity. Long-term observational data indicate that, in particular, the presence of CMD is associated with a higher risk of major adverse cardiovascular events [17]. Consequently, patients with ANOCA warrant regular clinical follow-up, comprehensive management of cardiovascular risk factors, and ongoing assessment of treatment adherence. Evaluation of therapeutic success should extend beyond ischemia testing alone and also incorporate patient-reported symptom burden and measures of health-related quality of life.
Clinical Implications
ANOCA should no longer be regarded as a benign angiographic finding but rather as a clinically significant syndrome associated with substantial symptom burden and adverse long-term cardiovascular outcomes. Multiple observational studies have demonstrated that patients with angina in the absence of obstructive coronary artery disease experience higher rates of major adverse cardiovascular events compared with asymptomatic populations, particularly when coronary microvascular dysfunction is present. These findings underscore the importance of systematic evaluation and long-term follow-up in this patient group rather than premature diagnostic closure after “normal” coronary angiography [3, 9].
From a diagnostic standpoint, reliance on anatomical imaging alone is insufficient in patients with persistent angina symptoms. Growing evidence supports the incorporation of functional coronary assessment to identify underlying mechanisms such as coronary microvascular dysfunction and vasospastic disorders. Invasive coronary functional testing, including measurement of coronary flow reserve, microvascular resistance, and vasoreactivity, enables precise phenotyping of ANOCA and facilitates mechanism-based clinical decision-making. This approach aligns with contemporary expert consensus recommendations and represents a shift toward positive diagnosis rather than exclusion-based labeling [4, 20].
Therapeutically, stratified management tailored to the dominant pathophysiological endotype has demonstrated clear clinical benefit. The CorMicA trial provided robust evidence that treatment guided by invasive functional testing significantly improves angina burden and health-related quality of life compared with standard empirical care. These data highlight that targeted therapy—such as beta-blockers and renin–angiotensin system inhibitors for microvascular dysfunction, or calcium channel blockers and nitrates for vasospastic angina—is superior to uniform antianginal treatment strategies. Accordingly, clinicians should prioritize individualized, mechanism-oriented therapy to optimize patient outcomes [13].
Beyond symptom control, aggressive management of cardiovascular risk factors remains a cornerstone of long-term care in ANOCA. Endothelial dysfunction and microvascular impairment are closely linked to traditional risk factors including hypertension, dyslipidemia, diabetes mellitus, and systemic inflammation. Comprehensive risk modification strategies therefore play a critical role in both symptom improvement and potential prognostic benefit. Taken together, these clinical considerations emphasize that ANOCA requires proactive diagnosis, targeted therapy, and structured follow-up rather than reassurance alone.
Prognosis of ANOCA
For many years, ANOCA was viewed as a benign clinical entity. Over the past two decades, however, accumulating observational and prospective data have challenged this perception. Patients who present with angina in the absence of obstructive coronary artery disease have been shown to carry a significantly higher long-term risk of major adverse cardiovascular events (MACE) [3]. This excess risk appears to be driven, to a large extent, by the presence of underlying coronary microvascular dysfunction. Consequently, ANOCA should not be considered merely a symptomatic condition, but rather a distinct clinical phenotype with important prognostic implications.
Among individuals with stable angina symptoms and no evidence of obstructive coronary artery disease, the risks of myocardial infarction, heart failure, and cardiovascular death are higher than those observed in asymptomatic populations [3]. Long-term event rates appear to be particularly increased in patients with objectively documented microvascular dysfunction. In this context, a reduced coronary flow reserve has emerged as an independent marker of adverse prognosis [25].
ANOCA is encountered more frequently in women. In female patients, myocardial ischemia occurring in the absence of epicardial coronary obstruction is associated with an elevated long-term cardiovascular risk. Data from the Women’s Ischemia Syndrome Evaluation (WISE) studies indicate that the risk of major adverse cardiovascular events in this population is substantially higher than previously appreciated [26].
Together, these observations reinforce that ANOCA represents a clinically meaningful condition in women and should neither be underestimated nor dismissed. Although mechanism-based diagnosis guided by invasive coronary functional testing has not yet been shown to directly reduce hard clinical endpoints, it has been consistently associated with substantial improvements in symptom control and health-related quality of life [13]. Moreover, reductions in symptom burden, enhanced physical activity, and fewer recurrent healthcare encounters may, over time, indirectly contribute to more favorable long-term clinical outcomes.
On the basis of current evidence, patients with ANOCA should not be labeled as having “normal coronary arteries” and discharged without further evaluation. Rather, this population warrants regular clinical follow-up, intensive management of cardiovascular risk factors, and implementation of mechanism-based treatment strategies aligned with the underlying pathophysiology.
Given the heterogeneous nature of ANOCA, prognostic assessment should be individualized and should extend beyond anatomical findings to integrate clinical presentation, functional abnormalities, and disease mechanisms. The principal pathophysiological substrates of ANOCA including functional and structural CMD, epicardial or microvascular vasospasm, and contributory processes such as endothelial dysfunction and autonomic imbalance are schematically illustrated in Figure 1.
Special Populations and Clinical Scenarios
ANOCA is encountered significantly more often in women than in men. In female patients, myocardial ischemia in the absence of obstructive epicardial coronary artery disease is most commonly attributable to coronary microvascular dysfunction and vasomotor abnormalities. Data from the Women’s Ischemia Syndrome Evaluation (WISE) studies indicate that women with ANOCA carry a substantial symptom burden that is accompanied by an increased risk of adverse long-term cardiovascular outcomes [26]. Collectively, these observations challenge the historical perception of ANOCA as a benign condition in women and underscore the importance of systematic diagnostic evaluation and targeted, mechanism-based management in this population.
In younger individuals, the diagnosis of ANOCA is frequently delayed or missed. Because traditional cardiovascular risk factors are often absent or less prevalent, symptoms may be attributed to non-cardiac causes, resulting in under-recognition and limited diagnostic evaluation. Nevertheless, vasospastic angina and coronary microvascular dysfunction can occur in younger patients and may lead to clinically meaningful angina, exercise intolerance, and impaired quality of life [12]. Raising awareness of ANOCA in this age group is therefore critical to prevent diagnostic dismissal and to facilitate timely and appropriate management.
Diabetes mellitus and metabolic syndrome have well-established detrimental effects on coronary microvascular function. In patients with diabetes, endothelial dysfunction, microvascular remodeling, and impaired vasodilatory capacity contribute to the development of ANOCA. Several studies indicate that coronary flow reserve is reduced in this population and that such impairment is linked to a less favorable cardiovascular prognosis [25].
Systemic autoimmune and inflammatory disorders, including systemic lupus erythematosus and rheumatoid arthritis, may contribute to the development of ANOCA through chronic inflammation, endothelial dysfunction, and microvascular injury. In such patients, ischemic symptoms occurring in the absence of obstructive coronary artery disease often point toward underlying inflammatory processes and warrant heightened clinical vigilance [27].
Current Guidelines and Ongoing Research
Contemporary clinical guidelines increasingly recognize the importance of ANOCA in the evaluation and management of patients with chronic coronary syndromes. Both the European Society of Cardiology guidelines on Chronic Coronary Syndromes and the European Association of Percutaneous Cardiovascular Interventions expert consensus document emphasize that, after obstructive coronary artery disease has been excluded, further functional assessment should be considered to identify underlying coronary microvascular dysfunction or vasomotor disorders [1, 4]. Despite this growing recognition, many current recommendations are derived from low- to moderate-quality evidence, reflecting the limited availability of large-scale prospective data in this field.
A major challenge in ANOCA research remains the scarcity of randomized controlled trials (RCTs) assessing hard clinical endpoints, including myocardial infarction, heart failure, and cardiovascular mortality. At present, most therapeutic strategies are informed by observational data, pathophysiological rationale, or small randomized studies that primarily target symptom relief rather than long-term outcomes. This limited body of high-quality RCT evidence constrains the strength and generalizability of current guideline recommendations and highlights the need for adequately powered, outcome-focused clinical trials [4].
In recent years, growing attention has been directed toward mechanistic studies aimed at deepening our understanding of ANOCA pathophysiology. Phenotyping based on invasive coronary functional testing has emerged as a promising strategy to inform targeted therapeutic decision-making. The CorMicA trial represents a landmark randomized study showing that stratified, mechanism-based treatment leads to meaningful improvements in angina burden and quality of life compared with standard care [13].
Nonetheless, larger multicenter trials with extended follow-up and more diverse patient populations are still needed to confirm these findings and to determine their impact on long-term clinical prognosis. Despite the expanding evidence base, several barriers continue to limit the widespread implementation of invasive coronary functional testing in routine clinical practice. These challenges include the requirement for specialized expertise, longer procedural times, additional costs, and variability in operator experience among centers. Addressing these obstacles is critical to enable standardized diagnostic pathways and to ensure broader and more equitable access to mechanism-based diagnosis and treatment for patients with ANOCA.
Future Directions and Knowledge Gaps in ANOCA
One of the most important unmet needs in ANOCA management is the absence of standardized diagnostic pathways. Once obstructive coronary artery disease has been excluded, diagnostic strategies often vary substantially between centers, resulting in delayed diagnosis or under-recognition of ANOCA in a considerable proportion of patients [4]. The lack of a uniform diagnostic algorithm fuels clinical uncertainty and contributes to inconsistent management. Future initiatives should therefore prioritize the development and implementation of standardized diagnostic pathways that incorporate both invasive and non-invasive functional testing into routine clinical care.
The capacity of non-invasive imaging modalities to reliably distinguish ANOCA endotypes remains limited. Cardiac magnetic resonance (CMR) and positron emission tomography (PET) perfusion imaging represent promising approaches for the assessment of coronary microvascular function; however, large prospective validation studies are still lacking [9]. Additional research is therefore required to define the diagnostic performance of these techniques in comparison with invasive coronary functional testing and to clarify their role within clinical decision-making algorithms.
Most pharmacological therapies currently used in ANOCA are extrapolated from evidence generated in populations with obstructive coronary artery disease. While mechanism-based treatment strategies have demonstrated consistent benefits with respect to symptom relief and quality of life, randomized controlled trials assessing their effects on hard clinical endpoints such as mortality and myocardial infarction remain scarce [5]. This gap in evidence underscores the need for dedicated clinical trials targeting specific ANOCA endotypes, particularly coronary microvascular dysfunction and vasospastic angina.
The higher prevalence of ANOCA in women further emphasizes the importance of sex-specific research. Available data suggest that coronary microvascular dysfunction may have a more prominent role in women; however, the clinical significance of these differences remains incompletely defined [26]. Future investigations should therefore prioritize the establishment of sex-specific diagnostic thresholds, deeper characterization of underlying pathophysiological mechanisms, and the development of tailored therapeutic strategies.
Patient-centered outcomes such as symptom burden, functional capacity, and quality of life remain underrepresented in ANOCA research. Given the association between ANOCA, recurrent healthcare utilization, and impaired quality of life, long-term follow-up studies are essential to determine the sustained effects of mechanism-based diagnostic and therapeutic strategies on both patient-reported and clinical outcomes [3].
Conclusion
Although ANOCA was long regarded as a benign clinical entity, accumulating evidence indicates that this perception is no longer justified. Among patients presenting with angina in the absence of obstructive coronary artery disease, the presence of coronary microvascular dysfunction and vasomotor disorders is associated with a substantial symptom burden and adverse long-term clinical outcomes. The heterogeneous pathophysiology of ANOCA therefore necessitates a mechanism-based approach to both diagnosis and management. Diagnostic strategies that rely exclusively on anatomical assessment are inadequate in this population, whereas the integration of invasive and non-invasive functional evaluations allows more accurate phenotyping and more informed therapeutic decision-making.
Treatment strategies guided by invasive coronary functional testing have demonstrated meaningful improvements in symptom control and quality of life. Nevertheless, large-scale randomized controlled trials evaluating the effects of mechanism-based therapies on hard clinical endpoints remain scarce, leaving important gaps in the current evidence base. Future research should prioritize the development of standardized diagnostic algorithms, deeper insight into sex-specific pathophysiological differences, and long-term studies focused on patient-reported outcomes. Collectively, such efforts will help shift ANOCA from a condition dismissed as “normal coronary arteries” to a clinically meaningful and treatable spectrum of disease.
- Knuuti J, Wijns W, Saraste A (2020) ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur Heart J. 41: 407-77.
- Patel MR, Peterson ED, Dai D (2010) Low diagnostic yield of elective coronary angiography. N Engl J Med. 362: 886-95.
- Jespersen L, Hvelplund A, Abildstrøm SZ (2012) Stable angina pectoris with no obstructive coronary artery disease is associated with increased risks of major adverse cardiovascular events. Eur Heart J. 33: 734-44.
- Kunadian V, Chieffo A, Camici PG (2020) An EAPCI Expert Consensus Document on Ischaemia with Non-Obstructive Coronary Arteries. Eur Heart J. 41: 3504-20.
- Ford TJ, Berry C (2019) How to diagnose and manage angina without obstructive coronary artery disease: Lessons from the British Heart Foundation CorMicA Trial. Interv Cardiol. 14: 76-82.
- Sedlak TL, Lee M, Izadnegahdar M, Merz CN, Gao M, et al. (2013) Sex differences in clinical outcomes in patients with stable angina and no obstructive coronary artery disease. Am Heart J. 166: 38-44.
- Shaw LJ, Bugiardini R, Merz CN (2009) Women and ischemic heart disease: evolving knowledge. J Am Coll Cardiol. 54: 1561-75.
- Bairey Merz CN, Pepine CJ, Walsh MN, Fleg JL (2017) Ischemia and no obstructive coronary artery disease (INOCA): Developing evidence-based therapies and research agenda for the next decade. Circulation. 135: 1075-92.
- Taqueti VR, Di Carli MF (2018) Coronary microvascular disease pathogenic mechanisms and therapeutic options. J Am Coll Cardiol. 72: 2625-41.
- Berry C, Camici PG, Crea F (2025) Clinical standards in angina and non-obstructive coronary arteries: A clinician and patient consensus statement. Int J Cardiol. 429: 133162.
- Camici PG, Crea F (2007) Coronary microvascular dysfunction. N Engl J Med. 356: 830-40.
- Ong P, Aziz A, Hansen HS, Prescott E, Athanasiadis A, et al. (2015) Structural and functional coronary artery abnormalities in patients with vasospastic angina pectoris. Circ J. 79: 1431-8.
- Ford TJ, Stanley B, Good R, (2018) Stratified medical therapy using invasive coronary function testing in angina: The CorMicA Trial. J Am Coll Cardiol. 72: 2841-55.
- Eslick GD, Jones MP, Talley NJ (2003) Non-cardiac chest pain: prevalence, risk factors, impact and consulting. Aliment Pharmacol Ther. 17: 115-24.
- Tamis-Holland JE, Jneid H, Reynolds HR (2019) Contemporary diagnosis and management of patients with myocardial infarction in the absence of obstructive coronary artery disease. Circulation. 139: e891- 908.
- Waheed N, Elias-Smale S, Malas W (2020) Sex differences in non-obstructive coronary artery disease. Cardiovasc Res. 116: 829- 40.
- Jespersen L, Abildstrøm SZ, Hvelplund A, Prescott E (2013) Persistent angina and its association with long-term anxiety, depression, and quality of life. Clin Res Cardiol. 102: 57-81.
- Beltrame JF, Crea F, Kaski JC (2017) International standardization of diagnostic criteria for vasospastic angina. Eur Heart J. 38: 2565-8.
- Woudstra J, Vink CEM, Schipaanboord DJM (2023) Coronary vasospasm in ANOCA patients: A systematic review and meta-analysis. Front Cardiovasc Med. 10: 1129159.
- Ford TJ, Ong P, Sechtem U (2020) Assessment of vascular dysfunction in patients without obstructive coronary artery disease. JACC Cardiovasc Interv. 13: 1847-64.
- Fearon WF, Balsam LB, Farouque HM (2003) Novel index for invasively assessing the coronary microcirculation. Circulation. 107: 3129-32.
- Crooijmans C, Jansen TPJ, Meeder JG (2025) Safety, feasibility, and diagnostic yield of invasive coronary function testing. JAMA Cardiol. 10: 384-90.
- Lanza GA, Crea F (2010) Primary coronary microvascular dysfunction: clinical presentation, pathophysiology, and management. Circulation. 121: 2317-25.
- JCS Joint Working Group (2014) Guidelines for diagnosis and treatment of patients with vasospastic angina. Circ J. 78: 2779-01.
- Murthy VL, Naya M, Taqueti VR (2014) Effects of sex on coronary microvascular dysfunction and cardiac outcomes. Circulation. 129: 251-27.
- Bairey Merz CN, Shaw LJ, Reis SE, (2006) Insights from the WISE Study: Gender differences in ischemic heart disease. J Am Coll Cardiol. 47: S21-9.
- Zanatta E, Colombo C, D'Amico G, d'Humières T, Dal Lin C, Tona F (2019) Inflammation and coronary microvascular dysfunction in autoimmune rheumatic diseases. Int J Mol Sci. 20:5563.

Tables at a glance
Figures at a glance