Green Synthesis and Characterization of Cadmium Sulfide (CdS) Nanoparticles Using Gracilaria edulis Seaweed Extract
Received Date: March 11, 2026 Accepted Date: March 31, 2026 Published Date: April 06, 2026
doi:10.17303/jmsa.2026.10.101
Citation: Gomathi Ramadoss, Gayathri Nagarajan, S. Sri Buvaneswari, Aravindh Muruganantham (2026) Green Synthesis and Characterization of Cadmium Sulfide (CdS) Nanoparticles Using Gracilaria edulis Seaweed Extract. J Mater sci Appl 10: 1-10
Abstract
The present study reports an eco-friendly approach for the green synthesis of cadmium sulfide (CdS) nanoparticles using marine red algae Gracilaria edulis extract as a natural reducing and stabilizing agent. The biosynthesis method provides a simple and sustainable alternative to conventional chemical methods for nanoparticle production. The formation and physicochemical properties of the synthesized CdS nanoparticles were investigated using various characterization techniques.UV–Visible spectroscopy revealed a characteristic absorption peak at 264 nm, confirming the formation of CdS nanoparticles and indicating a blue shift attributed to the quantum confinement effect. Fourier Transform Infrared (FTIR) analysis demonstrated the presence of bioactive functional groups in the seaweed extract that play a significant role in the reduction and stabilization of nanoparticles. X-ray diffraction (XRD) analysis confirmed the crystalline nature of the synthesized CdS nanoparticles, and the average crystallite size was estimated to be approximately 11.3 nm using the Debye–Scherrer equation. Scanning Electron Microscopy (SEM) images showed that the nanoparticles possess uniform morphology with predominantly spherical shapes, with particle sizes ranging from 20–50 nm.
The findings indicate that Gracilaria edulis seaweed extract is an effective biological resource for the green synthesis of CdS nanoparticles, offering a simple, environmentally friendly and cost-effective strategy for nanoparticle fabrication with potential applications in nanotechnology and environmental sciences.
Keywords: Green synthesis; Gracilaria edulis; Cadmium sulfide nanoparticles; Seaweed extract; XRD; SEM.
1. Introduction
Nanotechnology has emerged as an important interdisciplinary field involving the design, synthesis and application of materials at the nanoscale level, typically ranging from 1 to 100 nm. At this scale, materials exhibit unique physical and chemical properties that differ significantly from those of their bulk counterparts. These distinctive characteristics arise mainly from the large surface-to-volume ratio and quantum confinement effects associated with nanoscale materials. Because of these properties, nanomaterials have attracted considerable attention in various fields including medicine, electronics, catalysis and environmental remediation [1,2].
Nanomaterials can be broadly classified into carbon-based nanomaterials, metal nanoparticles, metal oxide nanoparticles and quantum dots. Carbon nanomaterials such as graphene and carbon nanotubes possess exceptional electrical, mechanical and thermal properties and are widely used in nanoelectronics and biomedical applications [3]. Similarly, metal oxide nanoparticles including titanium dioxide (TiO₂), zinc oxide (ZnO) and iron oxide (Fe₃O₄) have gained significant importance due to their applications in photocatalysis, sensors, antimicrobial coatings and environmental purification technologies [4].
Among different nanomaterials, metal nanoparticles have received considerable attention because of their remarkable optical, catalytic and antimicrobial properties. In particular, silver nanoparticles (AgNPs) are one of the most extensively studied nanomaterials due to their strong antibacterial, antifungal and antiviral activities [5]. Silver nanoparticles also exhibit unique physicochemical properties such as surface plasmon resonance, high electrical conductivity and excellent catalytic efficiency. These properties make them suitable for various biomedical and environmental applications including antimicrobial coatings, wound dressings, drug delivery systems and water purification technologies [6]..
Various physical, chemical and biological methods have been developed for the synthesis of nanoparticles. Conventional chemical synthesis methods often involve toxic reducing agents and may generate hazardous by-products. Therefore, environmentally friendly synthesis approaches known as green synthesis have gained increasing interest in recent years [7]. Green synthesis involves the use of plant extracts, microorganisms or natural biomolecules as reducing and stabilizing agents, offering advantages such as simplicity, cost-effectiveness and environmental sustainability [8].
Nanomaterials possess several unique properties including optical, magnetic, catalytic and mechanical characteristics, which make them highly useful in applications such as biosensors, catalysis, environmental monitoring and energy storage. However, despite their numerous advantages, the potential environmental and health impacts of nanoparticles must be carefully evaluated since their small size and high reactivity may lead to interactions with biological systems [9]. Therefore, the development of eco-friendly synthesis methods and the study of nanoparticle properties remain important areas of research in modern nanoscience.
2. Methodology
2.1. Seaweed Collection and Identification
Seaweed samples (Gracilaria edulis) were collected from the intertidal rocky shores of the Mandapam coast, Gulf of Mannar, India. Immediately after collection, the samples were rinsed with seawater to remove adhering sand, debris, and epiphytes. Any remaining epiphytes were gently removed using a soft brush.
In the laboratory, the samples were washed thoroughly with freshwater and then with distilled water to ensure the complete removal of salts and other impurities. The cleaned seaweeds were shade-dried at room temperature for five days. After drying, the material was cut into small pieces and used for further analysis, following the method described by [10].
2.2. Preparation of Seaweed Extract
For the preparation of the seaweed extract, 10 g of dried seaweed material was added to 100 mL of distilled water and heated in a water bath at 70 °C for 10 minutes. The mixture was then filtered using Whatman No. 1 filter paper, and the filtrate was collected in a 250 mL Erlenmeyer flask.
The resulting extract was stored at 4 °C and used for further experiments. The same procedure was followed for all seaweed samples, as described by [12].
2.3. Biosynthesis of Cadmium Sulfide (CdS) Nanoparticles
Cadmium sulfide (CdS) nanoparticles were synthesized using Gracilaria edulis seaweed extract as both a reducing and stabilizing agent. A 0.01 M solution of cadmium chloride was prepared in 100 mL of deionized water. To this, 5 mL of the seaweed extract was added under continuous stirring.
Subsequently, sodium sulfide solution was added dropwise to the reaction mixture while maintaining constant stirring. The reaction was allowed to proceed for 1 hour, leading to the formation of a yellow-colored precipitate, which indicates the formation of CdS nanoparticles [13].
2.4. Purification of Nanoparticles
Centrifugation of Metal Nanoparticles
The synthesized nanoparticles were purified by centrifugation to remove biological impurities and unreacted components. The reaction mixture was centrifuged at 12,000–15,000 rpm for 30 minutes. The collected nanoparticle precipitate was washed with sterile deionized water to remove residual biological molecules. The centrifugation and washing process was repeated three times to obtain purified nanoparticles.
3. Characterization Techniques
3.1. Seaweed
Seaweeds are primitive, non-flowering marine plants that lack true roots, stems and leaves. They are an important renewable marine resource and are widely distributed in marine environments. Seaweeds occur in intertidal, shallow and deep sea waters up to a depth of about 150 m. They are commonly found attached to rocky substrates, dead corals, pebbles and other solid surfaces, and may also grow as epiphytes on other marine plants.Based on their pigment composition, morphology and anatomical characteristics, seaweeds are broadly classified into three major groups: green algae (Chlorophyta), brown algae (Phaeophyta) and red algae (Rhodophyta). These groups differ in their biochemical composition, ecological distribution and biological functions.
3.2. Distribution
India possesses a rich diversity of marine algae along its extensive coastline. A large number of seaweed species are found along the southeast coast of Tamil Nadu, particularly in the region extending from Rameswaram to Kanyakumari, including the 21 islands of the Gulf of Mannar. Abundant seaweed populations are also reported from coastal regions such as Okha, Dwarka, Porbandar, Veraval, Diu and Gopnath along the Gujarat coast. In addition, significant seaweed resources occur in Lakshadweep, Andaman and Nicobar Islands, and along the western and eastern coasts of India including Mumbai, Ratnagiri, Goa, Karwar, Varkala, Vizhinjam and Visakhapatnam. Coastal lagoons such as Pulicat Lake and Chilika Lake also support diverse marine algae populations.
In Indian waters, approximately 271 genera and more than 1100 species of marine algae have been reported. These include about 213 species of green algae, 289 species of brown algae, 431 species of red algae and 220 species of blue-green algae. Later surveys have reported around 844 species belonging to the same number of genera in Indian coastal waters [14].
The southern coast of India is particularly rich in seaweed diversity. More than 200 species of seaweeds have been reported from this region. Surveys conducted by research organizations such as the Central Salt and Marine Chemicals Research Institute and the Central Marine Fisheries Research Institute have highlighted the significant potential of seaweed resources in the Gulf of Mannar region for industrial and biotechnological applications [15].
A distribution study of macroalgae conducted in southern districts of Tamil Nadu, including Kanyakumari, Tirunelveli, Thoothukudi and Ramanathapuram, recorded 57 taxa of marine algae belonging to 37 genera. These included 25 taxa of Rhodophyta, 18 taxa of Chlorophyta and 14 taxa of Phaeophyta. The highest number of algae species was reported from the Bay of Bengal coast (67.7%), followed by the Indian Ocean coast (25%) and the Arabian Sea coast (8%) [16].
3.3. Selection of Seaweed
Among the various marine algae available in the Gulf of Mannar region, the red alga Gracilaria edulis was selected for the present study due to its availability and potential biological activity. The samples were collected from the intertidal region of the Mandapam coast in the Gulf of Mannar, Rameswaram, Tamil Nadu. The collected seaweed was used for the biosynthesis of metal nanoparticles (Figure 1).
3.4. Taxonomical Classification
Gracilaria edulis is a species of red algae belonging to the division Rhodophyta. It is widely distributed in tropical marine environments and is commonly used in biotechnology and marine-based studies due to its rich bioactive compounds.
The taxonomical classification of Gracilaria edulis is as follows:
Kingdom: Plantae
Phylum: Rhodophyta
Class: Florideophyceae
Order: Gracilariales
Family: Gracilariaceae
Genus: Gracilaria
Species: Gracilaria edulis
3.5. Origin and Geographic Distribution
Gracilaria edulis is commonly found in tropical regions of the Indian and Pacific Oceans. In Southeast Asia, it has been reported from coastal areas of northern Burma (Myanmar), Malaysia, Indonesia, the Philippines and Papua New Guinea [17]. In India, it is widely distributed along the southeastern coast, particularly in the Gulf of Mannar region, where environmental conditions favour its growth.
3.6. Description
Gracilaria edulis is a marine red alga characterized by erect, flattened thalli that grow up to about 20 cm in height, although smaller individuals are commonly observed. The thalli are typically bright orange to red in color and arise from small discoid holdfasts that attach firmly to rocky substrates.
The thallus consists of several erect and overlapping flattened branches. Branching usually occurs in a single plane and is irregularly pinnate-alternate, forming rounded axils. The diameter of the primary branches generally does not exceed 7 mm. The terminal branches present at the distal portion of the thalli often show slightly expanded, curved or inrolled tips, while the lower lateral branchlets bear simple acute teeth.
In cross-section, the central axis is clearly visible and relatively narrow. It is surrounded by a large-celled pseudo-parenchymatous medulla and an outer cortex composed of smaller cells. In some regions of the thalli, the outer cortex contains numerous slightly enlarged gland cells.
Two morphological forms may occur within the same habitat: a rigid cartilaginous form and a more flexible, gelatinous form with finer branches. The life cycle of G. edulis is diplo-haplontic, triphasic and isomorphic.
Reproductive structures are present on fertile thalli. Tetrasporangia and spermatangia occur as pale patches on the surface, while cystocarps appear as wart-like nemathecia mainly along the edges of the thallus. The tetrasporangia form a solid layer without interspersed sterile cells and are zonately divided, measuring approximately 6 μm × 45 μm.
Female nemathecia contain semi-globose cystocarps that develop several gonimolobes separated by thin sterile partitions. The released carpospores are globose and measure about 9 μm in diameter. Spermatangia occur superficially in pustule-like regions that resemble pale tetrasporangial patches.
3.7. UV–Visible Analysis
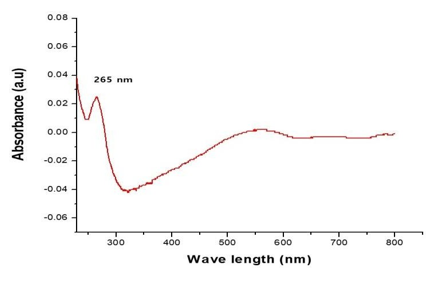
The UV–Visible spectrum of the synthesized CdS nanoparticles was recorded after 12 hours of reaction between CdCl₂ and Na₂S in the presence of Gracilaria edulis seaweed extract. The absorption spectrum was analyzed to confirm nanoparticle formation and to understand their optical properties.
The spectrum exhibited an absorption peak at 265 nm (Figure 3), which is attributed to the formation of CdS nanoparticles. UV–Vis analysis provides insight into particle size and distribution, particularly at the nanoscale.
The observed absorption at 265 nm shows a notable blue shift compared to bulk CdS, which typically absorbs at higher wavelengths. This shift can be attributed to the quantum confinement effect, where the reduction in particle size alters the electronic band structure. The presence of this blue shift indicates the formation of nanosized CdS particles. Similar shifts have been reported in earlier studies [1].
3.8. Fourier Transform Infrared (FTIR) Analysis
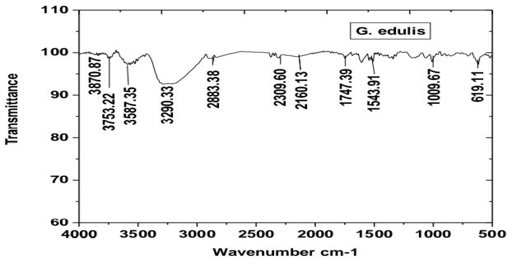
Fourier Transform Infrared (FTIR) spectroscopy was employed to identify the functional groups involved in the synthesis and stabilization of CdS nanoparticles. The synthesized CdS nanoparticles (Figure 4) were dried, and the obtained powder was analyzed in the range of 4000–400 cm⁻¹ using the KBr pellet method. The spectra were recorded using a SHIMADZU IR Prestige-21 FTIR spectrophotometer in transmittance mode with a resolution of 4 cm⁻¹.
FTIR analysis revealed the presence of functional groups such as –OH, –NH, and –COOH, which are attributed to biomolecules present in the Gracilaria edulis extract. These biomolecules play a significant role in the reduction and stabilization of CdS nanoparticles.
The FTIR spectrum exhibited several characteristic absorption peaks corresponding to different functional groups. The broad band observed around 3290 cm⁻¹ is assigned to –OH stretching vibrations, indicating the presence of alcohol or phenolic groups. A prominent peak at 1619 cm⁻¹ corresponds to C=C stretching vibrations, suggesting the presence of organic compounds associated with the nanoparticles [18]. The bands in the region of 1442–1357 cm⁻¹ are attributed to C=O stretching vibrations, while the peak at 1152 cm⁻¹ corresponds to ester functional groups. The absorption band at 1112 cm⁻¹ is assigned to C–H bending vibrations.
Importantly, a distinct peak observed at 619 cm⁻¹ is characteristic of Cd–S bond formation, confirming the successful synthesis of CdS nanoparticles [19]. The FTIR spectrum was further compared with that of bulk CdS, showing similar structural features and thereby supporting the formation of CdS nanoparticles.
3.9. Scanning Electron Microscope (SEM) Analysis
Scanning Electron Microscopy (SEM) with a JEOL JSM-6610LV instrument. The SEM images (Figure 5) clearly show the formation of CdS nanoparticles using Gracilaria edulis extract.
The nanoparticles were predominantly spherical in shape with slight agglomeration. Such aggregation is commonly observed in biologically synthesized nanoparticles due to the presence of biomolecules acting as stabilizing agents. The particle size was found to be in the range of 1-2 µm.
Overall, the SEM micrographs confirm the successful formation of CdS nanoparticles with well-defined morphology.
3. X-ray Diffraction (XRD) Analysis
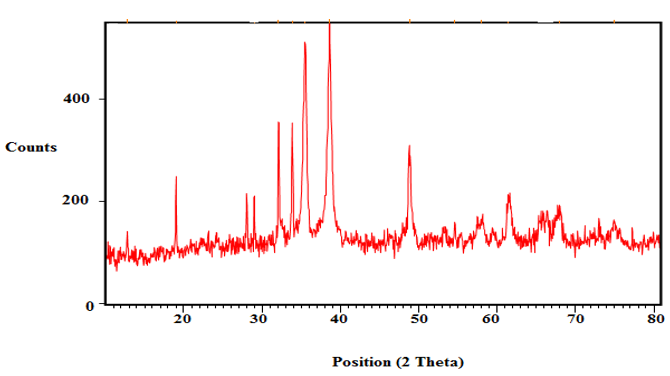
The crystalline nature of the synthesized CdS nanoparticles was investigated using X-ray diffraction (XRD) analysis. The XRD pattern of the synthesized CdS nanoparticles is shown in Figure 6. The diffraction peaks observed at 19.07°, 32.11°, 33.86°, 35.51°, 38.62° and 48.76° correspond to the crystalline planes of cadmium sulfide nanoparticles, confirming their crystalline structure [21].
The sharp and well-defined diffraction peaks indicate the high crystallinity of the synthesized nanoparticles. The peak broadening observed in the XRD pattern suggests that the particles are in the nanometer range.
The average crystallite size of the CdS nanoparticles was estimated using the Debye–Scherrer equation:
whereD=crystallite size,λ = wavelength of the X-ray radiation,β = full width at half maximum (FWHM) of the diffraction peak,θ = Bragg’s angle.
Using this equation, the average crystallite size of the synthesized CdS nanoparticles was calculated to be approximately 11.31 nm, confirming their nanoscale nature. This value is smaller than or comparable to previously reported sizes for biologically synthesized CdS nanoparticles (20–50 nm), indicating the strong reducing and stabilizing capability of Gracilaria edulis extract, which promotes the formation of finer nanoparticles.
Conclusion
In this study, cadmium sulfide (CdS) nanoparticles were successfully synthesized using an eco-friendly green approach with Gracilaria edulis seaweed extract acting as both a reducing and stabilizing agent. The formation of CdS nanoparticles was confirmed by UV–Visible spectroscopy, which showed a characteristic absorption peak at 265 nm, indicating the quantum confinement effect. FTIR analysis revealed the presence of bioactive functional groups in the seaweed extract, which are responsible for the reduction and stabilization of the nanoparticles. XRD results confirmed the crystalline nature of the CdS nanoparticles, with an average crystallite size of approximately 11.31 nm calculated using the Debye–Scherrer equation.
SEM analysis indicated that the particles were predominantly spherical with noticeable agglomeration, forming clusters in the range of 1–2 µm. This larger size is attributed to the aggregation of smaller primary nanoparticles, which is common in biologically synthesized systems. Overall, the results demonstrate that Gracilaria edulis extract provides a simple, cost-effective, and environmentally sustainable method for the synthesis of CdS nanoparticles, with potential applications in optoelectronic, catalytic, and environmental fields.
- Abbas R, et al. (2024) Silver nanoparticles: synthesis, characterization and emerging applications in nanotechnology. Nanomaterials.
- Zehra SH, et al. (2025) Recent advances in green synthesis of silver nanoparticles and their biomedical applications. Nanomaterials.
- Novoselov KS, et al. (2023) Two-dimensional materials and their applications in nanotechnology. Nature.
- Wang ZL, et al. (2023) Progress in nanomaterials for energy harvesting and environmental applications. Advanced Materials.
- Rizwana H, et al. (2023) Green synthesis and antimicrobial activity of plant-mediated silver nanoparticles. Scientific Reports.
- Jangid H, et al. (2024) Biomedical applications and antimicrobial mechanisms of silver nanoparticles. Frontiers in Pharmacology.
- Shahzadi S, et al. (2025) Green synthesis of silver nanoparticles using plant extracts and their catalytic and antibacterial properties. RSC Advances.
- Asefian S, et al. (2024) Eco-friendly synthesis of silver nanoparticles using biological methods and their antibacterial activity. BMC Biotechnology, 2024.
- Kumar V, et al. (2023) Metal and metal oxide nanoparticles: synthesis, properties and environmental applications. Materials Science and Engineering.
- Rai M, et al. (2024) Silver nanoparticles as antimicrobial agents: mechanisms and biomedical applications. Applied Microbiology and Biotechnology.
- Manilal A, Sujith S, Selvin J, Kiran GS, Shakir C (2009) Biopotentials of seaweeds collected from the southwest coast of India. Journal of Marine Science and Technology, 17: 67–73.
- Rengasamy KRR, Rajendran A, Natarajan J, Ahn YS (2013) Green synthesis of silver nanoparticles using seaweed extract and their antibacterial activity. Applied Nanoscience, 3: 353–60.
- Murugadoss G, Rajamannan B (2010) Synthesis and characterization of cadmium sulfide (CdS) nanoparticles. Chalcogenide Letters, 7: 693–8.
- Oza RM, Zaidi SH (2000) A Revised Checklist of Indian Marine Algae. Central Salt and Marine Chemicals Research Institute (CSMCRI), Bhavnagar, India.
- Jha B, Reddy CRK, Thakur MC, Rao MU (2009) Seaweeds of India: The Diversity and Distribution of Seaweeds of Gujarat Coast. Springer, Dordrecht.
- Rao PS, Mantri VA, Jha B (2014) Seaweed research and utilization in India. Botanica Marina, 57: 1–9.
- Guiry MD, Guiry GM (2023) AlgaeBase. World-wide electronic publication. National University of Ireland, Galway.
- Jha B, Reddy CRK, Thakur MC, Rao MU (2009) Seaweeds of India: The Diversity and Distribution of Seaweeds of Gujarat Coast. Springer, Dordrecht.
- Rizwana H, et al. (2023) Identification of functional groups in green-synthesized nanoparticles using FTIR. Scientific Reports, 13: 11204.
- Wang ZL, et al. (2023) FTIR analysis of bio-synthesized silver and cadmium nanoparticles. Advanced Materials, 35: 2204567.
- Jangid H, et al. (2024) Morphological studies of plant-mediated CdS and ZnO nanoparticles using SEM. Frontiers in Pharmacology, 15: 1012568.
- Shahzadi S, et al. (2025) Surface morphology and size analysis of biosynthesized nanoparticles via SEM. RSC Advances, 15: 4572–81.
- Asefian S, et al. (2024) Crystallographic analysis of green-synthesized metal nanoparticles using XRD. BMC Biotechnology, 24: 112.



Tables at a glance
Figures at a glance