Synthesis and Characterization of New 5-Benzylidene Thioimidazolone Esters
Received Date: January 27, 2026 Accepted Date: February 13, 2026 Published Date: February 16, 2026
doi: 10.17303/jocs.2026.4.101
Citation: Simon S Mnyakeni Moleele, Unarine Tshishonga, Mpelegeng V Bvumbi (2026) Synthesis and Characterization of New 5-Benzylidene Thioimidazolone Esters. J Org Chem Chem Sci 4: 1-13
Abstract
2-Thiohydantoin-containing compounds have garnered significant attention from researchers worldwide due to their bioactivity and therapeutic potential. In this project twenty two new 2-thiohydantoin derivatives (10a-v) were synthesized from the reaction of 5-arylidene-2-thiohydantoins (8a-e) with ethyl (2-chloroacetyl)-glycinate (9a), -alaninate (9b),-butanoate (9c), -valinate (9d), and -norvalinate (9e). The structures of synthesized compounds were characterized using Fourier-transform infrared spectroscopy (FT-IR), 1H- and 13C-nuclear magnetic resonance (NMR) spectroscopy, and mass spectrometry
Keywords: Imidazolone; Alaninate; Valinate; Norvalinate
Introduction
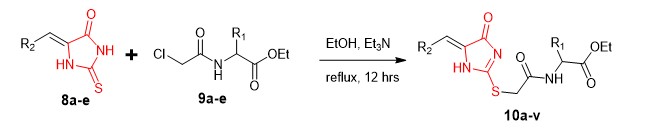
Thiohydantoins (thioxoimidazolidinones) are hydantoins (2, 4-imidazolidinediones) having one or both carbonyl groups substituted by thiocarbonyl groups. They are an important family of chemicals in pharmaceutical chemistry because they occur in a variety of pharmacologically active molecules with high bioactivities and are used in therapeutic medications. There are three known thiohydantoins, namely 2-thiohydantoin (2-thioxoximidazolin-4-one) [1], 4-thiohydantoin (4-thioxoximidazolin-2-one) [2] and 2, 4-dithiohydantoin (imidazolidine-2, 4-dithione) [3] [Figure 1].
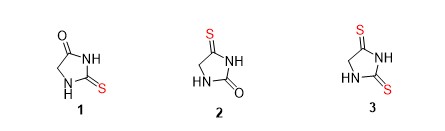
The 2-thiohydantoins are well-known analogues due to their numerous applications as intermediates and reagents in organic synthesis, pharmaceuticals and agricultural processes. Some of the pharmaceutical purposes that involve these active molecules are indicated in Figure 3. Buchynskyy et al. discovered the thiohydantoins 1-benzyl-3-aryl-2-thiohydantoins [4] as anti-Trypanosoma brucei agents. The most active analogues were reported to be 1-(4-fluorobenzyl)-3-(4-dimethylamino-3-chlorophenyl)-2-thiohydantoin (4a) and 1-(4-fluorobenzyl)-3-(4-dimethylamino-3-methoxyphenyl)-2-thiohydantoin (4b) and displayed high antiparasitic activity with IC50 3.2 and 1.9 μM respectively. [] Raghu Raj and co-workers reported the synthesis of a series of 7-chloroquinoline-thiohydantoins derivatives as antimalarial agents. Compounds 5a-d exhibited inhibitory activity against P. falciparum with promising low IC50 values of 39.84-57.21 μM and low toxicity [2]. Kiec´-Kononowicz reported 5-arylidine-2-thiohydantoins as mycobacterial agents. Compounds (6a) and (6b) displayed more than 90% inhibition of Mycobacterium tuberculosis growth with IC50 of 6.7 and 4.5 μM respectively [2]. Han and co-workers synthesized 5-arylidene-2-thiohydantoin derivatives with a substituent sulfonyl group in position 4 as a linker between two phenyl groups (e.g compound 7) and they exhibited high antifungal activity [2].
Our research group has embarked on a journey to synthesise 5-membered ring-containing compounds and test their biological activity against diabetes. With encouraging results obtained from glitazone, rhodanine and hydantoin-containing compounds, we envisaged that 2-thiohydantoin containing compounds could be active against diabetes. Thus this project focused on the synthesis of 2-thiohydantoin-containing compounds.
Results and Discussion
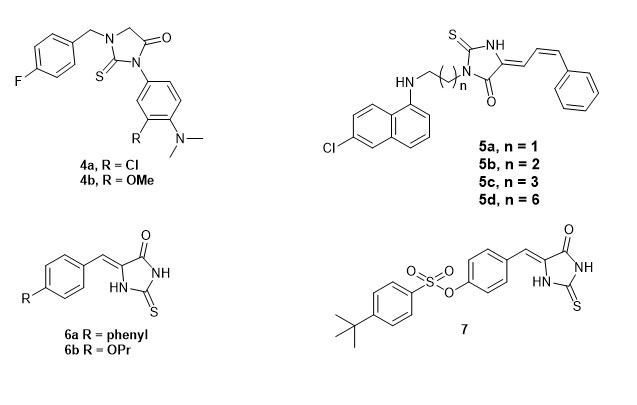
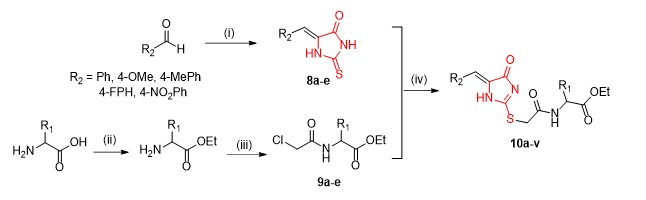
The synthesis of the desired ethyl [2-(5-benzylidene)-5-oxo-2-thioimidazolidin-3-yl]acetyl esters (10a-v) began by reacting commercial 2-thiohydantoin with different benzaldehydes to give benzylidene thiohydantoins 8a–e in good yields. (Scheme 1).[5, 6, 7, 8, 9]Concurrently, compounds 9a–e were synthesized by first protecting the amino acids with thionyl chloride in the presence of ethanol and subsequently reacting the intermediates with chloroacetyl chloride in the presence of sodium bicarbonate in good yields. , Finally, substitution reactions between benzylidene thiohydantoins 8 and compounds 9 followed by purification by column chromatography afforded the targeted ethyl [2-(4-benzylidene)-5-oxo-2-thioimidazolidin-3-yl]acetyl esters 10a–v in moderate to good yields (Table 1). These compounds were characterized by NMR and IR spectroscopies together with mass spectroscopic analyses. 1H NMR spectra of the products showed a singlet corresponding to one hydrogen (NH) signal at ~12 ppm, consistent with monoalkylation. The regioselectivity of S-alkylation of thiohydantoins 8 at the 2-position was established by analogy with the literature. [7] S-alkylation was further confirmed using the 1H-13C HMBC analysis of compound 10q. The spectrum showed coupling between a quaternary carbon at 160.92 ppm, corresponding to the 2-position of the imidazolone ring, and a singlet accounting for two protons at 4.12 ppm. Also, in the 1H NMR spectra of these compounds, doublets accounting for one proton ~8.7 ppm were observed, confirming the presence of the amidic proton. For the glycinate compounds 10a, 10e, 10j and 10o, this peak appeared as a triplet. The spectra were also characterized by a singlet accounting for two protons ~4.1 ppm confirming the methylene protons adjacent to the ring nitrogen. The 13C NMR spectra of compounds 10 were characterized by three carbonyl carbon signals and one thiocarbonyl carbon signals at ~172 ppm. IR spectra of the final compounds were characterized with peaks at ~1700 cm−1 and 1230 cm−1 confirming the C=O and C=S stretches respectively.
Reagents and conditions (i) Thiohydantoin, sodium acetate, acetic acid, reflux, (ii) SOCl2, EtOH, reflux, (iii) chloroacetyl chloride, H2O, NaHCO3, room temp, (iv) EtOH, Et3N, reflux.
Experimental Section
General All reagents used were analytical grade reagents from Sigma-Aldrich and Fluka. Thin-layer chromatography (TLC) was carried out using Macherey-Nagel Alugram Sil G/UV254 plates, pre-coated with 0.25mm silica gel 60. Detection was done under ultraviolet light at 254 nm. 1H NMR (400 MHz), 13C NMR (100 MHz) and 19F NMR (376.5 MHz) spectra were recorded on a Bruker 400 MHz spectrometer using DMSO-d6 or CDCl3 as solvents and TMS at 0.00 ppm as an internal standard. Values for the chemical shifts are expressed in parts per million (ppm). The following abbreviations are used: br.s for broad singlet, s for singlet, d for doublet, dd for doublet of doublets, q for quartet, quint for quintet and m for multiplet and (J) for coupling constants measured in hertz (Hz). All the melting points were determined on a Buchi melting point B-540 apparatus using capillary tubes. Infrared spectra were run on a Bruker platinum 22 vector Fourier Transform spectrometer (FTIR). Mass spectra (High Resolution) were recorded on a Waters GCT using a column called the Restek Rxi Wintegra Guard (15 m, 0.25 mm ID, 0.25 μm film thickness) mass spectrometer. The samples were dissolved in acetonitrile and injected at a volume of 1 μl at a mode of 10:1 and temperature of 280 °C. The source temperature was 100 °C and the de-solvation temperature was set at 300 °C. Helium gas was used as the carrier gas. The software used to control the hyphenated system and to do all data manipulation was MassLynx 4.1 (SCN 704).
General Synthetic Procedure for Target Compounds (10a-V)
A mixture of Knoevenagel condensation reaction products (8a-e) (1 mmol) dissolved in ethanol and triethylamine (2 mmol) was heated to 80 °C and stirred for 15 minutes. The reaction mixture was then refluxed for 12 hours with ethyl amino esters (9a-e) (1 mmol). After the reaction had cooled to room temperature, the solvent was removed in vacuo and the products were purified by column chromatography.
Ethyl (2-((5-benzylidene-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) glycinate (10a)
A reaction of 5-benzylidene-2-thiohydantoin 8a (0.50 g, 2.48 mmol) and ethyl (2-chloroacetyl)glycinate 9a (0.44 g, 2.48 mmol) gave compound 10a as a yellow solid (0.53 g, 63.79 %); m.p 187‒189 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 11.89 (s, 1H, NH), 8.83 (t, 1H, J 5.64 Hz, NH), 8.17 (d, 2H, J 7.32 Hz, 2 × ArH), 7.45‒7.37 (m, 3H, 3 × ArH), 6.76 (s, 1H, CCH), 4.14 (s, 2H, NCH2), 4.09 (q, 2H, J 7.12 Hz, OCH2CH3), 3.90 (d, 2H, J 5.72 Hz, NHCH2), 1.17 (t, 3H, J 7.08 Hz, CH3), 13C NMR (100 MHz, DMSO-d6) δC (ppm) 171.02 (COO), 169.97 (C=S), 167.57 (CONH), 164.26 (CON), 139.52 (ArC), 134.62 (ArC), 132.06 (ArCH), 130.02 (ArCH), 129.07 (ArCH), 121.70 (CCH), 60.99 (OCH2CH3), 41.63 (NCH2), 33.80 (NHCH2), 14.47 (CH3). IR (KBr cm-1): 3215 (N-H), 2977 (C-H), 2860 (C-H), 1753 (C=O), 1708 (C=O), 1663 (C=O), 1605 (C=C), 1226 (C=S),1170 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C16H17N3O4S: 348.0939; found: 348.1018.
Ethyl (2-((5-benzylidene-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) alaninate (10b)
A reaction of 5-benzylidene-2-thiohydantoin 8a (0.50 g, 2.48 mmol) and ethyl (2-chloroacetyl)alaninate 9b (0.47 g, 2.48 mmol) gave compound 10b as a grey solid (0.51 g, 57.95 %); m.p 180‒182 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 11.94 (s, 1H, NH), 8.81 (d, 1H, J 6.8 Hz, NH),8.18 (d, 2H, J 7.2, 2 × ArH), 7.37‒7.45 (m, 3H, 3 × ArH), 6.76 (s, 1H, CCH), 4.28 (quintet, 1H, J 7.2 Hz, CHCH3), 4.18 (s, 2H, NCH2), 4.06 (q, 2H, J 3.2 Hz, OCH2CH3), 1.30 (d, 3H, J 7.2 Hz, CHCH3), 1.15 (t, 3H, J 7.04 Hz, OCH2CH3), 13C NMR (100 MHz, DMSO-d6) δC (ppm) 172.71 (C=S), 171.01 (COO), 166.86 (CONH), 164.40 (CON), 139.52 (CCH), 134.64 (ArC), 132.08 (2 × ArCH), 130.62 (2 × ArCH), 129.23 (ArCH), 121.60 (CCH), 60.99 (OCH2CH3), 40.61 (NCH2), 33.83 (CHCH3), 17.50 (CHCH3), 14.43 (OCH2CH3). IR (KBr cm-1): 3312 (N-H), 2921 (C-H), 2851 (C-H), 1732 (C=O), 1710 (C=O), 1637 (C=O),1535 (C=C), 1272 (C=S),1190 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C17H19N3O4S: 362.1096; found: 362.1175.
Ethyl 2-(2-((5-benzylidene-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetamido) butanoate (10c).
A reaction of 5-benzylidene-2-thiohydantoin 8a (0.50 g, 2.48 mmol) and ethyl 2-(2-chloroacetamido)butanoate 9c (0.51 g, 2.48 mmol) gave compound 10c as a white solid (0.65 g, 71.30%); m.p 182‒184 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 11.64 (s, 1H, NH), 8.72 (d, 1H, J 7.32 Hz, NH), 8.18 (d, 2H, J 7.2, ArH), 7.38‒7.45 (m, 3H, 3 × ArH), 6.76 (s, 1H, CCH), 4.20 (quintet, 1H, J 7.02 Hz, CHCH2CH3), 4.14 (s, 2H, NCH2), 4.07 (q, 2H, J 5.7 Hz, OCH2CH3), 1.78‒1.53 (m, 2H, CHCH2CH3), 1.17 (q, 3H, J 7.20 Hz, CHCH2CH3), 1.15 (t, 3H, J 7.32 Hz, OCH2CH3), 13C NMR (100 MHz, DMSO-d6) δC (ppm) 172.08 (COO), 171.00 (C=S), 167.18 (CONH), 164.41 (CON), 139.48 (NHCCH), 134.60 (NHCCHC), 132.08 (ArCH), 130.62 (ArCH), 129.01 (ArCH), 121.64 (CCH), 61.02 (OCH2CH3), 54.33 (CHCH2CH3), 46.11 (NCH2), 24.63 (CHCH2CH3), 14.48 (CHCH2CH3), 10.57 (OCH2CH3). IR (KBr cm-1): 3296 (N-H), 2910 (C-H), 2805 (C-H), 1746 (C=O), 1711 (C=O), 1656 (C=O),1595 (C=C), 1255 (C=S),1173 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C18H23N3O4S: 376.1252; found: 376.1333.
Ethyl (2-((5-benzylidene-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) norvalinate (10d).
A reaction of (5-benzylidene-2-thiohydantoin 8a (0.50 g, 2.48 mmol) and ethyl (2-chloroacetyl)norvalinate 9e (0.54 g, 2.48 mmol) gave compound 10d as a white solid (0.53 g, 55.79 %), m.p 175‒177 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 11.90 (s, 1H, NH), 8.72 (d, 1H, J 7.4 Hz, NH), 8.18 (d, 2H, J 7.2, 2 × ArH), 7.39‒7.45 (m, 3H, 3 × ArH), 6.77 (s, 1H, CCH), 4.24 (quintet, 1H, J 7.8 Hz, CHCH2CH2CH3), 4.17 (s, 2H, NCH2), 4.06 (q, 2H, J 6.1 Hz, OCH2CH3), 1.69‒1.51 (m, 2H, CHCH2CH2CH3),1.32‒1.22 (m, 2H, CHCH2CH2CH3), 1.15 (t, 3H, J 7.1 Hz, OCH2CH3), 0.75 (t, 3H, J 7.2 Hz, CHCH2CH2CH3), 13C NMR (100 MHz, DMSO-d6) δC (ppm) 172.29 (COO), 170.99 (C=S), 167.17 (CONH), 164.36 (CON), 139.46 (NHCCH), 134.60 (CCHC), 132.09 (ArCH), 130.62 (ArCH), 129.07 (ArCH), 121.70 (CCH), 60.90 (OCH2CH3), 52.57 (CHCH2CH2CH3), 46.01 (NCH2), 33.61 (CHCH2CH2CH3), 18.95 (CHCH2CH2CH3), 14.46 (CHCH2CH2CH3),13.79 (OCH2CH3). IR (KBr cm-1): 3295 (N-H), 2956 (C-H), 2840 (C-H), 1709 (C=O), 1638 (C=O), 1537 (C=O), 1505 (C=C), 1248 (C=S),1174 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C19H23N3O4S: 390.1409; found: 390.1474.
Ethyl (2-((5-(4-methylbenzylidene)-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) glycinate (10e).
A reaction of 5-(4-methylbenzylidene)-2-thiohydantoin 8b (0.50 g, 2.29 mmol) and ethyl (2-chloroacetyl)glycinate 9a (0.41 g, 2.29 mmol) gave compound 10e as a yellow solid (0.54 g, 65.64%); m.p 165‒167 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 11.83 (s, 1H, NH), 8.80 (t, J 5.5 Hz, 1H, NH), 8.06 (d, J 7.8 Hz, 2H, 2 × ArH), 7.24 (d, J 7.9 Hz, 2H, 2 × ArH), 6.73 (s, 1H, CCH), 4.12 (s, 2H, NCH2), 4.08 (q, J 7.1 Hz, 1H, OCH2CH3), 3.91 (d, J 5.7 Hz, 2H, NHCH2), 2.35 (s, 3H, ArCH3), 1.18 (t, J 7.1 Hz, 3H, OCH2CH3). 13C NMR (100 MHz, DMSO-d6) δC (ppm) 171.02 (COO), 169.98 (C=S), 167.60 (CONH), 163.46 (CON), 140.05 (NHCCH), 138.86 (ArC), 132.10 (2 × ArCH), 131.92 (ArC), 129.76 (2 × ArCH), 121.96 (CCH), 60.98 (OCH2CH3), 41.64 (NHCH2), 33.75 (NCH2), 21.63 (ArCH3), 14.48 (OCH2CH3). IR (KBr cm-1): 3297 (N-H), 2915 (C-H), 2805 (C-H), 1757 (C=O), 1711 (C=O), 1664 (C=O),1605 (C=C), 1315 (C=S),1171 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C17H19N3O4S: 362.4215; found: 362.1851.
Ethyl (2-((5-(4-methylbenzylidene)-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) alaninate (10f).
A reaction of 5-(4-methylbenzylidene)-2-thiohydantoin 8b (0.50 g, 2.29 mmol) and ethyl (2-chloroacetyl)alaninate 9b (0.44 g, 2.29 mmol) gave compound 10f as a yellow solid (0.43 g, 49.79 %); m.p 203‒205 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 11.82 (s, 1H, NH), 8.80 (d, J 6.8 Hz, 1H, NH), 8.08 (d, J 7.8 Hz, 2H, 2 × ArH), 7.24 (d, J 7.8 Hz, 2H, 2 × ArH), 6.74 (s, 1H, CCH), 4.28 (q, J 6.9 Hz, 1H, CHCH3), 4.12 (s, 2H, NCH2), 4.09 (m, J 7.1 Hz, 2H, OCH2CH3), 2.35 (s, 3H, ArCH3), 1.30 (d, J 7.2 Hz, 3H, CHCH3), 1.16 (t, J 7.1 Hz, 3H, OCH2CH3). 13C NMR (100 MHz, DMSO-d6) δC (ppm) 172.71 (COO), 171.01 (C=S), 166.90 (CONH), 163.57 (CON), 140.04 (NHCCH), 138.85 (ArC), 132.12 (2 × ArCH), 131.92 (ArC), 129.77 (2 × ArCH), 121.92 (CCH), 61.00 (OCH2CH3), 48.67 (CHCH3), 33.78 (NCH2), 21.65 (ArCH3), 17.50 (CHCH3), 14.42 (OCH2CH3). IR (KBr cm-1): 3310 (N-H), 2975 (C-H), 2840 (C-H), 1732 (C=O), 1708 (C=O), 1640 (C=O),1603 (C=C), 1273 (C=S),1190 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C18H21N3O4S: 376.1252; found: 376.1321.
Ethyl 2-(2-((5-(4-methylbenzylidene)-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetamido) butanoate (10g).
A reaction of 5-(4-methylbenzylidene)-2-thiohydantoin 8b (0.50 g, 2.29 mmol) and ethyl 2-(2-chloroacetamido)butanoate 9c (0.48 g, 2.29 mmol) gave compound 10g as a brown solid (0.61 g, 69.22%); m.p 167‒169 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 11.85 (s, 1H, NH), 8.72 (d, J 7.4 Hz, 1H, NH), 8.07 (d, J 8.1 Hz, 1H, 2 × ArH), 7.24 (d, J 8.1 Hz, 2H, 2 × ArH), 6.73 (s, 1H, CCH), 4.19 (q, J 7.9 Hz, 1H, CHCH2CH3), 4.15 (s, 2H, NCH2), 4.07 (q, J 5.2 Hz, 2H, OCH2CH3), 2.34 (s, 3H, ArCH3), 1.83–1.52 (m, 2H, CHCH2CH3), 1.20 (t, J 7.3 Hz, 3H, OCH2CH3), 0.85 (t, J 7.4 Hz, 3H, CHCH2CH3). 13C NMR (100 MHz, DMSO-d6) δC (ppm) 172.08 (COO), 170.98 (C=S), 167.21 (CONH), 163.61 (CON), 140.02 (NHCCH), 138.82 (ArC), 132.12 (2 × ArCH), 131.90 (ArC), 129.76 (2 × ArCH), 121.93 (CCH), 60.95 (OCH2CH3), 54.32 (CHCH2CH3), 33.72 (NCH2), 24.88 (CHCH2CH3), 21.66 (ArCH3), 14.48 (OCH2CH3), 10.57 (CHCH2CH3). IR (KBr cm-1): 3021 (N-H), 2896 (C-H), 2810 (C-H), 1724 (C=O), 1650 (C=O), 1598 (C=O),1476 (C=C), 1229 (C=S),1155 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C19H23N3O4S: 390.1426; found: 390.1473.
Ethyl (2-((5-(4-methylbenzylidene)-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) valinate (10h).
A reaction of 5-(4-methylbenzylidene)-2-thiohydantoin 8b (0.50 g, 2.29 mmol) and ethyl (2-chloroacetyl)valinate 9d (0.51 g, 2.29 mmol) gave compound 10h as a white solid (0.54 g, 58.94%); m.p 208‒210 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 11.83 (s, 1H, NH), 8.57 (d, J 8.2 Hz, 1H, NH), 8.07 (d, J 7.8 Hz, 2H, 2 × ArH), 7.24 (d, J 7.9 Hz, 2H, 2 × ArH), 6.74 (s, 1H, CCH), 4.23 (t, J 4.4 Hz, CHCH(CH3)2), 4.20 (s, 2H, NCH2), 4.08 (q, J 6.2 Hz, 2H, OCH2CH3), 2.34 (s, 3H, ArCH3), 2.04 (m, 1H, CHCH(CH3)2), 1.16 (t, J 7.1 Hz, 3H, OCH2CH3), 0.86 (d, J 6.8 Hz, 6H, CHCH(CH3)2). 13C NMR (100 MHz, DMSO-d6) δC (ppm) 171.61 (COO), 171.00 (C=S), 167.42 (CONH), 163.68 (CON), 140.02 (NHCCH), 138.81 (ArC), 132.10 (2 × ArCH), 131.88 (ArC), 129.78 (2 × ArCH), 121.99 (CCH), 60.92 (OCH2CH3), 58.28 (CHCH(CH3)2), 33.66 (NCH2), 30.56 (CHCH(CH3)2), 21.65 (ArCH3), 19.28 (CHCH(CH3)2), 18.49 (CHCH(CH3)2), 14.50 (OCH2CH3). IR (KBr cm-1): 3280 (N-H), 2965 (C-H), 2820 (C-H), 1736 (C=O), 1699 (C=O), 1673 (C=O),1555 (C=C), 1271 (C=S),1175 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C20H24N3O4S: 404.1565; found: 404.1630.
Ethyl (2-((5-(4-methylbenzylidene)-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) norvalinate (10i)
A reaction of (Z)-5-(4-methylbenzylidene)-2-thiohydantoin 8b (0.50 g, 2.29 mmol) and ethyl (2-chloroacetyl)norvalinate 9e (0.51 g, 2.29 mmol) gave compound 10i as a white solid (0.72 g, 78.31 %); m.p = 201‒203 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 11.83 (s, 1H, NH), 8.69 (d, J 7.5 Hz, 1H, NH), 8.07 (d, J 7.8 Hz, 2H, 2 × ArH), 7.24 (d, J 7.9 Hz, 2H, 2 × ArH), 6.73 (s, 1H, CCH), 4.24 (q, J 7.7 Hz, 1H, NHCH), 4.18 (s, 2H, NCH2), 4.07 (q, J 4.7 Hz, 2H, OCH2CH3), 2.34 (s, 2H, ArCH3), 1.73–1.48 (m, 2H, CH2CH2CH3), 1.31‒1.29 (m, 2H, CH2CH2CH3), 1.15 (t, J 7.1 Hz, 3H, OCH2CH3), 0.77 (t, J 7.3 Hz, 3H, CH2CH2CH3). 13C NMR (100 MHz, DMSO-d6) δC (ppm) 172.30 (COO), 171.01 (C=S), 167.19 (CONH), 163.62 (CON), 139.98 (CCH), 138.81 (C-9), 132.02 (2 × ArCH), 129.76 (2 × ArCH), 121.92 (CCH), 60.96 (OCH2CH3), 52.74 (NHCH), 33.68 (NCH2), 33.63 (CH2CH2CH3), 21.65 (ArCH3), 18.98 (CH2CH2CH3), 14.47 (OCH2CH3), 13.82 (CH2CH2CH3). IR (KBr cm-1): 3302 (N-H), 2957 (C-H), 2921 (C-H), 1732 (C=O), 1709 (C=O), 1640 (C=O),1603 (C=C), 1284 (C=S), 1142 (C-O). HRMS (ESI-TOF)
Ethyl (2-((5-(4-fluorobenzylidene)-4-oxo-4,5-dihydro-1H-imidazol-2-yl) thio) acetyl) glycinate (10j)
A reaction of (Z)-5-(4-fluorobenzylidene)-2-thiohydantoin 8d (0.50 g, 2.24 mmol) and ethyl (2-chloroacetyl)glycinate 9a (0.40 g, 2.24 mmol) gave compound 10j as a yellow solid (0.55 g, 68.89%); m.p = 231‒232 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 11.86 (s, 1H, NH), 8.80 (t, J 5.6 Hz, 1H, NH), 8.25 (dd, JHH 7.2 Hz, 4JFH 6.4 Hz, 2H, 2 × ArH), 7.26 (t, JHH 8.4 Hz, 3JFH 17.2 Hz, 2H, 2 × ArH), 6.78 (s, 1H, CCH), 4.12 (s, 2H, NCH2), 4.07 (q, J 7.1 Hz, 2H, OCH2CH3), 3.91 (d, J 5.7 Hz, 2H, NHCH2), 1.17 (t, J 7.0 Hz, 3H, OCH2CH3). 13C NMR (100 MHz, DMSO-d6) δC (ppm) 170.95 (C=S), 170.01 (COO), 167.57 (CONH), 164.32 (1JFC 258.0 Hz, ArCF), 161.79 (CON), 139.19 (CCH), 134.37 (3JFC 8.0 Hz, 2 × ArCH), 131.35 (ArC), 120.42 (CCH), 116.10 (2JFC 8.0 Hz, 2 × ArCH), 60.97 (OCH2CH3), 41.63 (NHCH2), 33.77 (NCH2), 14.47 (OCH2CH3). IR (KBr cm-1): 3279 (N-H), 2920 (C-H), 2852 (C-H), 1749 (C=O), 1709 (C=O), 1662 (C=O),1597 (C=C), 1220 (C=S), 1155 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C16H16N3O4FS: 366.0845; found: 366.0918.
Ethyl (2-((5-(4-fluorobenzylidene)-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) alaninate (10k).
A reaction of 5-(4-fluorobenzylidene)-2-thiohydantoin 8d (0.50 g, 2.24 mmol) and ethyl (2-chloroacetyl)alaninate 9b (0.44 g, 2.24 mmol) gave compound 10k as a white solid (0.49 g, 59.81%); m.p = 222‒224 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 11.88 (s, 1H, NH), 8.80 (d, J 6.9 Hz, 1H, NH), 8.36 (dd, JHH 7.6 Hz, 4JFH 6.4 Hz, 2H, 2 × ArH), 7.25 (t, JHH 8.8 Hz, 3JFH 17.6 Hz, 2H, 2 × ArH), 6.78 (s, 1H, CCH), 4.28 (quintet, J 7.1 Hz, 1H, CHCH3), 4.12 (s, 2H, NCH2), 4.06 (q, J 7.1 Hz, 2H, OCH2CH3), 1.28 (d, J 7.2 Hz, 3H, CHCH3), 1.15 (t, J 7.1 Hz, 3H, OCH2CH3). 13C NMR (100 MHz, DMSO-d6) δC (ppm) 172.72 (COO), 170.93 (C=S), 166.85 (CONH), 164.35 (1JFC 269.0 Hz, ArCF), 161.76 (CON), 139.21 (CCH), 134.41 (3JFC 8.0 Hz, 2 × ArCH), 131.38 (ArC), 120.37 (CCH), 116.09 (2JFC 21.0 Hz, 2 × ArCH), 60.99 (OCH2CH3), 48.65 (CHCH3), 33.81 (NCH2), 17.51 (CHCH3), 14.41 (OCH2CH3). IR (KBr cm-1): 3305 (N-H), 2922 (C-H), 2851 (C-H), 1746 (C=O), 1713 (C=O), 1643 (C=O),1597 (C=C), 1206 (C=S),1159 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C17H18N3O4FS: 380.1002; found: 380.1068.
Ethyl 2-(2-((5-(4-fluorobenzylidene)-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetamido) butanoate (10l).
A reaction of 5-(4-fluorobenzylidene)-2-thiohydantoin 8d (0.50 g, 2.24 mmol) and ethyl 2-(2-chloroacetamido)butanoate 9c (0.47 g, 2.24 mmol) gave compound 10l as a white solid (0.69 g, 80.81%); m.p = 209‒211 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 11.89 (s, 1H, NH), 8.70 (d, J 7.4 Hz, 1H, NH), 8.53 (t, JHH 7.2 Hz, 4JFH 6.8 Hz, 2H, 2 × ArH), 7.25 (t, JHH 8.4 Hz, 3JFH 17.2 Hz, 2H, 2 × ArH), 6.78 (s, 1H, CCH), 4.20 (q, J 8.4 Hz, 1H, NHCH), 4.13 (s, 2H, NCH2), 4.06 (q, J 7.1 Hz, 2H, OCH2CH3), 1.89–1.46 (m, 2H, CH2CH3), 1.15 (t, J 7.1 Hz, 3H, OCH2CH3), 0.84 (t, J 7.3 Hz, 3H, CH2CH3). 13C NMR (100 MHz, DMSO-d6) δC (ppm) 172.11 (C=S), 170.95 (COO), 167.18 (CONH), 164.38 (1JFC 248.0 Hz, ArCF), 161.75 (CON), 139.18 (CCH), 134.40 (3JFC 8.0 Hz, 2 × ArCH), 131.36 (4JFC 3.0 Hz, ArC), 120.39 (CCH), 116.07 (2JFC 21.0 Hz, 2 × ArCH), 60.95 (OCH2CH3), 54.30 (NHCH), 33.75 (NCH2), 24.92 (CH2CH3), 14.46 (OCH2CH3), 10.54 (CH2CH3). IR (KBr cm-1): 3303 (N-H), 2972 (C-H), 2852 (C-H), 1745 (C=O), 1711 (C=O), 1640 (C=O),1597 (C=C), 1292 (C=S),1159 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C18H20N3O4FS: 394.1158; found: 394.1224.
Ethyl (2-((5-(4-fluorobenzylidene)-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) valinate (10m)
A reaction of 5-(4-fluorobenzylidene)-2-thiohydantoin 8d (0.50 g, 2.24 mmol) and ethyl (2-chloroacetyl)valinate 9d (0.50 g, 2.24 mmol) gave compound 10m as a white solid (0.47 g, 52.93%); m.p = 175‒177 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 11.89 (s, 1H, NH), 8.57 (d, J 8.3 Hz, 1H, NH), 8.38 (dd, JHH 7.2 Hz, 4JFH 6.8 Hz, 2H, 2 × ArH), 7.23 (dd, JHH 8.4 Hz, 3JFH 17.2 Hz, 2H, 2 × ArH), 6.78 (s, 1H, CCH), 4.23 (t, J 4.3 Hz, 1H, NHCH), 4.20 (s, 2H, NCH2), 4.09 (q, J 4.1 Hz, 2H, OCH2CH3), 2.03 (m, 1H, CH(CH3)2), 1.15 (t, J 7.0 Hz, 3H, OCH2CH3), 0.84 (d, J 6.7 Hz, 6H, CH(CH3)2). 13C NMR (100 MHz, DMSO-d6) δC (ppm) 171.63 (COO), 170.92 (C=S), 167.41 (CONH), 164.39 (1JFC 248.0 Hz, ArCF), 161.75 (CON), 139.14 (CCH), 134.38 (3JFC 8.0 Hz, 2 × ArCH), 131.32 (4JFC 3.0 Hz, ArC), 120.46 (CCH), 118.04 (2JFC 22.0 Hz, 2 × ArCH), 60.92 (OCH2CH3), 58.26 (NHCH), 33.68 (NCH2), 30.58 (CH(CH3)2), 19.25 (CH(CH3)2), 18.45 (CH(CH3)2), 14.47 (OCH2CH3). IR (KBr cm-1): 3259 (N-H), 2967 (C-H), 2885 (C-H), 1738 (C=O), 1715 (C=O), 1643 (C=O),1596 (C=C), 1307 (C=S), 1153 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C19H22N3O4FS: 408. 1303; found: 408.1384.
Ethyl (2-((5-(4-fluorobenzylidene)-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) norvalinate (10n).
A reaction of 5-(4-fluorobenzylidene)-2-thiohydantoin 8d (0.50 g, 2.24 mmol) and ethyl (2-chloroacetyl)norvalinate 9e (0.50 g, 2.24 mmol) gave compound 10n as a white solid (0.65 g, 72.51% ); m.p = 196‒198 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 11.73 (s, 1H, NH), 8.71 (d, J 7.5 Hz, 1H, NH), 8.28 (dd, JHH 8.0 Hz, 4JFH 6.4 Hz Hz, 2H, 2 × ArH), 7.25 (dd, JHH 8.8 Hz, 3JFH 17.6 Hz, 2H, 2 × ArH), 6.79 (s, 1H, CCH), 4.26 (q, J 7.9 Hz, 1H, NHCH), 4.14 (s, 2H, NCH2), 4.06 (q, J 6.5 Hz, 2H, OCH2CH3), 1.94–1.47 (m, 2H, CH2CH2CH3), 1.34–1.21 (m, 2H, CH2CH2CH3), 1.16 (t, J 7.2 Hz, 3H, OCH2CH3), 0.75 (t, J 7.3 Hz, 3H, CH2CH2CH3). 13C NMR (100 MHz, DMSO-d6) δC (ppm) 172.29 (COO), 170.93 (C=S), 167.18 (CONH), 164.35 (1JFC 247.0 Hz, ArCF), 161.76 (CON), 139.15 (CCH), 134.40 (3JFC 8.0 Hz, 2 × ArCH), 131.35 (4JFC 3.0 Hz, ArC), 120.40 (CCH), 116.06 (2JFC 21.0 Hz, 2 × ArCH), 60.95 (OCH2CH3), 52.72 (NHCH), 46.17 (CH2CH2CH3), 33.68 (NCH2), 18.95 (CH2CH2CH3), 14.43 (C-19), 13.75 (CH2CH2CH3). IR (KBr cm-1): 3286 (N-H), 2922 (C-H), 2852 (C-H), 1736 (C=O), 1711 (C=O), 1643 (C=O),1598 (C=C), 1269 (C=S), 1154 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C19H22N3O4FS: 408. 1303; found: 408.1384.
Ethyl (2-((5-(4-methoxybenzylidene)-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) glycinate (10o).
A reaction of 5-(4-methoxybenzylidene)-2-thiohydantoin 8c (0.50 g, 2.13 mmol) and ethyl (2-chloroacetyl)glycinate 9a (0.38 g, 2.13 mmol) gave compound 10o as a yellow solid (0.38 g, 47.51%); m.p = 201‒202 oC; 1H NMR (400 MHz, DMSO) δH 11.77 (s, 1H, NH), 8.79 (t, J 5.5 Hz, 1H, NH), 8.15 (d, J 8.4 Hz, 2H, 2 × ArH), 7.74 (d, J 8.5 Hz, 2H, 2 × ArH), 6.74 (s, 1H, CCH), 4.12 (s, 2H, NCH2), 4.10 (q, J 5.7 Hz, 2H, OCH2CH3), 3.91 (d, J 5.7 Hz, 2H, NHCH2), 3.81 (s, 3H, OCH3), 1.17 (t, J 7.1 Hz, 3H, OCH2CH3). 13C NMR (101 MHz, DMSO) δc (ppm) 171.02 (COO), 170.00 (C=S), 167.65 (CONH), 162.43 (ArCOCH3), 160.94 (CON), 137.74 (CCH), 133.98 (2 × ArCH), 127.29 (ArC), 122.07 (CCH), 114.83 (2 × ArCH), 60.98 (OCH2CH3), 55.80 (OCH3), 41.65 (NHCH2), 33.72 (NCH2), 14.47 (OCH2CH3). IR (KBr cm-1): 3308 (N-H), 2979 (C-H), 2833 (C-H), 1759 (C=O), 1708 (C=O), 1662 (C=O),1598 (C=C), 1251 (C=S), 1164 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C17H19N3O5S: 378.1045; found: 378.1114.
Ethyl (2-((5-(4-methoxybenzylidene)-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) alaninate (10p).
A reaction of 5-(4-methoxybenzylidene)-2-thiohydantoin 8c (0.50 g, 2.13 mmol) and ethyl (2-chloroacetyl)alaninate 9b (0.41 g, 2.13 mmol) gave compound 10p as a yellow solid (0.11 g, 73.53%); m.p 174‒176 oC; 1H NMR (400 MHz, DMSO) δH 11.77 (s, 1H, NH), 8.81 (d, J 6.8 Hz, 1H, NH), 8.17 (d, J 8.3 Hz, 2H, 2 × ArH), 6.99 (d, J 8.3 Hz, 2H, 2 × ArH), 6.74 (s, 1H, CCH), 4.28 (quintet, J 13.9, 6.9 Hz, 2H, CHCH3), 4.11 (s, 2H, NCH2), 4.09 (q, J 5.7 Hz, 2H, OCH2CH3), 3.82 (s, 3H, OCH3), 1.15 (t, J 7.0 Hz, 3H, OCH2CH3), 0.83 (d, J 4.0 Hz, 3H, CHCH3). 13C NMR (101 MHz, DMSO) δc (ppm) 172.73 (COO), 171.00 (C=S), 166.93 (CONH), 162.53 (ArCOCH3), 160.91 (CON), 137.74 (CCH), 134.03 (2 × ArCH), 127.40 (ArC), 122.00 (CCH), 114.66 (2 × ArCH), 60.99 (OCH2CH3), 55.70 (CHCH3), 48.64 (OCH3), 33.76 (NCH2), 17.50 (CHCH3), 14.43 (OCH2CH3). IR (KBr cm-1): 3285 (N-H), 2921 (C-H), 2851 (C-H), 1732 (C=O), 1708 (C=O), 1659 (C=O),1597 (C=C), 1254 (C=S), 1112 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C18H21N3O5S: 392.1209; found: 392.1267.
Ethyl 2-(2-(4-(4-methoxybenzylidene)-5-oxo-2-thioxoimidazolidin-1-yl) acetamido) butanoate (10q)
A reaction of 5-(4-methoxybenzylidene)-2-thiohydantoin 8c (0.50 g, 2.13 mmol) and ethyl 2-(2-chloroacetamido)butanoate 9c (0.44 g, 2.13 mmol) gave compound 10q as a yellow solid (0.58 g, 67.63%); m.p = 162-164 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 11.74 (s, 1H, NH), 8.69 (d, J 7.4 Hz, 1H, NH), 8.16 (d, J 8.3 Hz, 2H, 2 × ArH), 6.99 (d, J 8.3 Hz, 2H, 2 × ArH), 6.74 (s, 1H, CCH), 4.20 (q, J 5.8 Hz, 1H, NHCH), 4.12 (s, 2H, NCH2), 4.06 (q, J 6.8 Hz, 2H, OCH2CH3), 3.81 (s, 3H, OCH3), 1.80-1.68 (m, 2H, CHCH2CH3), 1.15 (t, J 7.1 Hz, 3H, OCH2CH3), 0.85 (t, J 7.3 Hz, 3H, CHCH2CH3). 13C NMR (101 MHz, DMSO) δC (ppm) 172.09 (COO), 171.00 (C=S), 167.29 (CONH), 162.58 (ArCOCH3), 160.92 (CON), 137.72 (CCH), 133.86 (ArCH), 127.37 (ArC), 121.93 (CCH), 114.65 (ArCH), 60.95 (OCH2CH3), 55.69 (NHCH), 54.31 (OCH3), 33.70 (NCH2), 24.91 (NHCH2CH3), 14.46 (OCH2CH3), 10.53 (NHCH2CH3). IR (KBr cm-1): 3306 (N-H), 2974 (C-H), 2901 (C-H), 1741 (C=O), 1707 (C=O), 1640 (C=O),1596 (C=C), 1251 (C=S),1139 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C19H23N3O5S: 406.1358; found: 406.1423.
Ethyl (2-((5-(4-methoxybenzylidene)-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) valinate (10r)
A reaction of (5-(4-methoxybenzylidene)-2-thiohydantoin 8c (0.50 g, 2.13 mmol) and ethyl (2-chloroacetyl)valinate 9d (0.47 g, 2.13 mmol) gave compound 10r as a solid (0.55 g, 61.59%); m.p = 184-186 oC; 1H NMR (400 MHz, DMSO) δH 12.10 (s, 1H, NH), 8.56 (d, J 8.3 Hz, 1H, NH), 8.15 (d, J 8.6 Hz, 2H, ArH), 7.73 (d, J 8.6 Hz, 2H, ArH), 6.74 (s, 1H, CCH), 4.21 (dd, J 8.5, 6.8 Hz, 1H, CHCH(CH3)2), 4.15 (s, 2H, NCH2), 4.08 (q, J 7.2 Hz, 2H, OCH2CH3), 3.81 (s, 3H, OCH3), 2.06 (m, 1H, CHCH(CH3)2), 1.15 (t, J 7.0 Hz, 3H, OCH2CH3), 0.85 (d, J 6.8 Hz, 6H, CHCH(CH3)2). 13C NMR (101 MHz, DMSO) δC (ppm) 171.61 (COO), 170.99 (C=S), 167.52 (CONH), 162.63 (ArC), 160.70 (CON), 137.69 (CCH), 132.58 (ArCH), 127.35 (ArC), 122.11 (CCH), 114.59 (ArCH), 60.92 (OCH2CH3), 58.27 (CHCH(CH3)2), 55.79 (OCH3), 33.64 (NCH2), 30.57 (CHCH(CH3)2), 19.28 (CHCH(CH3)2), 14.50 (OCH2CH3). IR (KBr cm-1): 3198 (N-H), 2969 (C-H), 2891 (C-H), 1706 (C=O), 1630 (C=O), 1596 (C=O),1510 (C=C), 1226 (C=S),1167 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C20H25N3O5S: 420.1514; found: 420.1582.
Ethyl (2-((5-(4-methoxybenzylidene)-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) norvalinate (10s).
A reaction of 5-(4-methoxybenzylidene)-2-thiohydantoin 8c (0.50 g, 2.13 mmol) and ethyl (2-chloroacetyl)norvalinate 9e (0.47 g, 2.13 mmol) gave compound 10s as a yellow solid (0.43 g, 48.05%); m.p = 214-216 oC; 1H NMR (400 MHz, DMSO-d6) δH (ppm) 12.01 (s, 1H, NH), 8.68 (d, J 7.5 Hz, 1H, NH), 8.16 (d, J 8.3 Hz, 2H, ArH), 7.74 (d, J 8.3 Hz, 2H, ArH), 6.74 (s, 1H, CCH), 4.25 (q, J 7.6 Hz, 1H, CHCH2CH2CH3), 4.14 (s, 2H, NCH2), 4.07 (q, J 5.2 Hz, 2H, OCH2CH3), 3.81 (s, 3H, OCH3), 1.74–1.46 (m, 2H, CHCH2CH2CH3), 1.36–1.19 (m, 2H, CHCH2CH2CH3), 1.15 (t, J 7.1 Hz, 3H, OCH2CH3), 0.76 (t, J 7.2 Hz, 3H, CHCH2CH2CH3). 13C NMR (101 MHz, DMSO) δC (ppm) 172.30 (COO), 171.00 (C=S), 167.28 (CONH), 162.55 (ArC), 160.92 (CON), 137.69 (CCH), 134.01 (ArCH), 127.37 (C-6), 122.07 (C-5), 114.66 (ArCH), 60.96 (OCH2CH3), 55.83 (CHCH2CH2CH3), 52.73 (OCH3), 46.31 (CHCH2CH2CH3), 33.63 (C-11), 18.96 (CHCH2CH2CH3), 14.45 (OCH2CH3), 13.81 (CHCH2CH2CH3). IR (KBr cm-1): 3201 (N-H), 2962 (C-H), 2855 (C-H), 1707 (C=O), 1643 (C=O), 1597 (C=O),1505 (C=C), 1255 (C=S), 1167 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C20H25N3O5S: 420.1514; found: 420.1582.
Ethyl (2-((5-(4-nitrobenzylidene)-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) alaninate (10t)
A reaction of 5-(4-nitrobenzylidene)-2-thiohydantoin 8e (0.50 g, 2.06 mmol) and ethyl (2-chloroacetyl)alaninate 9b (0.39 g, 2.06 mmol) gave compound 10t as a yellow solid (0.54 g, 66.82%); m.p = 232-234 oC; 1H NMR (400 MHz, DMSO) δH 11.81 (s, 1H, NH), 8.86 (d, J 6.6 Hz, 1H, NH), 8.45 (d, J 8.1 Hz, 2H, ArH), 8.24 (d, J 8.5 Hz, 2H, ArH), 6.85 (s, 1H, CCH), 4.35–4.24 (m, 1H, CHCH3), 4.15 (s, 2H, NCH2), 4.06 (q, J 6.6 Hz, 2H, OCH2CH3), 1.28 (d, J 7.1 Hz, 3H, CHCH3), 1.19 (t, J 8.0 Hz, 3H, OCH2CH3). 13C NMR (101 MHz, DMSO) δc (ppm) 172.42 (COO), 170.29 (C=S), 167.19 (CONH), 166.56 (CON), 146.87 (ArC), 142.11 (CCH), 141.24 (ArC), 132.41 (ArCH), 122.99 (CCH), 117.47 (ArCH), 60.16 (OCH2CH3), 48.70 (CHCH3), 33.84 (NCH2), 17.55 (OCH2CH3), 14.40 (CHCH3). IR (KBr cm-1): 3305 (N-H), 2920 (C-H), 2849 (C-H), 1750 (C=O), 1710 (C=O), 1650 (C=O), 1538 (C=C), 1305 (C=S),1150 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C17H18N4O6S: 407. 1409; found: 407. 1475.
Ethyl 2-(2-(4-(4-nitrobenzylidene)-5-oxo-2-thioxoimidazolidin-1-yl) acetamido) butanoate (10u).
A reaction of 5-(4-nitrobenzylidene)-2-thiohydantoin 8e (0.50 g, 2.06 mmol) and ethyl 2-(2-chloroacetamido)butanoate 9c (0.42 g, 2.06 mmol) gave compound 10u as a yellow solid (0.21 g, 75.14%); m.p = 237-239 oC; 1H NMR (400 MHz, DMSO) δH 1H NMR (400 MHz, DMSO) δ 11.99 (s, 1H, H-1NH), 8.74 (d, J 7.4 Hz, 1H, NH), 8.45 (d, J 8.3 Hz, 2H, ArH), 8.24 (d, J 8.4 Hz, 2H, ArH), 6.85 (s, 1H, CCH), 4.26 (q, J 8.0 Hz, 2H, OCH2CH3), 4.15 (q, J 15.5 Hz, 1H, CHCH2CH3), 4.12 (s, 2H, NCH2), 1.86–1.38 (m, 2H, CHCH2CH3), 1.14 (t, J 7.1 Hz, 3H, OCH2CH3), 0.83 (t, J 7.3 Hz, 3H, CHCH2CH3). 13C NMR (101 MHz, DMSO) δc (ppm) 13C NMR (101 MHz, DMSO) δ 172.12 (COO), 170.72 (C=S), 167.70 (CONH), 167.03 (CON), 147.18 (ArC), 142.10 (CCH), 141.37 (ArC), 132.71 (ArCH), 123.98 (CCH), 117.99 (ArCH), 60.97 (OCH2CH3), 54.32 (CHCH2CH3), 33.97 (NCH2), 24.97 (CHCH2CH3), 14.46 (OCH2CH3), 10.54 (CHCH2CH3). IR (KBr cm-1): 3304 (N-H), 2980 (C-H), 2858 (C-H), 1739 (C=O), 1715 (C=O), 1651 (C=O),1505 (C=C), 1338 (C=S), 1181 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C18H20N4O6S: 421.1103; found: 421.1170.
Ethyl (2-((5-(4-nitrobenzylidene)-4-oxo-4, 5-dihydro-1H-imidazol-2-yl) thio) acetyl) norvalinate (10v)
A reaction of 5-(4-nitrobenzylidene)-2-thiohydantoin 8e (0.50 g, 2.06 mmol) and ethyl (2-chloroacetyl)norvalinate 9e (0.44 g, 2.06 mmol) gave compound 10v as a yellow solid (0.53 g, 61.50%); m.p = 211–213 oC; 1H NMR (400 MHz, DMSO) δH 11.87 (s, 1H, NH), 8.73 (d, J 7.5 Hz, 1H, NH), 8.43 (d, J 8.6 Hz, 2H, ArH), 8.22 (d, J 8.7 Hz, 2H, ArH), 6.82 (s, 1H, CCH), 4.33 (q, J 8.1 Hz, 1H, CHCH2CH2CH3), 4.14 (s, 2H, NCH2), 4.06 (q, J 8.8 Hz, 2H, OCH2CH3), 1.79–1.46 (m, 2H, CHCH2CH2CH3), 1.34–1.19 (m, 2H, CHCH2CH2CH3), 1.13 (t, J 7.1 Hz, 3H, OCH2CH3), 0.72 (t, J 7.3 Hz, 3H, CHCH2CH2CH3). 13C NMR (101 MHz, DMSO) δc (ppm) 172.32 (COO), 170.72 (C=S), 167.69 (CONH), 167.04 (CON), 147.13 (ArC), 142.07 (CCH), 141.37 (ArC), 132.71 (ArCH), 123.95 (CCH), 117.95 (ArCH), 60.97 (OCH2CH3), 52.80 (CHCH2CH2CH3), 46.20 (NCH2), 33.82 (CHCH2CH2CH3), 18.98 (CHCH2CH2CH3), 14.42 (OCH2CH3), 13.74 (CHCH2CH2CH3). IR (KBr cm-1): 3109 (N-H), 2959 (C-H), 2916 (C-H), 1743 (C=O), 1714 (C=O), 1649 (C=O),1594 (C=C), 1337 (C=S), 1146 (C-O). HRMS (ESI-TOF): m/z [M+H]+ calculated for C19H22N4O6S: 435.1260; found: 435.1324.
Conclusion
Twenty-two new 2-thiohydantoin containing compounds 10 were synthesized in this project over four reaction steps. The synthesis started from the Knoevenagel condensation of 2-thiohydantoin with five different benzaldehydes to obtain intermediates 8. On the other hand chloroacetyl compounds 9 were synthesized by first protecting the amino acids with thionyl chloride in the presence of ethanol and subsequently reacting the intermediates with chloroacetyl chloride in the presence of sodium bicarbonate in good yields. Intermediates 8 and 9 were reacted in the presence of triethylamine in refluxing ethanol over 12 hours to obtain compounds 10 in acceptable to good yields varying from 47.51% to 80.81%.
- Buchynskyy A, Gillespie JR, Herbst ZM, Ranade RM, Buckner FS, et al. (2017) Medicinal Chemistry Letters. 8: 886-91.
- Raj R, Gut J, Rosenthal PJ, Kumar V (2014) Bioorganic & Medicinal Chemistry Letters. 24: 756-9.
- Kieć-Kononowicz K, Szymańska E (2002) Il Farmaco. 57: 909-16.
- Han J, Dong H, Xu Z, Lei J, Wang M, et al. (2013) International Journal of Molecular Sciences. 14: 12484-95.
- Tshiluka NR, Bvumbi MV, Ramaite IDI, Mnyakeni Moleele SS (2020) Arkivoc, part v, 161-75.
- Tshiluka NR, Bvumbi MV, Mnyakeni Moleele SS (2023) Russian Journal of Bioorganic Chemistry, 49: 384-9.
- Tshiluka NR, Bvumbi MV, Tshishonga U, Mnyakeni Moleele SS (2022) Journal of Chemical Research, 46:1-7
- Burgy G, Tahtouh T, Durieu E, Foll-Josselin B, Limanton E, et al. (2013) European Journal of Medicinal Chemistry. 62: 728-37.
- Zaitseva ER, Smirnov AY, Mishin AS, Baranov MS (2020) Russian Journal of Bioorganic Chemistry, 46: 458-61.
- Brun E, Safer A, Carreaux F, Bourahla K, L’helgoua’ch JM, et al. (2015) Molecules, 23: 11617-31.
- Kaczor A, Szemerédi N, Kucwaj‐Brysz K, Dąbrowska M, Starek M, et al. (2021) ChemMedChem. 16: 2386-401.
- Singh V, Singh A, Singh G, Verma RK, Mall R, et al. (2021) Medicinal Chemistry Research, 30: 1905-14.
- Tropiano M, Blackburn OA, Tilney JA, Hill LR, Placidi MP, et al. (2013) Chemistry–A European Journal, 19: 16566-71.
- Tshishonga U, MSc dissertation (2024) University of Venda.
- Christoff RM, Soares da Costa TP, Bayat S, Holien JK, Perugini MA, et al. (2021) Bioorg. Med. Chem. 52: 116518

Tables at a glance
Figures at a glance