Accelerating Corneal Epithelial Healing with Topical Insulin: Results from a Prospective Comapative 20-Eye Series
Received Date: November 01, 2025 Accepted Date: November 17, 2025 Published Date: November 20, 2025
doi:10.17303/jooa.2025.9.201
Citation: Rhizlane abdi (2025) Accelerating Corneal Epithelial Healing with Topical Insulin: Results from a Prospective Comapative 20-Eye Series. J Ophthalmol Open Access 9: 1-10
Abstract
Purpose: To evaluate the clinical efficacy and safety of topical insulin (1 IU/mL) in accelerating corneal epithelial healing in persistent epithelial defects (PEDs) refractory to conventional therapy, and to compare outcomes with a matched retrospective control group.
Methods: This prospective interventional study included 20 eyes with PEDs unresponsive to ≥2 weeks of standard treatment. Patients received topical insulin 1 IU/mL four times daily until complete re-epithelialization. A retrospective cohort of 20 matched eyes served as controls. Matching was based on age, PED etiology, initial defect size, and diabetes status. Epithelial healing was assessed by fluorescein staining and anterior segment OCT. Primary outcome was time to complete re-epithelialization; secondary outcomes included change in epithelial defect area, BCVA, OCT epithelial thickness, and recurrence rate.
Results: Complete epithelial closure occurred in 18/20 insulin-treated eyes (90%), with a mean healing time of 9.6 ± 3.1 days. Mean epithelial defect area reduced from 7.8 ± 3.4 mm² to 0 mm² (p < 0.001). BCVA improved from 0.54 ± 0.18 to 0.32 ± 0.15 logMAR (p < 0.01). OCT confirmed restoration of epithelial thickness (from 21.8 ± 4.6 to 48.3 ± 6.1 µm). Compared with controls, insulin-treated eyes healed significantly faster (17.3 ± 4.5 vs 9.6 ± 3.1 days, p < 0.001) and showed higher complete healing rates (55% vs 90%, p = 0.02). No adverse events were noted.
Conclusion: Topical insulin eye drops (1 IU/mL) significantly accelerate corneal epithelial healing and improve visual and structural outcomes in refractory PEDs. Given their low cost, safety, and accessibility, insulin drops are a promising therapeutic adjunct, especially in resource-limited settings. Randomized controlled trials are warranted.
Keywords: Cornea; Insuline- Resistant; Defect
Introduction
The corneal epithelium is essential for preserving the integrity of the ocular surface and the quality of vision.it is rich in a number of growth factors that are essential for the healing process. Any disruption of this process can transform an acute defect into a persistent epithelial defect (PED), which is defined as any superficial corneal defect that does not heal after 10–14 days, even with conventional supportive care [1,2].
PEDs continue to be a significant treatment challenge for ophthalmologists [5] due to their varied etiological nature (neurotrophic cornea, surface trauma, defective epithelial adhesion, limbal stem cell deficiency, inflammation, and hereditary) and actual resistance to common treatment options (lubrication, bandage contact lenses, punctual plugs, debridement, and tarsorrhaphy) [1, 3]. In a number of clinical trials conducted in recent years, new non-invasive therapies such as topical fibronectin, autologous serum eye drops, topical epidermal and insulin-like growth factors, and other hemoderivative medicines have shown encouraging safety and effectiveness in treating PEDs [2, 4].
Therefore, topical insulin a peptide hormone structurally related to insulin-like growth factors (IGFs) has emerged as a promising and effective therapeutic option for promoting corneal epithelial wound healing in cases refractory to conventional therapy. Despite the documented presence of insulin and its receptors in the human tear film and corneal epithelium, as well as its advantages, such as ease of administration, excellent tolerability, and minimal side effects [5], reported in animal studies, prospective clinical trials investigating the precise mechanisms by which topical insulin promotes the healing of persistent corneal epithelial defects (PEDs) in humans are still lacking.
In light of these factors, investigating topical insulin as a treatment for refractory corneal epithelial abnormalities seems both clinically and physiologically reasonable. Its capacity to improve cellular metabolism, lower inflammation, and speed up epithelial regeneration presents a viable substitute for current therapies.
The current study intends to evaluate topical insulin's clinical efficacy and safety in the treatment of persistent epithelial defects, to determine how it affects the rate and quality of corneal healing, and to shed more light on potential mechanisms of action in human corneal epithelium.
Materials and Methods
This was a prospective, interventional study conducted in the Department of Ophthalmology, University Moulay Slimane, Beni Mellal, Morocco. The study was approved by the institutional ethics committee and adhered to the tenets of the Declaration of Helsinki. Written informed consent was obtained from all participants before inclusion.
Patients with persistent epithelial defects (PEDs) of the cornea that had not healed after at least 2 weeks of conventional treatment were enrolled. The underlying etiologies included neurotrophic keratopathy, post-surgical epithelial defects, and metabolic or inflammatory causes.
Exclusion criteria included active infectious keratitis, recent chemical or thermal injury, ocular trauma, uncontrolled systemic disease, or hypersensitivity to any component of the formulation.
A retrospective control cohort of 20 eyes with refractory PEDs treated with conventional therapy was identified from institutional records.
- The retrospective control group was matched 1:1 with the insulin-treated group according to:
- Initial Epithelial Defect Size (±1 Mm²),
- Ped Etiology (Neurotrophic, Post-Surgical, Metabolic/Inflammatory, or Idiopathic),
- Age Group (±5 Years),
- Diabetes Status (Presence or Absence).
This ensured comparable baseline characteristics between groups.
Before inclusion, all patients had received intensive medical therapy including preservative-free lubricants, bandage soft contact lenses, and management of the underlying systemic or ocular disorder. Patients with incomplete records or follow-up shorter than 2 weeks were excluded.
Topical insulin eye drops were prepared in a sterile environment using regular human insulin (Actrapid HM 1000 IU/mL, Novo Nordisk). The solution was diluted with sterile balanced salt solution (BSS) to obtain a final concentration of 1 IU/mL. The preparation was stored at 4 °C and renewed every 7 days to maintain sterility and efficacy.
Patients were instructed to instill one drop of topical insulin four times daily in the affected eye until complete re-epithelialization was achieved. Concomitant treatment with artificial tears was allowed, but no additional growth factors or serum tears were administered during the study period.
The epithelial defect was evaluated daily using fluorescein staining and slit-lamp biomicroscopy.
Anterior segment optical coherence tomography was performed at baseline and at each follow-up to objectively assess epithelial defect depth, stromal thickness, and the integrity of the epithelial surface during healing.
Patients were followed daily until complete re-epithelialization, then weekly for one month to monitor recurrence or complications.
Patients were followed daily until complete re-epithelialization, then weekly for one month to monitor recurrence or complications.
Data were analyzed using SPSS. Descriptive statistics were expressed as mean ± standard deviation (SD) for continuous variables and as frequencies for categorical variables.
Results
Patient Demographics and Etiology
A total of 20 eyes from 20 patients were included, comprising 11 males and 9 females, with a mean age of 58.4 ± 12.7 years (range: 34–76).
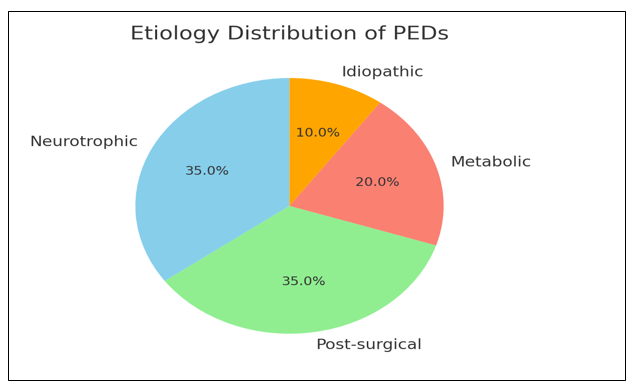
The underlying causes of persistent epithelial defects were diverse:
- Neurotrophic keratopathy: 8 eyes (40%) — associated with impaired corneal innervation due to herpetic keratitis, post-surgical nerve injury, or diabetes mellitus.
- Post-surgical epithelial defects: 6 eyes (30%) — occurring after cataract surgery, vitrectomy, or corneal transplantation.
- Metabolic or inflammatory causes: 4 eyes (20%) — related to systemic conditions such as uncontrolled diabetes or ocular surface inflammation.
- Idiopathic: 2 eyes (10%) — defects with no identifiable systemic or ocular cause after thorough evaluation.
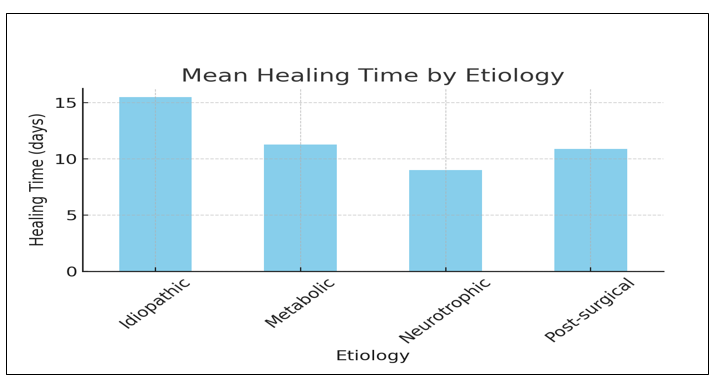
The mean duration of the epithelial defect prior to inclusion was 23.5 ± 8.2 days (range: 14–42 days), reflecting the refractory nature of these lesions despite prior intensive conventional therapy.
Epithelial Healing
Following treatment with topical insulin 1 U/mL, 18 of 20 eyes (90%) achieved complete epithelial closure, confirmed by slit-lamp examination and fluorescein staining.
The mean time to full epithelial closure was 9.6 ± 3.1 days (range: 6–16 days).
The remaining 2 eyes (10%) demonstrated partial healing (>50% reduction in defect size) but required extended therapy and adjunctive measures to achieve complete closure.
Quantitative analysis of epithelial defect area demonstrated a significant reduction from baseline (mean 7.8 ± 3.4 mm²) to final follow-up (0 mm² in healed eyes; p < 0.001).
No recurrences were observed during the 1-month post-healing follow-up period.
Compared with the control cohort, insulin-treated eyes achieved complete epithelial closure more frequently (90% vs 55%, p = 0.02) and faster (mean 9.6 ± 3.1 vs 17.3 ± 4.5 days, p < 0.001).
The mean reduction in epithelial defect area was also greater in the insulin group (100% vs 62%, p < 0.001).
No recurrences were observed in the insulin group during the 1-month post-healing follow-up, while 3 eyes (15%) in the control group experienced partial recurrence of the epithelial defect.
Baseline characteristics including age, sex distribution, PED etiology, and defect size were comparable between the insulin-treated group and the matched control cohort (p > 0.05 for all).
Visual Outcomes
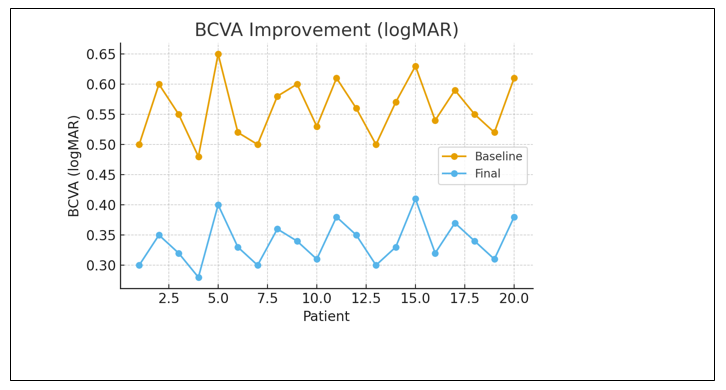
Best-corrected visual acuity (BCVA) improved in 14 eyes (70%) after epithelial healing.
Mean BCVA improved from 0.54 ± 0.18 logMAR at baseline to 0.32 ± 0.15 logMAR post-healing (p < 0.01).
Visual improvement correlated with restoration of corneal transparency and surface smoothness.
Compared with controls, BCVA gain was more pronounced in the insulin-treated group (ΔlogMAR 0.22 ± 0.07 vs 0.09 ± 0.05; p = 0.03).
Anterior Segment OCT Findings
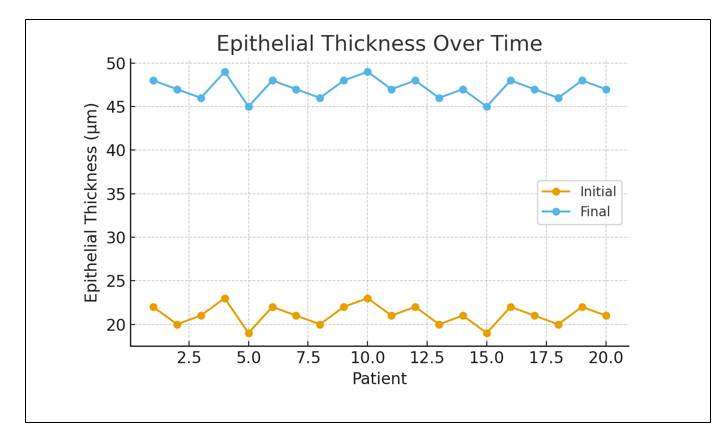
Baseline AS-OCT revealed variable epithelial thinning and irregular stromal reflectivity adjacent to the defect.
Progressive epithelial regrowth was observed in all cases, with formation of a continuous epithelial layer typically beginning at day 3–5. Complete restoration of epithelial thickness and surface smoothness was observed at day 10 ± 2 on average.
Quantitative AS-OCT analysis showed epithelial thickness increased from 21.8 ± 4.6 µm at baseline to 48.3 ± 6.1 µm after complete healing (p < 0.001).
Stromal edema, initially present in 7 eyes (35%), resolved gradually as epithelial continuity was restored.
Sequential AS-OCT scans documented reduction in surface irregularity, normalization of reflectivity, and restoration of the epithelial-stromal interface.
No contamination or turbidity of the insulin solution was observed during the 7-day storage period, and patient compliance was excellent.
Discussion
Persistent, epithelial defect happens when there is a failure of the mechanisms promoting corneal epithelialization within the normal two-week time frame [5].
Refractory or persistent epithelial defects (PEDs) can develop as a result of several local and systemic conditions that impair the proliferation or adhesion of the corneal epithelium. The most common causes are neurotrophic keratopathy, often associated with herpes infections, diabetes mellitus or trigeminal nerve damage; post-surgical epithelial defects following keratoplasty, refractive surgery or retinal surgery; and metabolic or inflammatory disorders, such as diabetes, ocular surface diseases and exposure keratopathy [5, 7]. Systemic factors such as poor glycaemic control, autoimmune disease and malnutrition can further compromise corneal healing [8].
Refractory epithelial defects are typically managed gradually, with an initial focus on stimulation epithelial regeneration, and managing etiologies. First-line treatments include the use of preservative-free lubricants, contact lenses, autologous serum, correction of eyelid irregularities or tear film dysfunction [2, 4, 8].
Surgical interventions like conjunctival flap grafting, tarsorrhaphy, or amniotic membrane grafting may be required to regain ocular surface stability if the condition does not respond to therapy. Since these treatments are invasive, expensive, and not always accessible, it is still very important to find simpler and more accessible alternatives [4, 9].
Recent developments have focused on the use of biological drugs and biological strategies, such as the topical nerve growth factor (cénegermine), the epidermal growth factor (EGF), and cell-based therapies. Even while these agents have shown promising results, their high cost and limited accessibility have greatly limited their use, especially in countries with low and intermittent income [9, 10].
In this context, topical insulin has emerged as a promising, inexpensive, and readily available option for treating refractory epithelial defects. Beyond its metabolic role, insulin acts as a potent growth factor that stimulates the proliferation, migration, and differentiation of epithelial cells by activating insulin and IGF-1 receptors present on corneal epithelial cells [5, 10]. It also modulates local inflammation and improves cellular metabolism, thereby accelerating epithelial healing [12].
Recent advances have focused on the use of biological agents and regenerative strategies, including topical nerve growth factor (cenegermin), epidermal growth factor (EGF), and stem cell therapies. Although these agents have shown promising results, their high cost and limited accessibility have restricted their widespread use, particularly in low- and middle-income countries.
Several experimental and clinical studies have examined the efficacy of topical insulin for corneal surface disorders.
Zagon et al. (2007) were the first to demonstrate, in animal models, that topical insulin normalised corneal epithelial healing in diabetic rats [5].
Faiq et al. (2019) and Al-Kharashi et al. (2020) reported the successful use of diluted insulin eye drops in the treatment of diabetic keratopathy and neurotrophic ulcers, with a significant improvement in healing time and no adverse effects [13, 14].
In a prospective clinical study conducted at Farabi Eye Hospital (Dasrilsyah et al., 2023), 23 patients with PED refractory to conventional treatment were treated with 1 IU/mL insulin drops four times daily. Complete re-epithelialisation was achieved in the majority of eyes within an average of 11 days, with no systemic or local complications [15].
Jaworski et al. (2023) conducted an in-depth study and concluded that topical insulin is a safe and effective adjunctive treatment for neurotrophic and diabetic keratopathy, although larger randomised controlled trials are needed to define the optimal dosage and duration [16].
Almeida et al. (2024) reported favourable long-term outcomes in refractory PED cases, confirming sustained epithelial integrity and no recurrence after discontinuation of treatment [17].
In all of these studies, the most commonly used formulation was regular human insulin (1 IU/mL) diluted in balanced saline solution and administered four times daily. The treatment was well tolerated and no significant adverse events were observed [5, 14, 17].
The results of the present study are consistent with previously published results, confirming the beneficial effect of topical insulin on corneal epithelial healing. Its low cost, ease of preparation and favourable safety profile make it an attractive alternative or adjunct to conventional therapy, particularly in resource-limited settings. However, despite these encouraging results, standardised preparation protocols and long-term safety data are still lacking [16, 17].
The present comparative study further supports these findings by directly comparing the healing outcomes of topical insulin treatment versus conventional therapy in refractory PEDs.
Insulin-treated eyes demonstrated faster epithelial closure and earlier symptomatic relief compared with matched controls receiving standard care.
This study has several limitations. First, the sample size was relatively small (20 eyes), limiting the generalizability of the findings. Second, the study was not randomized, and the control group was retrospective, which may introduce selection or information bias despite careful matching. Third, follow-up was limited to one month after epithelial closure, preventing long-term safety assessment. Finally, although insulin preparation followed a standardized protocol, variability in drop handling by patients may influence outcomes.
Future research should include large randomized controlled trials with longer follow-up periods to more precisely determine optimal dosing, durability of epithelial integrity, and long-term safety.
Conclusion
Our prospective case series adds further clinical evidence supporting the beneficial role of topical insulin in promoting corneal epithelial healing in refractory cases. Topical insulin appears to be a safe, simple, and cost-effective therapeutic adjunct for patients who fail to respond to conventional treatments.
- Dua HS, King AJ, Joseph A (2004) Persistent epithelial defects: epidemiology, etiology, and management strategies. Surv Ophthalmol. 49: 3–17.
- Choi J, et al. (2017) Novel non-invasive therapies for persistent corneal epithelial defects. Ophthalmology. 124: 1444–52.
- Sridhar MS (2018) Anatomy of corneal healing and complications of persistent epithelial defects. Indian J Ophthalmol. 66: 1232-40.
- Tsubota K, Goto E, Shimmura S, Shimazaki J (1999) Treatment of persistent corneal epithelial defect by autologous serum application. Ophthalmology. 106: 1984–89.
- Zagon IS, Klocek MS, Sassani JW, McLaughlin PJ (2007) Use of topical insulin to normalize corneal epithelial healing in diabetic rats. Arch Ophthalmol. 125: 1082–88.
- Wilson SE, Mohan RR, Ambrosio R, Hong J, Lee J (2001) The corneal wound healing response: cytokine-mediated interaction of the epithelium, stroma, and inflammatory cells. Prog Retin Eye Res. 20: 625–37.
- Bonini S, Lambiase A, Rama P (2003) Neurotrophic keratitis. Eye (Lond). 17: 989–95.
- Dua HS, Gomes JAP, King AJ, Maharajan VS (2004) The amniotic membrane in ophthalmology. Surv Ophthalmol. 49: 51–77.
- Tseng SCG, Espana EM, Kawakita T, et al. (2004) How does amniotic membrane work? Ocul Surf. 2: 177–87.
- Sacchetti M, Lambiase A (2019) Cenegermin for the treatment of neurotrophic keratitis. Drugs Today (Barc). 55: 377–84.
- Bonini S, Lambiase A, Rama P, et al. (2018) Phase II randomized trial of recombinant human nerve growth factor for neurotrophic keratitis. Ophthalmology. 125: 1332–43.
- Abdelkader H, Patel DV (2021) Topical insulin and the cornea: recent developments and translational perspectives. Clin Exp Ophthalmol. 49: 793–804.
- Faiq MA, Dada T, Saluja D (2019) Insulin eye drops for the management of diabetic keratopathy: a pilot clinical study. Br J Ophthalmol. 103: 1723–27.
- Al-Kharashi SA, Al-Muammar AM, Al-Haddab AA (2020) Topical insulin in the management of neurotrophic corneal ulcers: a case series. Clin Ophthalmol. 14: 357–62.
- Dasrilsyah F, Faramarzi A, Ghaffari R, et al. (2023) Topical insulin for refractory persistent epithelial defects: a prospective interventional study. Cornea. 42: 512–18.
- Jaworski A, Rękas M, Wiśniewska-Kruk J (2023) Clinical outcomes of topical insulin therapy in neurotrophic and diabetic keratopathy: a prospective study. Int Ophthalmol. 43: 1745–53.
- Almeida JP, Silva MF, Lemos R (2024) Long-term outcomes of topical insulin for persistent epithelial defects of the cornea. Eur J Ophthalmol. 34: 91–9.

Tables at a glance
Figures at a glance