Cost-Effectiveness and Cost-utility Evaluation of Clopidogrel Compared to Prasugrel in Secondary Prevention of Myocardial Infarction
Received Date: February 21, 2025 Accepted Date: March 21, 2025 Published Date: March 24, 2025
doi:10.17303/jpdm.2025.8.102
Citation: Amirsadri M, Hajhashemi V, Azadi M (2025) Cost-Effectiveness and Cost-Utility Evaluation of Clopidogrel Compared to Prasugrel in Secondary Prevention of Myocardial Infarction. J Pharmacol Drug Metab 8: 1-15
Abstract
Background and Purpose: Cardiovascular diseases are the most common cause of death in most countries of the world, including Iran, and the most important cause of disability. Despite rapid advances in diagnosis and treatment, one-third of patients who have a heart attack still die, and two-thirds of those who survive never fully recover and return to normal life. These diseases impose a huge cost on the health care systems of countries. However, cardiovascular diseases are one of the most preventable non-communicable diseases of humans. Coronary artery diseases cause mortality, morbidity, and disability in the Iranian population and are responsible for approximately 50% of all deaths per year. While the age-specific mortality rate from coronary heart disease is decreasing in developed countries, there is evidence that this disease is increasing in Iran.
Clopidogrel and prasugrel are two of the commonly prescribed antiplatelet drugs for secondary prevention of myocardial infarction (MI). Prasugrel, a more potent P2Y12 inhibitor, offers potential advantages compared to clopidogrel, but its cost-effectivenss for this indication has not been evaluated in Iran.
Objective: To evaluate the cost-effectiveness of prasugrel compared to clopidogrel in a 55-year-old Iranian population with a history of MI.
Methods: A Markov-model was developed to simulate the long-term health outcomes and costs associated with each treatment. The analysis was conducted from a payer perspective, considering a lifetime horizon. The outcomes were evaluated in terms of Quality-Adjusted-Life-Years (QALY) and Life-Years-Gained (LYG).
Results: Prasugrel was found to be the cost-effective alternative compared to clopidogrel, with a cost saving of 1,999,522 Rials and more effectiveness in terms of QALY (0.067) and LYG (0.073) per patient.
Conclusion: The findings suggest that prasugrel is a cost-effective treatment option for secondary prevention of MI in Iran.
Keywords: Cost-Effectiveness; Myocardial-Infarction; Prasugrel; Clopidogrel; Markov-Model
Introduction
Myocardial infarction (MI) is characterized by the permanent and irreversible destruction and subsequent cell death of a portion of the cardiac muscle (myocardium). This necrosis is a consequence of diminished blood flow and severe ischemia in the affected myocardial tissue. The cessation of blood flow may manifest acutely, without preceding symptoms, or may occur following multiple episodes of angina pectoris. The primary etiology of MI is the obstruction of arteries supplying the heart [1].
CVD is a prominent cause of disability and mortality. Despite considerable advancements in diagnostic and therapeutic modalities, approximately one-third of patients experiencing a myocardial infarction do not survive, and two-thirds of those who do survive fail to achieve a complete recovery to pre-event functional status [2].
Coronary artery disease is a leading cause of mortality and morbidity within the Iranian population, accounting for approximately 50% of annual deaths [3].
Reviews of post-MI prescription patterns indicate that clopidogrel is the most frequently selected antiplatelet agent, with gastrointestinal bleeding identified as a significant adverse effect associated with its use [4].
A study conducted in 2021, encompassing seven studies with 32,951 participants, reported that reduced-dose prasugrel was associated with a reduced risk of major adverse cardiovascular events (MACE), including cardiovascular death, myocardial infarction (MI), or ischemic stroke (OR 0.08, 95% CI 0.67-0.97), compared to clopidogrel. However, with the exception of MI (OR 0.74, 95% CI (0.98-0.56), there was no statistically significant difference between reduced-dose prasugrel and clopidogrel in terms of primary safety endpoint of all bleeding events (OR: 1.31, 95% CI (1.98-0.78)), but reduced-dose prasugrel carried a significantly higher risk of minor bleeding (OR: 1.73, 95% CI (2.41-1.25)) [5].
Although clopidogrel is used for the prevention of blood clots, it is often prescribed prophylactically in patients with atherosclerosis who are at risk of MI and peripheral vascular disease. Clopidogrel irreversibly blocks the platelet ADP receptor. The initial dose is typically 300 mg, followed by a maintenance dose of 75 mg once daily [6].
Prasugrel is metabolized by the hepatic CYP system, specifically CYP2B6 and CYP2C9/19 (minor), leading to the formation of an active thiol metabolite and an inactive metabolite [7].
The recommended initial loading dose of prasugrel is 60 mg, administered upon the recognition of an acute coronary syndrome. For patients weighing over 60 kg, the standard maintenance dose is 10 mg/day. However, the risk of bleeding is elevated in patients with low body weight (<60 kg) taking prasugrel, making a lower dose of 5 mg/day a consideration in these patients. Studies have indicated that prasugrel at a 5 mg dose in patients with low body weight achieves a similar reduction in platelet reactivity as a 10 mg dose in patients with higher body weight (>60 kg). For research purposes, the default dose for this study was established at 10 mg prasugrel [8].
Cardiovascular diseases (CVDs) are the leading cause of death in many countries, including Iran, and are a significant contributor to disability. These diseases impose substantial costs on healthcare systems worldwide. However, CVDs are among the most preventable non-communicable diseases.
Given the critical role of anticoagulant drugs in secondary prevention of heart attacks, cost-effectiveness studies are essential for evidence-based drug decisions and policies. This study aims to evaluate the cost-effectiveness of clopidogrel compared to prasugrel to provide scientific evidence for optimal decision-making.
Materials and Methods
Pharmacoeconomics is a branch of economics that employs analyses such as cost-effectiveness and cost-utility to compare drug products and treatment strategies. Economic evaluations provide valuable information to healthcare decision-makers, enabling optimal allocation of limited resources (9).
In cost-effectiveness analyses, the clinical output of interventions is measured using similar physical or natural units. The most common unit is life-years-gained (LYG), representing the additional years added to a patient’s life expectancy as a result of an intervention or medication [10].
Cost-Utility Analysis is recognized as the gold standard in pharmaceutical analysis. This type of analysis evaluates both the quality and quantity of life, with the most common measure being quality-adjusted-life-years (QALY) [11].
Modeling in pharmacoeconomics serves as a framework for aggregating and utilizing information and evidence from various sources, as well as predicting the outcomes of different interventions over time. Modeling facilitates predictions and understanding by collecting necessary information from diverse sources.
The choice of modeling method should align with the type of analysis and disease being studied. Markov models are particularly effective for analyzing chronic and recurring diseases, such as cardiovascular diseases [12].
For the economic evaluation, the Markov modeling method was employed using Microsoft Excel 2017, conducting economic analyses of cost-effectiveness and cost-utility.
A hypothetical population of 1,000 individuals was created, averaging 55 years old, all with a history of MI. The effects of using clopidogrel compared to prasugrel were examined within this population. Patient modeling continues until death or until reaching the age of 100 years, whichever occurs first.
The time horizon selected for Markov modeling is divided into equal time periods known as Markov cycles. Each cycle represents the duration an individual spends in one health state before transitioning to another. Throughout each cycle, all information and conditions remain constant. At the end of each cycle, the model reassesses each patient’s status to determine the proportion of the population transitioning from one health state to another and the proportion remaining stable in their current state.
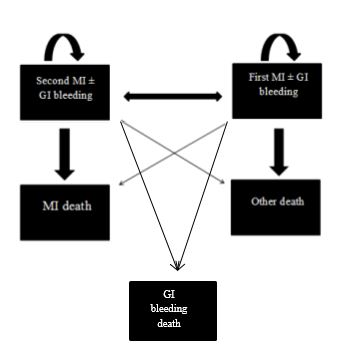
This study utilized separate health states for the first year following a non-fatal heart attack and for subsequent years. In the studied model, after the initial heart attack, patients may experience another heart attack, die from a MI, or die from other causes. If none of these events occur, the patient transitions to the next cycle as a person with a history of heart attack, considering the associated probabilities and costs.
These individuals can either remain in their current state or transition to other states (recurrence of stroke or death). If none of these events occur, the patient transitions to the next cycle of the model as an individual with a history of myocardial infarction, accounting for the probabilities and costs associated with that condition, which differ from those in the first year of the disease (figure 1).
In this study, the payer’s perspective was adopted. This perspective considers all expenses incurred by the patient, healthcare system, or health insurance, providing valuable insights for healthcare planning and policy-making. While this view may not encompass all aspects, such as productivity and indirect costs, the results can still inform the planning of the Ministry of Health and Medical Education and insurance organizations [13].
To calculate Quality-Adjusted Life Years (QALY), the Utility-Weight of each health state was utilized. In this context, a utility-weight of one represents a state of perfect health, while a utility weight of zero corresponds to death [14,15].
For this model, the utility weight is set at 0.98 for a patient who has experienced a GI bleeding and 0.76 for a patient who has experienced a heart attack and 0.88 for a patient in the years following a heart attack, as determined in previous studies using the EQ-5D questionnaire method [16,17].
Discounting is used to estimate the present value of costs and outcomes that occur over different time periods. Discount rates of 5% and 5.8% were considered for effects and costs, respectively according to the Iranian FDA [15].
To deal with uncertainties, one-way sensitivity analysis was conducted as a method in which only one input parameter in the model is altered within a defined range, and the effect of this change on the final model outcome is examined [18].
Also Probabilistic-Sensitivity-Analysis (PSA) was conducted to investigate uncertainties in the input variables, considering beta distribution for probabilities and log-normal distribution for costs, relative risks and discount rates [18].
The Cost-Effectiveness-Acceptability-Curve (CEAC) illustrates the probability that one intervention is cost-effective compared to another across a range of maximum willingness-to-pay values (the amount an individual is willing to pay for each additional unit of effectiveness). In this study, the CEAC for prasugrel versus clopidogrel for the secondary prevention of myocardial infarction was examined [19,20].
Relative Risks represent the ratio of the probability of an event occurring in a population receiving a desired intervention to the probability of the same event occurring in a control group that does not receive the intervention. (Table 1.)
Transition probability estimates the number of patients who will move from one health state to another over a specified period. The results of clinical studies and previous research inform the determination of these transition probabilities, which are presented in Table 2 [28,29].
In this model, a hypothetical cohort of 1,000 patients with a history of myocardial infarction was included. These individuals can either remain in their current state or transition to other states (recurrence of stroke or death). If none of these events occur, the patient transitions to the next cycle of the model as an individual with a history of myocardial infarction, accounting for the probabilities and costs associated with that condition, which differ from those in the first year of the disease. Patient modeling continues until death or until reaching the age of 100 years, whichever occurs first (Figure 2).
The treatment protocol was established as follows: in the first group, during the first year post-heart attack, each individual receives 75 mg of clopidogrel, 80 mg of aspirin, and one of the statins (atorvastatin). In the second group, patients take 10 mg of prasugrel, 80 mg of aspirin, and one of the statins (atorvastatin). In subsequent years of the disease, individuals continue to use aspirin and statins, and only in the event of a recurrent heart attack is clopidogrel or prasugrel reintroduced into the treatment regimen.
The time horizon selected for Markov modeling is divided into equal time periods known as Markov cycles. Each cycle represents the duration an individual spends in one health state before transitioning to another. Throughout each cycle, all information and conditions remain constant. At the end of each cycle, the model reassesses each patient’s status to determine the proportion of the population transitioning from one health state to another and the proportion remaining stable in their current state.
The cost assessment was conducted based on the selected perspective (payer).(Tables 3 and 4)
In this study, two scenarios were examined: one based on public tariffs (the base scenario) and another based on private tariffs.
Also two scenarios were considered for the discount rate:
1. No discounting applied (0%).
2. Discount rates of 5.8% for costs and 5% for outcomes, based on the Iranian FDA.
The following formula is used for discounting:
- R = cost or effectiveness in the future
- i = discount rate
- t = time
Incremental Cost-Effectiveness Ratio (ICER)
The results obtained from cost-effectiveness and cost-utility analyses were calculated as the Incremental Cost-Effectiveness Ratio (ICER) using the following formula:
Sensitivity Analyses
One-way sensitivity analysis involves changing only one input parameter each time in the model within a specified range and assessing the effect of this change on the final model outcome.
Probabilistic sensitivity analysis is a valuable method for assessing the level of uncertainty that helps decision-makers in decision-making.
The Cost-Effectiveness Acceptability Curve (CEAC) illustrates the probability that an intervention is cost-effective compared to another intervention across a range of maximum willingness-to-pay values for the defined outcome.
Costs and Outcomes Assessment Results
Outcomes were measured in terms of Life Years Gained (LYG) for cost-effectiveness assessment and Quality-Adjusted Life Years (QALY) for cost-utility assessment. The results indicate that the prasugrel group achieved higher outcomes than the clopidogrel group (table 5).
Patients in the clopidogrel group incur higher costs than those in the prasugrel group across all scenarios (table 5). Despite the lower public tariff compared to the private tariff, the incremental cost difference is greater under the public tariff.
The resulted ICERS for public tariffs are presented in table 6:
Sensitivity Analyses Results
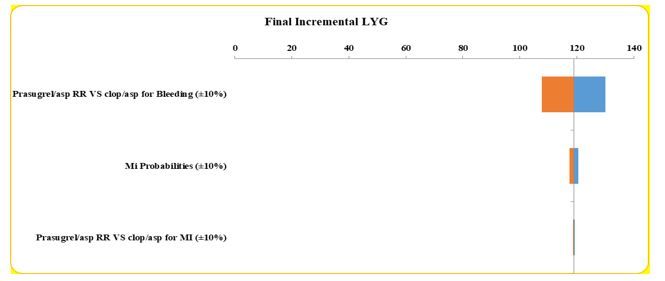
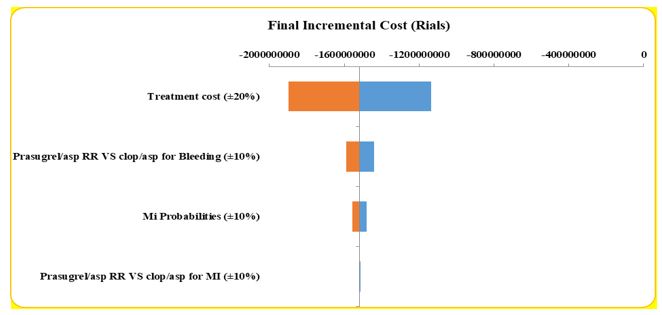
One-way sensitivity analysis results are depicted using tornado diagrams in Figures 2-4. These diagrams illustrate the parameters that were altered and their respective ranges of change. This analysis showed that the results are particularly sensitive to to the risk of gastrointestinal bleeding with prasugrel and aspirin versus clopidogrel and aspirin and the treatment costs.
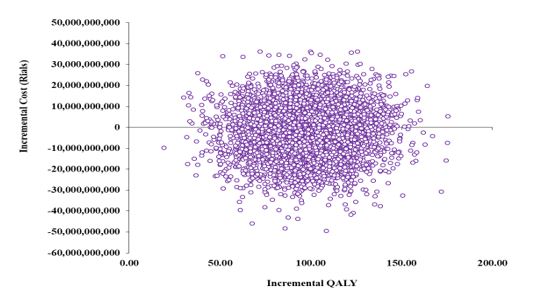
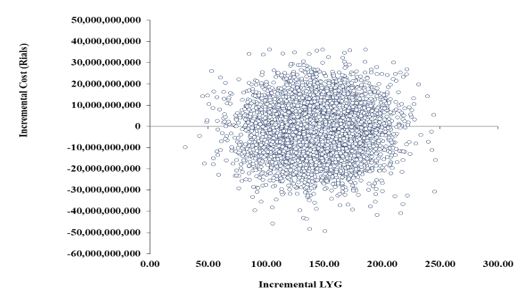
The results of PSA for 5,000 iterations of the analysis, using randomly selected numbers within the specified ranges according to each parameter’s statistical distribution are presented in figures 5 and 6.
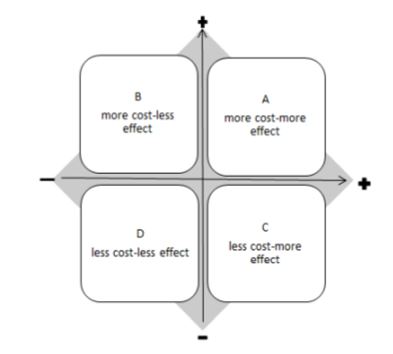
the cost-effectiveness diagram includes four quadrants (figure 7):
- Area A, where both costs and effects are higher.
- Area C, which represents the most favorable outcome, showing lower costs and higher effects.
- Area D, where both costs and effects are negative.
- Area B, which shows interventions with higher costs and lower effects.
In the sensitivity analysis of this model, as illustrated in the scatter diagrams (Figures 5 and 6), 44% of the points show saving in cost with the use of prasugrel while all the iterations resulted in higher outcomes for the use of prusugrel.
While Iran has not established a threshold limit for cost-effectiveness analyses, WHO guidelines suggest that if the incremental cost-effectiveness per QALY is less than the country’s GDP per capita, the analysis is considered very cost-effective. All of the 5000 iterations in the PSA were below this threshold.
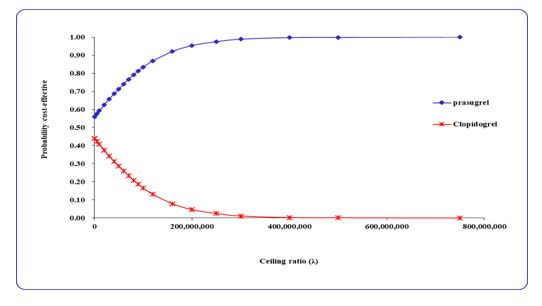
The results of the CEAC are presented in Figure 8. According to this diagram, at any willingness to pay per additional QALY the cost-effectiveness of prasugrel for secondary MI prevention is likely to be favorable (Figure 8).
Discussion
According to the public sector tariffs, the cost per patient over the model time horizon in the main scenario (with no discounting) is 233446743 Rials in the prasugrel group and 234964173 Rials in the clopidogrel group, indicating that the use of prasugrel in the public tariff will result in a lower cost compared to clopidogrel for each patient.
In the private sector tariff, the cost per person when using prasugrel is 639007826 Rials and in the clopidogrel receiving group is 639029260 Rials, which means that a patient receiving clopidogrel with a private tariff pays an average of 21434 Rials more than each patient in the prasugrel group.
According to the estimates made above and considering the costs, although the public tariff is lower than the private tariff, the difference in costs (incremental cost) in the public tariff is higher than the private one. According to Table 5, with a zero discount rate, an average of 0.11 QALYs was obtained for each person throughout life, indicating that in this case, the use of prasugrel throughout life brings more QALYs for each person.
Although in Iran, a threshold for cost-effectiveness analysis has not been specified, according to the WHO proposal, if the incremental cost-effectiveness per unit of QALY is less than the gross domestic product (GDP) per capita of a country, the relevant analysis is considered highly cost-effective. In our study, none of the PSA points exceeded this threshold [14].
Many studies have been conducted in the field of comparing the effects and side effects of prasugrel compared to clopidogrel in the world [20,33]. The cost-effectiveness of these drugs has also been examined separately [22,34,35]. For example, in 2015, a study was conducted in Canada by Husam Abdel-Qadir et al. over 12 months in which the costs and consequences of using prasugrel, clopidogrel, and ticagrelor were compared. In this study, prasugrel was estimated to be more cost-effective compared to clopidogrel with higher cost than clopidogrel [35]. However, in our study, which was conducted for the entire life of patients after myocardial infarction, prasugrel was reported to be both more effective and, in most cases, less expensive than clopidogrel.
Since none of the studies examined the simultaneous use of clopidogrel and aspirin in comparison with prasugrel and aspirin in preventing recurrent myocardial infarction, and these studies differ from the present study in terms of subject matter, the adopted perspective, time horizon, and specifications of the model used, it is not possible to compare them directly with our study.
Limitation
Although recurrent MI is not the only cardiovascular complication, however, in order to make modeling feasible, MI was considered as the only cardiovascular complication of the patient.
Given the price difference between the foreign and Iranian brands of clopidogrel and prasugrel, only the Iranian generic type, which is conditionally approved by insurance, was considered in this study. However, in the sensitivity analysis, the effect of price changes on the final results of the model was examined.
Although the latest announced tariffs were used in this study, due to the frequent changes in costs, it is necessary to consider this issue when using this study as a background for other studies.
In this study, due to the lack of domestic resources to determine the utility weight for calculating QALY, international resources were used, although the possible impact of these cases was examined in the sensitivity analysis.
Since in modeling studies for a hypothetical cohort population, the average costs related to a patient must be considered, despite the differences in treatment methods and treatment costs between individual patients, an average treatment method has been considered.
Although this payer perspective cannot include all aspects such as productivity and indirect costs, the results of this study can be used in the planning of the Ministry of Health and Medical Education, as well as insurance organizations.
The generalizability of these results to other settings should be approached with caution.
Conclusion
This study showed that prasugrel in patients who have previously had a heart attack could be considered a very cost-effective intervention according to WHO criteria for annual GDP per capita in Iran.
Acknowledgment
This study was financially supported by the Vice-Chancellery for Research of Isfahan University of Medical Sciences, Isfahan through Grant No. 3991084.
Conflict of Interest Statement
The authors declare that they have no conflicts of interest.
Authors’ Contribution
M. Amirsadri and V. Hajhashemi contributed to the conception of the work. M. Amirsadri, V. Hajhashemi and M. Azadi contributed to the data acquisition. M. Amirsadri contributed to the model design and analyses, interpretation of results. V. Hajhashemi and M. Azadi contributed to the manuscript preparation. M. Amirsadri Supervised the work. All authors approved the final version of the manuscript.
- NICE (2010) prevention of cardiovascular disease.NICE clinical guideline.No. 25. Manchester: NICE.
- Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M, et al. (2015) Heart Disease and Stroke Statistics-- 2015 Update A Report From the AHA. Circulation. 131: 00-00.
- WHO (2017) Cardiovascular diseases. Available from:https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds).
- Lin CC, Hu HY, Luo JC, Peng YL, Hou MC, Lin HC, et al. (2013) Risk factors of gastrointestinal bleeding in Clopidogrel users: a nationwide population-based study. Alimentary pharmacology & therapeutics, 38: 1119-28.
- Lee CH, Huang MS, Chao TH, Lin SH, Li YH (2021) Reduced-Dose Prasugrel versus Clopidogrel for Patients Undergoing Percutaneous Coronary Intervention. Int Heart J. 62: 246-55.
- Wu W, Liu J, Yu H, Jiang Z (2020) Antiplatelet therapy with or without PPIs for the secondary prevention of cardiovascular disease in patients at high risk of upper gastrointestinal bleeding: A systemic review and meta-analysis. Experimental and Therapeutic Medicine. 19: 3595-603.
- Mitsis A, Gragnano F (2021) Myocardial Infarction with and without ST-segment Elevation: a Contemporary Reappraisal of Similarities and Differences. Curr Cardiol Rev. 17: e230421189013.
- Ancrenaz, V, Daali, Youssef & Fontana, Pierre & Besson, Marie & Samer, Caroline & Dayer, P & Desmeules, Jules (2010) Impact of Genetic Polymorphisms and Drug – Drug Interactions on Clopidogrel and Prasugrel Response Variability. Current drug metabolism. 11: 667-77.
- Rascati KL (2009) Essentials of Pharmaeconomics.Texas: Lippincott Williams & Wilkins.
- Townsend R (2024) Principle of pharmacoeconomics, https://www.worldcat.org/title/principles-of-pharmacoeconomics/oclc/456758033.
- Cooper K, Brailsford SC, Davies R (2007) Choice of modelling technique for evaluating health care interventions. Journal of the ORS. 58: 168-76.
- Scuffham PA, Whitty JA, Mitchell A, Viney R (2008) The use of QALY weights for QALY calculations.PharmacoEconomics. 26: 297-310.
- Weintraub WS (2003) editor. CHCE. Totowa (NJ): Humana Press.
- Edejer T, Baltussen R, Adam T, Hutubessy R, Acharya A, Evans DB, et al. (2003) Making choices in health: WHO Guide to Cost-effectiveness Analysis.
- Drummond MF. Methods for the Economic Evaluation of Health Care Programmes: Oxford University Press.
- Lamotte M, Annemans L, Evers T, Kubin M (2006) A multi-country economic evaluation of low-dose aspirin in the primary prevention of cardiovascular disease. Pharmacoeconomics. 24: 155-69.
- Goodacre S, Nicholl J, Dixon S, Cross E, Angelini K, Arnold J, Revill S, Locker T, Capewell SJ, Quinney D, Campbell S, Morris F (2004) Randomised controlled trial and economic evaluation of a chest pain observation unit compared with routine care. BMJ. 328: 254.
- Jain R, Grabner M, Onukwugha E (2011) Sensitivity analysis in cost-effectiveness studies. PharmacoEconomics. 29: 297-314.
- Barton GR, Briggs AH, Fenwick EA (2008) Optimal cost-effectivness decisions: the role of the cost-effectivness acceptability curve(CEAC),the cost-effectivness acceptability frontier (CEAF), and the expected value of perfection information (EVPI). Value in Health. 11: 886-97.
- Bastian Wein, Michael Coslovsky, Reza Jabbari, Søren Galatius, Matthias Pfisterer, Christoph Kaies (2017) Prasugrel vs. clopidogrel in contemporary Western European patients with acute coronary syndromes receiving drug-eluting stents: Comparative cost-effectiveness analysis from the BASKET-PROVE cohorts,International Journal of Cardiology, 248: 20-7.
- Collaboration AT (2002) Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ. 324: 71-86
- Schleinitz MD, Weiss JP, Owens DK (2004) Clopidogrel versus aspirin for secondary prophylaxis of vascular events: a cost-effectiveness analysis. AJM, 116: 797-806.
- Antiplatelet Trialists' Collaboration (1994) Collaborative overview of randomised trials of antiplatelet therapy Prevention of death, myocardial infarction, and stroke by prolonged antiplatelet therapy in various categories of patients. Bmj, 308: 81-106
- Berger JS (2018) Oral Antiplatelet Therapy for Secondary Prevention of Acute Coronary Syndrome. Am J Cardiovasc Drugs, 18: 457-72.
- Lin KJ, De Caterina R, Rodríguez LAG (2014) Low-dose aspirin and upper gastrointestinal bleeding in primary versus secondary cardiovascular prevention: a population-based, nested case–control study. Circulation: Cardiovascular Quality and Outcomes, 7: 70-7.
- Peng CH, Huang TP, Chen YH, Hsu CH, Cheng IL (2022) Clinical efficacy and safety of reduced-dose prasugrel versus clopidogrel in patients undergoing percutaneous coronary intervention: a systematic review and meta-analysis of randomized controlled trials. Acta Cardiologica Sinica, 38: 425.
- García Rodríguez LA, Lin KJ, Hernández-Díaz S, Johansson S (2011) Risk of upper gastrointestinal bleeding with low-dose acetylsalicylic acid alone and in combination with clopidogrel and other medications. Circulation. 123: 1108-15.
- Who.int.America: Life tables by country Iran (Islamic Republic of). updated 2020-12-06. Available from: https://apps.who.int/gho/data/view.searo.60760?lang=en
- Who.int.America: Incidence of acute myocardial infarction in Islamic Republic of Iran: a study using national registry data in 2012.
- Tariff and relative value of health services. https://rvu.behdasht.gov.ir/.
- Iran FDA, list and price of pharmaceutical items. https://ifdana.fda.gov.ir/fa/News/12347
- Amirsadri M, Hassani A (2015) Cost-effectiveness and cost-utility analysis of OTC use of simvastatin 10 mg for the primary prevention of myocardial infarction in Iranian men. Daru. 23: 56.
- Davies A, Bakhai A, Schmitt C, Barrett A, Graham-Clarke P, Sculpher M (2013) Prasugrel vs clopidogrel in patients with acute coronary syndrome undergoing percutaneous coronary intervention: a model-based cost-effectiveness analysis for Germany, Sweden, the Netherlands, and Turkey. Journal of Medical Economics, 16: 510-21.
- Husam Abdel-Qadir, Idan Roifman, Harindra C (2015) Wijeysundera Cost-effectiveness of clopidogrel, prasugrel and ticagrelor for dual antiplatelet therapy after acute coronary syndrome: a decision-analytic model, 3: E438-46.
- Mauskopf JA, Graham JB, Bae JP, Ramaswamy K, Zagar AJ, Magnuson EA, Meadows ES (2011) Cost-effectiveness of prasugrel in a US managed care population. Journal of Medical Economics, 15: 166-74.


Tables at a glance
Figures at a glance