Preparation and Characterization of Transdermal Patch for Arthritis
Received Date: March 05, 2026 Accepted Date: March 10, 2026 Published Date: March 16, 2026
doi:10.17303/jra.2026.1.105
Citation: Muktamadhavi, Rajesh Kumar Nema, Gyanesh Kumar Sahu (2026) Preparation and Characterization of Transdermal Patch for Arthritis. J Rheumatol Arthritis 1: 105
Abstract
Millions of people worldwide suffer from arthritis, a common inflammatory disease that requires efficient and focused treatment approaches to reduce pain and enhance joint function. In order to treat arthritis locally, this study focuses on the creation and characterization of a Transdermal patch intended for the sustained release of anti-inflammatory drugs.
To improve drug solubility and skin permeability, a combination of biocompatible polymers, such as Polyvinyl Pyrrolidine (PVP) and Hydroxypropyl methylcellulose (HPMC), were used in the formulation of transdermal patches. The patches' physical characteristics, such as their thickness, tensile strength, and moisture content, were assessed after they were loaded with Ibuprofen, a model anti-inflammatory medication. The results showed a pattern of controlled and sustained release. The Transdermal patches were applied to a rat model of Carrageenan /Kaolin induced arthritis for in vivo assessment. Clinical scoring of joint inflammation is used to evaluate the effectiveness of the treatment. In addition to a decrease in inflammatory markers, the results showed that the treated group experienced a significant reduction in pain and swelling when compared to the control group.With benefits like increased patient compliance and fewer systemic side effects, this study shows that Transdermal patches have the potential to be a successful delivery method for the treatment of arthritis. In larger animal models and clinical trials, more research is required to optimize formulation parameters and examine the long-term safety and effectiveness of this novel therapeutic approach.
Keywords: Transdermal Patch; Arthritis; Anti-inflammatory; Invivo Study; Ibuprofen.
Highlights
- As of now, a number of compounds have been developed in the management of the various forms of arthritis.
- However, the use of these compounds via oral and parenteral routes have a number of disadvantages.
- Transdermal delivery not only increases passive permeation of the drug but also improves compliance towards the patients that are on treatment.
- To enhance the speed of the movement from the clinic to the market, the safety considerations have to be looked at very early.
Introduction
- As of now, a number of compounds have been developed in the management of the various forms of arthritis.
- However, the use of these compounds via oral and parenteral routes have a number of disadvantages.
- Transdermal delivery not only increases passive permeation of the drug but also improves compliance towards the patients that are on treatment.
- To enhance the speed of the movement from the clinic to the market, the safety considerations have to be looked at very early.
Arthritis originates from the Greek word, meaning ‘a disease of the joints.’ It is described as joint inflammation, usually acute or chronic. Pain and structural damage typically accompany it [1].
Arthritis and arthralgia are not the same. While the latter describes pain confined to a joint, the source of which may or may not be inflammation, arthritis is not limited to inflammation of the joint. It is believed arthritis afflicted Neanderthals and ancient Egyptians. Dr. John K. Spencer first used “osteoarthritis” in 1886. It is estimated that there are over 100 types of arthritis, with osteoarthritis (also known as degenerative arthritis) being the most common. Osteoarthritis is non-inflammatory. Inflammatory arthritis can occur in several conditions, caused by autoimmune processes (as in rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, etc.), inflammation induced by deposition of crystals (in gout, pseudogout, basic calcium phosphate disease), or infections like septic arthritis and Lyme’s arthritis. Other autoimmune connective tissue diseases may accompany inflammatory arthritis, such as systemic lupus erythematosus, Sjogren’s syndrome, scleroderma, myositis, inflammatory bowel disease, celiac disease, etc. [2].
The different forms of arthritis are characterized by unique variations. For instance, in the case of osteoarthritis, age, gender (female), joint injuries, and obesity are key contributory factors. Certain genetic considerations have also been noted such as mutations affecting collagen types II, IV, V, and VI [3].
Rheumatoid arthritis (RA) is classified as an autoimmune condition wherein multiple joints are simultaneously affected along with the auxiliary tissues like bones, cartilage and synovial tissues leading to osteitis, synovitis and bone destruction. The bone damage by joint subchondral sclerotic erosion degenerates the condition, progressively reducing the joint space, causing severe joint destruction, functional loss, and ultimately disability [4].
RA primary targets include the knees, wrists, fingers, and interphalangeal joints of legs. The inflamed joints that are stiff in the morning usually are warm and painful to touch. On palpation, the rheumatoid patients display pain in the joints and osteoarthritic changes [5].
Patients suffering from rheumatoid arthritis (RA) tend to become disabled within a decade of diagnosis, which calls for more focused efforts on early diagnosis to contain inflammation and damage to the cartilage and bone of joints [6].
Prognostic markers pertinent to arthritis are grouped into two classes; those that pertain to predicting the signs and symptoms of rheumatoid arthritis (RA) and those that assess the damage done to bone and cartilage commonly affected as entails from the age course of 40 to 60, although this condition can affect children as well. The condition is progressive in nature, exacerbated by age, and a noted hallmark is joint stiffness. Ultimately, mobility becomes highly restricted and painful sensations are experienced during movement of the joints [7].
It has been documented that RA causes the activation of multiple different pathological signaling pathways, which leads to the progressive and irreversible destruction of joints. The development and persistence of fully developed RA necessitate the involvement of multiple cells, including synovial cells, dendritic cells, macrophages, and several other immune cells, such as rheumatoid arthritis synovial fibroblasts (RASF), in contrast to other inflammatory disorders [8].
The pathogenesis of RA is significantly influenced by a number of signaling pathways, such as the innate immune system's Toll-like receptor (TLR), apoptotic effector molecules, intracellular kinases, transcriptional factors, and pro-inflammatory cytokine mediators such as interleukins (IL-6, IL-1B), tumor necrosis factor-alpha (TNF-α), and C-reactive proteins [9].
Local cellular component interactions cause cellular proliferation and joint destruction. RASF are not only activated by the early response genes, proto-oncogenes, and intracellular signaling mechanism that led to the production and release of tissue-damaging molecules; Pro-inflammatory cytokines also directly relate to synovial inflammation. The most implicated cytokines in RA by means of RASF activation are IL-1B, IL-6, and TNF-\u03b1. Activated macrophages abundant in the synovial tissues of the affected joints release these cytokines [10]. In the treatment of RA, non-steroidal anti-inflammatory medications (NSAIDs) are advised as first-line agents. By blocking the COX enzyme, NSAIDs act to stop prostaglandin production [11].
Ibuprofen, diclofenac, piroxicam, ketoprofen, naproxen, and flurbiprofen are among the NSAIDs mostly suggested for the treatment of chronic pain [12].
Ibuprofen (IB), which is taken orally in doses of 200 and 400 mg, is deemed a well-known, safe, and widely used among other NSAIDs by the WHO's list of essential medications. In conditions including RA, osteoarthritis, muscular discomfort, common cold, and uveitis, IBU is used to control moderate to severe pain, fever, and inflammation. The biopharmaceutical classification system (BCS) classifies Ibuprofen as BCS class II with low solubility and high permeability profile [13].
Etiology
Age, female gender, obesity, anatomical factors, muscle weakness, and joint injury (occupation/sports activities) are among the risk factors for developing osteoarthritis. The most prevalent subtype of primary osteoarthritis is the disease. Though linked with the risk factors, it is diagnosed in the absence of a predisposing trauma or disease [14].
Secondary osteoarthritis develops with a preexisting joint anomaly. Predisposing diseases include trauma or injury, congenital joint disorders, inflammatory arthritis, avascular necrosis, infectious arthritis, Paget disease, osteopetrosis, osteochondritis dissecans, metabolic disorders (hemochromatosis, Wilson's disease), hemoglobinopathy, Ehlers-Danlos syndrome, or Marfan syndrome [15].
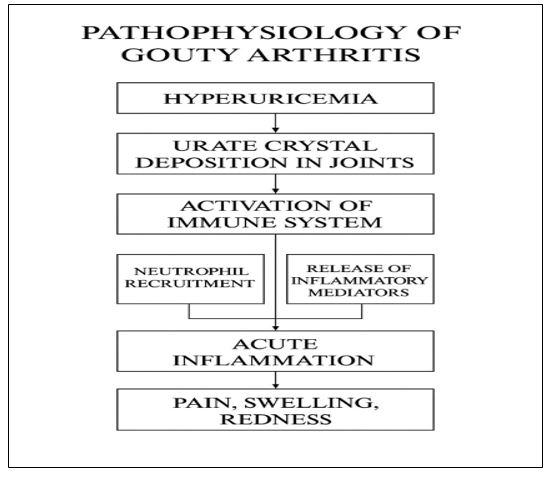
Prolonged hyperuricemia in gout causes uric acid buildup in the joints, which in turn causes inflammation. Less than 10% of gout is caused by hyperuricemia, which can be caused by a number of genetic mutations. Most gout sufferers are under-excretors, meaning they are unable to eliminate all of the uric acid generated by their body's endogenous or exogenous purine metabolism. Other risk factors for hyperuricemia and gout include male sex, aging, chronic kidney disease, alcoholism, and certain medications like diuretics [17].
Patients with pre-existing risk factors, such as immunodeficiency, aging, diabetes mellitus, prosthetic joints, rheumatoid arthritis, and intravenous drug abuse, are more susceptible to septic arthritis, an acute form of arthritis that is uncommon in the general population [18].
One of the most prevalent clinical characteristics in patients with systemic lupus erythematosus (SLE) is arthritis, which is also commonly observed in patients with other autoimmune diseases. Inflammatory bowel disease, psoriasis, celiac disease, Sjogren syndrome, systemic sclerosis, dermatomyositis, mixed connective tissue disease (MCTD), and others are often linked to arthritis [19].
Pathophysiology
A degenerative cascade of progressive cartilage loss that results in bone damage is the hallmark of osteoarthritis. Osteophytes, subchondral plate thickening, and subchondral cysts are characteristic findings. Joint collage is broken down by proteolytic enzymes like matrix metalloproteinases, serine proteases, and cysteine proteinases that are induced by interleukin-6, monokines, interferon-induced protein-10, and macrophage chemotactic protein. The cartilaginous matrix becomes thinner and eventually disintegrates when the surrounding articular cartilage calcifies. Additionally, a decline in chondrocyte function is linked to aging, increasing the risk of osteoarthritic degeneration [20].
Generally speaking, rheumatoid arthritis symptoms are worse than those of osteoarthritis. An autoimmune reaction to an environmental trigger results in rheumatoid arthritis, a chronic, systemic inflammatory disease. Endothelial cell activation and synovial cell hyperplasia occur before cartilage and eventually bone deterioration. After exposure to an antigenic pathogen, the pathology develops as a result of the abnormal production of inflammatory mediators, including interleukins 1, 6 and 8, tumor necrosis alpha, and others [21].
Gout's monosodium urate salts form needle-shaped crystals when they precipitate. Cooler body parts and acidic environments are more likely to experience this crystallization. The usual acute flare-up of gouty arthritis is caused by an inflammatory response mediated by IL-1 when these deposited intraarticular uric acid crystals become unstable. In pseudogout, the process is different because calcium pyrophosphate dihydrate is created when chondrocyte-derived inorganic pyrophosphate combines with calcium. This crystal is placed in joint spaces that are prone to osteoarthritis. Osteophytes, subchondral cysts, and bone and cartilage fragmentation are all examples of pseudoout crystal damage. Calcium pyrophosphate deposition is more likely in metabolic diseases like hemochromatosis, hyperparathyroidism, or hypomagnesemia [22].
An inflammatory reaction to a monobacterial infection is usually the cause of septic arthritis. Cytokines, chemokines, and proteases are released when bacteria enter the synovial fluid, breaking down cartilage and causing the synovial membrane to swell. Bacterial toxins also have a detrimental effect on the joint space itself. The most prevalent pathogen in adults is Staphylococcus aureus, though streptococci strains are also prevalent. Gram-negative bacterial infections are more frequently observed in the elderly and very young, as well as in those who have experienced trauma, intravenous drug use, or immunosuppression [23].
Drug Profile
A popular nonsteroidal anti-inflammatory medicine (NSAID) with analgesic, anti-inflammatory, and antipyretic effects is ibuprofen. Since its initial introduction in the 1960s as a less dangerous substitute for aspirin in the treatment of rheumatoid arthritis, it has grown to become one of the most widely used over-the-counter drugs globally. Ibuprofen, which is chemically categorized as a derivative of propionic acid, works by non-selectively inhibiting the cyclooxygenase (COX) enzymes, COX-1 and COX-2, which lowers the production of prostaglandins that are implicated in pain and inflammation. It is appropriate for treating mild to moderate pain, fever, and chronic inflammatory diseases such rheumatoid arthritis and osteoarthritis because to its superior safety profile, particularly at low dosages, and comparatively short half-life. Modern drug delivery technologies, such as transdermal patches, nanoformulations, and mucoadhesive preparations, are designed to maximize the therapeutic benefits of ibuprofen while reducing the gastrointestinal adverse effects that are frequently connected to oral administration. Additionally, there is ongoing research into ibuprofen's possible function in regulating neuroinflammation and its effects on illnesses other than pain management, like cancer and Alzheimer's. Despite its extensive use, dosage, duration, and patient-specific risks need to be considered, particularly in those with renal or cardiovascular impairment [24].
Genetic predisposition combined with environmental triggers most certainly causes rheumatoid arthritis (RA). Although genetics are important, environmental elements such smoking, work exposures, and even dietary practices can greatly affect the evolution and progression of RA. Rheumatoid arthritis (RA) risk has been linked to several genetic and environmental variables. Amongst these, the most notable correlations have been with female sex, a family history of RA, the genetic component the 'shared epitope' and with tobacco smoke exposure [16].
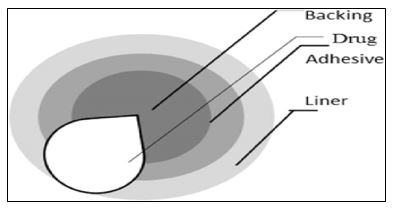
Transdermal Patch
To administer a particular dosage of medication through the skin and into the bloodstream, a medicated adhesive patch known as a transdermal patch or skin patch is applied to the skin. Often, this promotes healing to an injured area of the body. In December 1979, the U.S. Food and Drug Administration authorized the first prescription patch with scopolamine for motion sickness that was sold commercially [40].
The nicotine patch, which distributes nicotine to aid in quitting tobacco use, was the best-selling transdermal patch in the US. In 2007, Europe approved the first vapor patch to decrease smoking that was sold commercially. Fentanyl, an analgesic for severe pain, nitroglycerin patches for angina, lidocaine patches, marketed as Lidoderm, which relieve the peripheral pain of shingles (herpes zoster), and buprenorphine, marketed as Butrans, which is an analgesic for moderate to severe chronic pain are among the other patches that are on the market [41].
Types of Transdermal Drug Delivery System
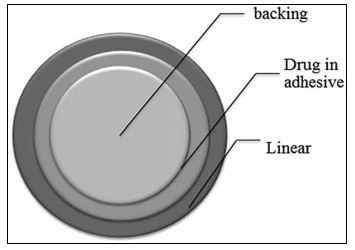
Single-layer Drug-in-Adhesive
The Single-layer Drug-in-Adhesive system is characterized by the inclusion of the drug directly within the skin contacting adhesive. The adhesive in this transdermal system design acts as both the basis for the formulation, holding the medicine and all of the excipients under a single backing film, in addition to attaching the system to the skin. Diffusion across the skin determines the drug's release rate from this kind of device [42].
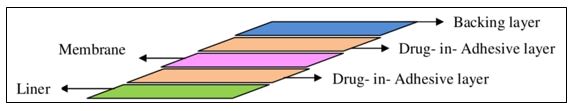
Multi-layer Drug-in-Adhesive
Similar to single-layer drug-in-adhesive, multi-layer drug-in-adhesive incorporates the drug directly into the adhesive. Multiple drug-in-adhesive layers under a single backing film or the inclusion of a membrane between two separate drug-in-adhesive layers are both included in the multi-layer [43].
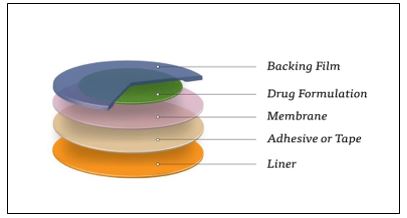
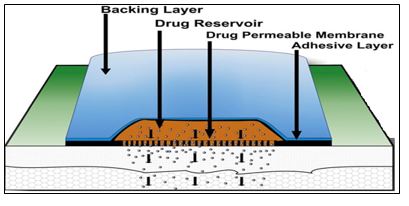
Drug Reservoir-in-Adhesive
A liquid compartment with a medication solution or suspension that is isolated from the release liner by an adhesive and semi-permeable membrane defines the Reservoir transdermal system design. The product's adhesive ingredient that promotes skin adhesion can be integrated in a concentric pattern around the membrane or as a continuous layer between the membrane and the release liner [44].
Drug Matrix-in-Adhesive
A semisolid matrix containing a medication solution or suspension that comes into direct touch with the release liner is what defines the matrix system design. The skin-adhesive component is integrated into an overlay and surrounds the semisolid matrix in a concentric pattern [45].
Components of Transdermal Patch
The basic components of transdermal patch consist of:
A Polymer Must Meet the Following Requirements in Order to Be Utilized in A Transdermal System:
The Advantages of Delivering Drugs Through the Skin Includes: [50-52]
The Disadvantages of the Transdermal Delivery System: [53]
Materials [54]
Methods
Determining the maximum wavelength of ibuprofen:
- Polymer matrix / Drug reservoir
- Active ingredient (drug)
- Permeation enhancers
- Pressure sensitive adhesive (PSA)
- Backing laminates
- Release liner
- And other excipients like plasticizers and solvents [46].
- Polymer Matrix
- Polymers are the important parameter of TDDS, which control the release of the drug from the device. Polymer matrix can be prepared by dispersion of drug in liquid or solidstate synthetic polymer base.
- Natural Polymers
- e.g. cellulose derivatives, zein, gelatin, shellac, waxes, gums
- Synthetic Elastomers
- e.g. polybutadiene, hydrin Rubber, polyisobutylene, silicon acrylonitrile,Neoprene, butylrubber etc.
- Synthetic Polymers
- e.g. polyvinyl alcohol, polyvinylchloride, polyethylene, polyacrylate, polyamide, polyurea, polyvinylpyrrolidone etc.
- The polymer's molecular weight and chemical properties should be such that a particular drug diffuses and is delivered through it in an appropriate manner. The polymer should be easily produced into the required product, stable, and non-reactive. Both the polymer and the byproduct of its breakdown must not be harmful to the host [47–49].
- Compared to oral administration, a lower dosage of medication was required with the Transdermal Drug Delivery System (TDDS).
- For drugs with gastrointestinal discomfort and poor absorption, skin administration is an appropriate route of administration.
- Lower dosage frequency leads to better patient compliance. steady and optimal blood concentration time profile minimizes the adverse effects.
- Parenteral therapy avoids the risks, discomfort, and inconvenience.
- The release period is longer than that of sustained oral drug delivery systems.
- Skin inflammation is a potential risk.
- Drug binding to the skin may cause dosage dumping.
- It is unable to develop for large-molecule drugs.
- Could result in an allergic reaction.
- Younger and adult skin is more porous than older skin.
- Ibuprofen (API)
- Quercetin (Bioenhancer)
- HPMC (Polymer)
- Polyvinyl pyrolidine (copolymer)
- Polyethylene Glycol (Plasticizer)
- Toluene: Ethanol(solvent)
The maximum wavelength of ibuprofen was determined by measuring the absorbance of 10u g/mL of ibuprofen solution in PH 7.4 phosphate buffer solution. By measurement, it was determined that the maximum wavelength of ibuprofen was 222nm.
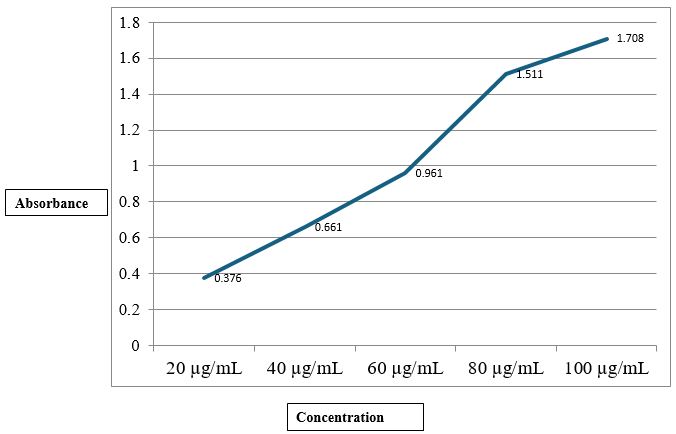
Determination of calibration curve of ibuprofen:
The ibuprofen treatment is then diluted in PH 7.2 phosphate buffer to yield a series of dilutions containing 20,40,60,80,100 of ibuprofen per milliliter of solution. The absorbance of the above dilutions was measured on a UV-visible spectrophotometer at 221nm and 262 nm.
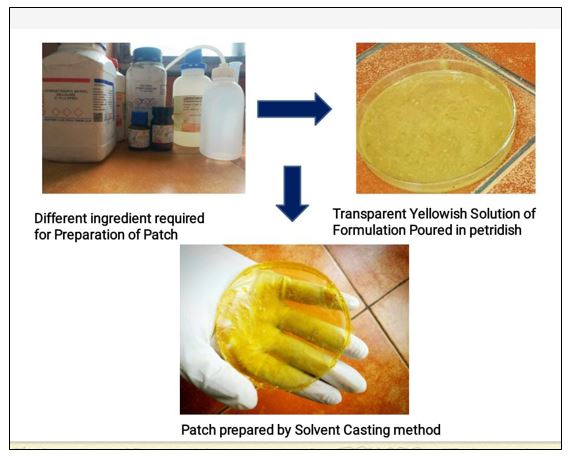
Preparation of patch by Solvent casting Method:
The HPMC and Polyvinyl pyrrolidine were accurately weighed and dissolved in mixture of ethanol solvent
The drug ibuprofen and bioenhancer quercetin was dissolved in toluene and then dispersed in the polymeric solution and plasticizer of Polyethylene Glycol was added with continuous stirring using a magnetic stirrer to obtain homogenous mixture
Glycerin was spread into petridish and the resulting mass was poured into a Petri dish
The Petridish was left undisturbed at room temperature for 24 hrs.
The patch was obtained intact by slowly lifting from the Petridish and transdermal patches were cut into radius of 2 cm square.
Evaluation of Transdermal Patch
Physical Appearance
Every transdermal film was examined organoleptically and visually for color, transparency, shape, surface texture, patch thickness, and film formation (no shrinkage) after drying [55].
Thickness of Patch
A screw gauge was used to measure each patch's thickness at five distinct locations, and the average was computed [56].
Thickness range – 0.12 ± 0.025 to 0.25 ± 0.022
Weight Uniformity
Patches with a diameter of 4 cm and a radius of 2 cm were cut. Five patches' weights were measured, and the weight variation was estimated [57].
Folding Endurance
A uniformly sliced patch with a radius of 2 cm and a diameter of 4 cm was folded repeatedly in the same spot until it broke. The folding endurance value is determined by counting the number of times the film was folded in the same spot without breaking [58].
Percentage Moisture Content
The prepared films were weighed individually and kept in a desiccator containing fuse calcium chloride at room temperature for 24h. After 24h, the films were reweighed and determined the percentage moisture content from the mentioned formula [59].
Percentage Moisture Uptake
To maintain 84% relative humidity, the weighted films were stored in desiccators with a saturated potassium chloride solution at room temperature for 24 hours. The films were reweighed after a day, and the % moisture uptake was calculated using the formula [60] below.
Drug Content
A phosphate buffer solution was used to dissolve the designated patch area. The film was dissolved by stirring the contents. A volumetric flask was used to hold the contents. The drug content was ascertained by measuring the solution's absorbance at a wavelength of 262 nm [61].
Result of Evaluation Studies
Physical Appearance
The patch was visually inspected for color, surface texture, shape. It helps to explain the physical appearance of patch. Transdermal patch cut in 2cm2.
Thickness of Patch
The thickness of the prepared patch was measured by Screw Guage. The mean thickness was measured at different point of the film were given in table.
Thickness range – 0.12 ± 0.025 to 0.25 ± 0.022
The thickness of patch was determined. It was found that F4 (0.22mm) shows less thickness & F1 (0.29mm) shows more thickness whereas marketed patch (F5) shows 0.22 mm thickness which is approx. similar to prepared patch,
Uniformity of Weight
The quantified area of 2cm2 radius is to be cut at different parts of the patch and weigh in digital balance. The average weight calculated from individual weight are shown in table.
Uniformity of weight was measured. It was found that F3 (597mg) shows more weight &F4 (590mg) shows less weight whereas marketed patch shows Uniformity of Weight approx. similar to prepared patch.
Folding Endurance
The folding endurance of the Patches are given below:
Folding endurance of prepared transdermal patches were noted. It was found that more folding endurance value is seen in F2 (30) and less folding endurance value in F4 (26) whereas Folding Endurance of marketed patch (F5) was found to 25 mm which is approx. similar to prepared patch.
Moisture Uptake
Moisture uptake of prepared transdermal patch F1 to F4 were determined. The results were tabulated in Table.
Moisture contents in various formulated patch were determined. It shows that F2 (2.75%) has more moisture content and F3 (2.12%) shows less moisture content whereas marketed patch shows less moisture uptake than prepared patch.
Moisture Loss
Moisture loss of prepared transdermal patch F1 to F4 were determined. The results are tabulated in Table.
Moisture loss were determined and found that F4 (0.90%) shows more moisture loss and F1 (0.68%) shows less loss in moisture whereas marketed patch shows moisture loss approx similar to prepared patch.
In Vivo Animal Study
Materials
Drug Ibuprofen, Quercetin, and Carrageenan were Purchased from ibuychemicals. HPMC, PVP, Methanol, Propylene glycol and Isoflurane was obtained from laboratory of Rungta Institute of pharmaceutical Sciences, Bhilai (C.G). All Reagents were of analytical grade.
Animals
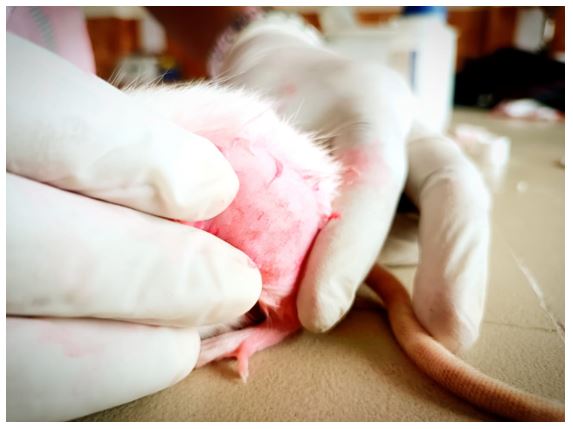
Throughout, 18 male albino rats from the Animal Laboratory House of the Rungta Institute of Pharmaceutical Sciences in Bhilai (C.G.) were utilized. The animals were kept in an air-conditioned animal room with a 12-hour light/dark cycle (08:00–20:00 h light, 20:00–08:00 h dark), at 23 ± 5 °C and 55 ± 10% RH, and given water and a laboratory meal for a week prior to the tests. Animals weighing 150–200 g on average were used in the investigation. The rats were given anesthesia and had their belly hairs cut off for the experiment. Using distilled water, the application site (3.14 cm2) was cleansed. The experimental protocols were conducted in compliance with the CPCSEA's recommendations for animal care. They have been split into three groups:
Induction of Arthritis
A rat model of arthritis produced by 5% carrageenan/kaolin was created. For a limited period, 3% isoflurane in a N2O combination was used to put the animals to sleep. A single intraplantar injection of 3% carrageenan/kaolin suspended in 100 μL of pyrogen-free sterile saline was administered to the left knee joints to cause arthritis. Knee thickness was assessed to assess the arthritic progression in rats with arthritis produced by carrageenan/kaolin. After six days of monitoring, these are regarded as behavioral indications of arthritis caused by carrageenan or kaolin. Redness, swelling, and arthritic pain began to manifest as the arthritis worsened, peaking on the first day following the carrageenan/kaolin injection. Every test was conducted in a blinded manner [62].
Result
Male albino rats with artificially generated arthritis were used in this investigation to evaluate the ibuprofen transdermal patch in vivo. The left knee joint was injected intraplantarly with a kaolin and carrageenan suspension to simulate the subacute joint inflammation and edema that are typical of arthritic diseases. Three (n = 6) groups of animals were randomly assigned to each of the following: The Normal group (non-arthritic, untreated), the Standard group (arthritis-induced, treated with commercialized diclofenac patch), and the Treatment group (arthritis-induced, treated with the developed ibuprofen transdermal patch).
After being given kaolin and carrageenan, rats in the Standard and Treatment groups showed obvious symptoms of joint inflammation, including stiffness, redness, edema, and decreased mobility. At regular intervals, measurements were made of the joint diameter and paw thickness. The Treatment group saw a notable decrease in joint swelling 24 hours after the patch was applied, and this improvement persisted throughout the trial period. By day 7, there were noticeable anti-inflammatory benefits.
We tracked body weight as a measure of overall health. Rats in the Standard group were likely to maintain body weight than those in the Treatment group, which may indicate a decrease in systemic inflammation. Joint soreness and limited mobility.
These findings highlight the possibility of the developed ibuprofen transdermal patch as a non-invasive option for treating arthritis-related inflammation by confirming that it successfully reduced kaolin-carrageenan-induced arthritis symptoms and gave long-lasting anti-inflammatory action.
Conclusion
The current work used an in vivo model of arthritis caused by kaolin and carrageenan in male albino rats to assess the anti-arthritic effectiveness of a transdermal patch loaded with ibuprofen. When compared to the group receiving normal treatment, the results clearly showed that the transdermal patch effectively reduced inflammation and arthritic symptoms. Rats that were given kaolin and carrageenan to induce arthritis showed obvious symptoms of joint swelling, redness, and limited mobility, which closely resembled subacute arthritis-like diseases.
Rats given the ibuprofen transdermal patch demonstrated a marked decrease in paw edema and joint inflammation from the beginning of treatment. Similar to the usual medicine group, the treatment group showed a greater improvement in joint flexibility and a decrease in swelling. Furthermore, the therapy group maintained a lower body weight over the course of the trial, indicating less inflammatory stress and greater systemic health.
Bypassing the gastrointestinal issues linked to oral NSAIDs, a transdermal administration method had the extra benefit of preserving a localized and long-lasting anti-inflammatory action. The ibuprofen transdermal patch may be a viable substitute for treating arthritis-related inflammation, especially for localized or chronic joint problems, according to the in vivo results. To confirm its long-term safety, effectiveness, and potential for patient compliance, more research involving pharmacokinetic evaluation, chronic arthritis models, and clinical trials is advised. In conclusion, the developed ibuprofen transdermal patch showed notable therapeutic efficacy in treating rats' arthritis caused by kaolin and carrageenan, and it might be a good non-invasive alternative for managing arthritis.
- Ma L, Cranney A, Holroyd-Leduc JM et al. (2009) Acute monoarthritis: what is the cause of my patient's painful swollen joint.
- Reginato AM, Olsen BR (2002) The role of structural genes in the pathogenesis of osteoarthritic disorders. Arthritis Res.
- Siva C, Velazquez C, Mody A, Brasington R (2003) Diagnosing acute monoarthritis in adults: a practical approach for the family physician. Am Fam Physician.
- M Qindeel et al. (2020) Recent trends, challenges and future outlook of transdermal drug delivery systems for rheumatoid arthritis therapy J. Cont.
- M Bilal et al. (2021) Marine-Derived Biologically Active Compounds for the Potential Treatment of Rheumatoid Arthritis Marine Drugs.
- E Myasoedova et al. (2020) is the epidemiology of rheumatoid arthritis changing? Results from a population-based incidence study.
- J Bullock et al. (2019) Rheumatoid arthritis: a brief overview of the treatment.
- P Conigliaro et al. (2019) Challenges in the treatment of rheumatoid arthritis.
- Beck MA, Ferrada KA, Sikora AK, Ombrello JC, Collins W, Pei N, Balanda DL, Ross D, Cardona Z., B.
- Y. Cao et al. (2020) Pain Mechanism in Rheumatoid Arthritis: From Cytokines to Central Sensitization Mediators Inflammation.
- E.R. Vina et al. (2020) Nonsteroidal anti-inflammatory drug use in chronic arthritis pain: variations by ethnicity.
- M. Korani et al. (2020) The Effect of Aqueous Extract of Trachyspermum ammi Seeds and Ibuprofen on Inflammatory Gene Expression in the Cartilage Tissue of Rats with Collagen-Induced Arthritis.
- M. Statelova et al. (2020) Factors Affecting Successful Extrapolation of Ibuprofen Exposure from Adults to Pediatric Populations After Oral Administration of a Pediatric Aqueous Suspension.
- Krishnan Y, Grodzinsky AJ (2018) Cartilage diseases. Matrix Biol.
- Donahue, SWKrogh's (2018) principle for musculoskeletal physiology and pathology. J Musculoskelet Neuronal Interact.
- Shan F, Rashid, MI, Jamal SF, Ahmad I (2025) Environmental and Genetic Etiology of Rheumatoid Arthritis.
- Badwaik C, Nandgave M, (2022) Gout-a Review on Pathophysiology, Etiology, and Treament.
- Beutler BD, Chang C, Chang EY (2025) April. Septic Arthritis: Current Concepts. In Seminars in Musculoskeletal Radiology 29: 293-301.
- Justiz Vaillant AA, Goyal A, Varacallo MA (2023) StatPearls [Internet]. StatPearls Publishing; Treasure Island (FL): Systemic Lupus Erythematosus.
- Struglics A, Larsson S, Kumahashi N, Frobell R, Lohmander LS (2015) Changes in Cytokines and Aggrecan ARGS Neoepitope in Synovial Fluid and Serum and in C-Terminal Crosslinking Telopeptide of Type II Collagen and N-Terminal Crosslinking Telopeptide of Type I Collagen in Urine Over Five Years After Anterior Cruciate Ligament Rupture: An Exploratory Analysis in the Knee Anterior Cruciate Ligament, Nonsurgical Versus Surgical Treatment Trial. Arthritis Rheumatol.
- de Hair MJ, van de Sande MG, Ramwadhdoebe TH, Hansson M, Landewé R, et al. (2014) Features of the synovium of individuals at risk of developing rheumatoid arthritis: implications for understanding preclinical rheumatoid arthritis. Arthritis Rheumatol.
- Kleiber Balderrama C, Rosenthal AK, Lans D, Singh JA, Bartels CM (2017) Calcium Pyrophosphate Deposition Disease and Associated Medical Comorbidities: A National Cross-Sectional Study of US Veterans. Arthritis Care Res (Hoboken).
- Mathews CJ, Coakley G. (2008) Septic arthritis: current diagnostic and therapeutic algorithm. Curr Opin Rheumatol.
- Chen J, Zhang Y, Wang H (2023) Recent advances in ibuprofen formulations and delivery strategies: A comprehensive review. Drug Development and Industrial Pharmacy.
- Segal, Marian, Patches, Pumps and Timed Release: New Ways to Deliver Drugs, Food and Drug Administration. Retrieved on 2007.
- Berner B, John VA (2010) Pharmacokinetic characterization of Transdermal delivery systems, J Clin Pharmacokin.
- Godbey KJ (1996) Improving patient comfort with nonocclusive transdermal backings, American Association of Pharmaceutical Scientists.
- Pastore MN, Kalia YN, Horstmann M, Roberts MS (2015) Transdermal patches: History, development and pharmacology. Br. J. Pharmacol.
- Montia D, Saettone Mf, Giannaccini B, Angeli Dg (1995) Enhancement of Transdermal Penetration of Dapiprazole Through Hairless Mouse Skin. J Control Release.
- Bharadwaj S, Gupta Gd, Sharma Vk (2012) Topical Gel: A Novel Approach for Drug Delivery. J Chem. Bio. Phy. Sci.
- Aggarwal G. Development (2009) Fabrication and Evaluation of Transdermal Drug Delivery -A Review, Pharmainfo.net.
- Chien YW (1992) Novel drug delivery systems, Drugs and the Pharmaceutical Sciences: New York: Marcel Dekker.
- Zurdo SI, Franke P, Schaefer UF, Lehr CM (2007) Delivery of ethinyl estradiol from film forming polymeric solutions across human epidermis in vitro and in vivo in pigs. J Control Release.
- Babu RJ, Pandit JK (2005) Effect of permeation enhancers on the transdermal delivery of buprenolol through rat skin. Drug Delivery.
- Hanumanaik M, Patil U, Kumar G, Patel SK, Singh I, Jadatkar K (2012) Design, Evaluation and Recent Trends in Transdermal Drug Delivery System: A Review. IJPSR.
- Nidhi S (2018) A brief review on transdermal patches. Int J Org Med Chem.
- Thornfeldt CR (1998) Potent Penetration Enhancers. US Patent, No. 5760096.
- Thorat R, Mohalkar AV et al. (2024) Formulation and Evaluation of Transdermal Patch of Ibuprofen.
- Abrams GJ, Solomon L, Meyers OL (2016) A long-term study of diclofenac sodium in the treatment of rheumatoid arthritis and osteo-arthrosis. South African Medical Journal.
- Gowda DV, Rajesh N, Somashekhara CN, Siddaramaiah (2010) Development and evaluation of Aceclofenac loaded transdermal film. International Journal of Pharmtech Research 2.
- Soral et al. (2021) Formulation and Evaluation of Transdermal Patch of Rabeprazole Sodium.
- C. Tiwari et al. (2021) A Novel Approach for Transdermal Drug Delivery.
- P Kriplani et al. (2018) Formulation and evaluation of transdermal patch of diclofenac sodium.
- PK Yadav et al. (2017), Transdermal patch of an antihypertensive drug: its development and evaluation.
- M Akhlaq et al. (2016) Formulation and evaluation of anti-rheumatic dexibuprofen transdermal patches: a quality-by-design approach.
- P Ossowicz et al. (2022) Evaluation of the structural modification of ibuprofen on the penetration release of ibuprofen from a drug-in-adhesive matrix type transdermal patch.
- G Praveen, B Meenaksi et al. (2014) Development of ibuprofen nanoliposome for transdermal delivery: Physical characterization, in vitro/in vivo studies, and anti-inflammatory activity.









Tables at a glance
Figures at a glance