Solid Pseudo Papillary Neoplasm of the Pancreas in a 45-Year-Old Woman: A Case Report
Received Date: June 03, 2024 Accepted Date: July 03, 2024 Published Date: July 06, 2024
doi:10.17303/jspcr.2024.6.202
Citation: Dr. Vinay HG (2024) Solid Pseudo Papillary Neoplasm of the Pancreas in a 45-Year-Old Woman: A Case Report. J Surg Proce Case Rep 6: 1-9
Abstract
We present a distinctive case of solid pseudo papillary neoplasm as seen in a 45-year-old woman who presented with pain abdomen and abdominal mass. It was discovered that she possessed a significant mass covering the pancreatic body and tail, exhibiting typical physical and histological characteristics. Pancreatic mass appear variegated with solid grey-brown areas, necrosis and hemorrhagic areas noted. Also seen are cystic areas with necrosis and mucin with Calcification. She subsequently had a successful surgery, and she is currently in remission.
Keywords: Calcification; Solid Pseudo Papillary Neoplasm; Necrosis; Pancreas
Introduction
Frantz's tumor, also known as solid pseudo papillary neoplasm, is an uncommon condition that makes up 0.2–2.7 percent of all pancreatic tumors [1]. The majority of patients are female and in their second or third decade of life; very few involve children [2]. They are often discovered by accident, showing up as a slowly expanding abdominal mass with vague symptoms like soreness in the abdomen. With a prevalence of malignant transformation of around 15%, surgical excision is the preferred treatment for this low-grade malignant tumor, which offers a favorable long-term prognosis [3].
SPN is a rare primary pancreatic tumor that primarily affects young women. Law et al.'s analysis of the literature on SPN publications revealed information on 2744 patients during a 51-year span. According to this analysis, 88% of the patients were female, whereas tumors in men are uncommon. At the time of clinical presentation, the mean age was 28.5 years. Abdominal pain and moderate gastrointestinal symptoms are among the nonspecific clinical presentations that occur most frequently. Patients with no symptoms are typically identified by accident [3].
90% of SPN cases occur in adolescent and young adult females, indicating a strong female inclination [4]. Progesterone receptors and the predominance of SPN in young females at the start of the reproductive period suggest that female hormones play a role in the formation of this tumor [5].
SPN typically has a well-circumscribed, mixed cystic and solid appearance on radiological examination. Punctuate calcification may be linked to intracystic degeneration. However, severe eggshell calcification is a rare symptom of SPN. Due to tumor degradation and subsequent calcification, this condition affects older individuals. Primary cystic pancreatic neoplasms are included in the differential diagnosis of SPN. Of these, different degrees of calcification can be seen in pseudocyst, oligocystic serous cystadenoma, and mucinous cystic neoplasm (MCN). While serous cystadenoma calcification is centrally localized, MCN may have peripheral linear calcification [6].
We discuss the case of a 45-year-old woman who presented with an abdominal mass and was subsequently diagnosed with SPN.
Case Report
A 45-year-old woman with symptomatology such as lump in the abdomen and abdominal pain. With no significant past medical history and otherwise in good health. On physical examination, vague mass occupying the left lumbar and epigastrium, with tenderness was present.
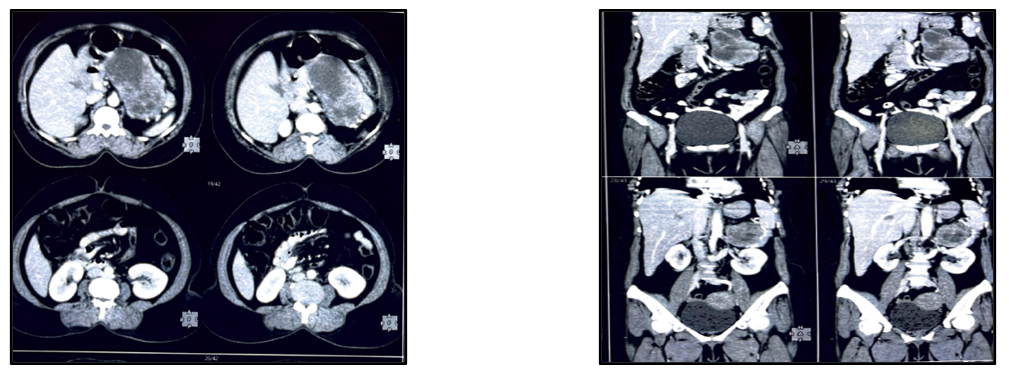
Computerized tomography scan Figure 1. demonstrated large heterogeneously enhancing solid and cystic lesion measuring 12.0x8.0x6.0cms arising from the distal body and tail of pancreas showing coarse central calcifications. It is causing mass effect on the left adrenal gland. Splenic vessels are displaced and coursing along the inferior margin of the lesion. No evidence of pancreatic duct dilation. The lesion shows heterogeneous enhancement of post contrast study -Neoplastic etiology.
PET CT Scan was done showed heterogeneously enhancing solid and cystic lesion arising from the distal body and tail of pancreas showing coarse central calcifications causing mass effect on the left adrenal gland of neoplastic etiology, likely representing pancreatic cystadenocarcinoma. No significant pancreatic lymph nodes were seen, No any evidence of hepatic, pulmonary or skeletal metastases.
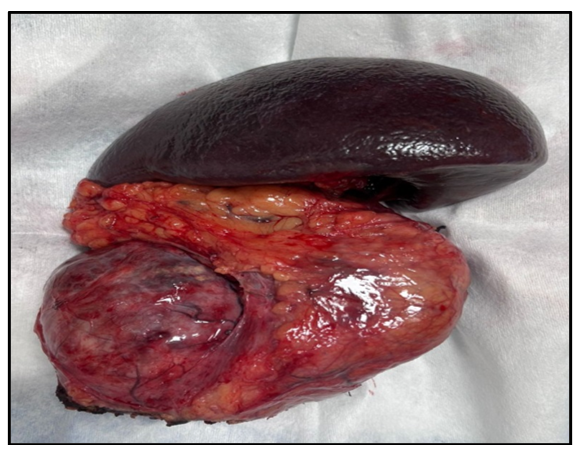
Figure2. presented the excised specimen. Findings on gross examination shows large heterogeneous lesion seen in left hypochondrium. Well defined soft tissue density lesion measuring 12.0x8.0x6.0cms noted, involving the distal body and tail of pancreas. Few calcifications noted within it. Pancreatic mass showed appears variegated with solid grey-brown areas, necrosis and hemorrhagic areas noted. Also seen are cystic areas with necrosis and mucin with Calcification. Surrounding pancreatic tissue noted. Lymph nodes isolated at the hilum of spleen, largest measuring 0.5cm, showed no tumor deposit.
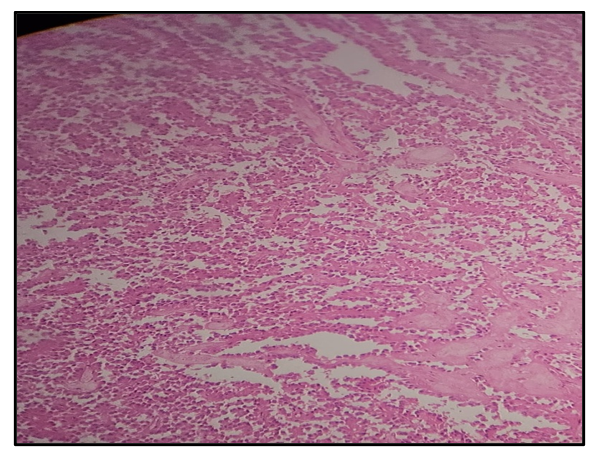
On microscopic examination, multiple sections studied shows pancreatic tissue with tumors exhibiting heterogeneous, variable admixture of solid and pseudo papillary areas. The solid areas are comprised of uniform cells admixed with capillary sized blood vessels. Pseudo papillae areas shows tumor cells detached from blood vessels forming fibro vascular stalks or rosette-like structures. Stroma shows various degrees of hyalinization, degeneration, such as hemorrhage, foamy macrophages, calcification and cholesterol clefts. Figure 3, the tumor cells have a moderate amount of eosinophilic cytoplasm and perinuclear vacuoles. Relatively uniform nuclei with fine chromatin, inconspicuous nucleoli and longitudinal grooves with occasional mitosis seen. Tumor is seen infiltrating into the surrounding pancreatic tissue. Microscopic section from pancreatic margin is free of tumor, section from spleen shows congestion, section from the vessel appears unremarkable and 6 lymph nodes with reactive change seen.
The patient was discharged after five days in a satisfactory condition with advice and post treatment. The post-operative recovery was significant. The patient has been followed up for over 1-month without any complication.
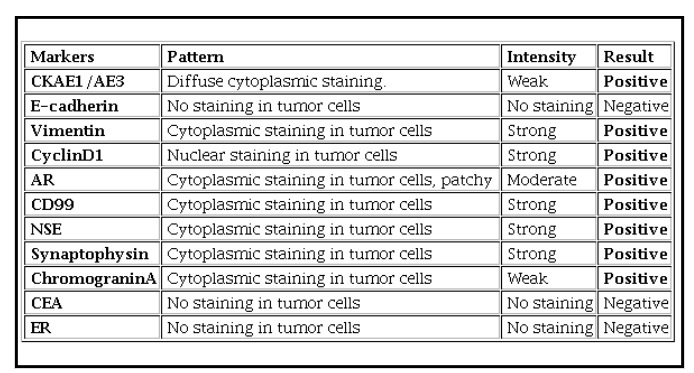
IHC was performed by Manual HIER on the formalin fixed paraffin block (Figure 4). Immunohistochemistry favors Solid pseudo papillary neoplasm.
Discussion
SPN is an uncommon pancreatic exocrine tumor with a low risk of cancer and a fair prognosis [7], which recurs infrequently after removal [8,9]. Also referred to as Frantz's tumor, it was first identified as a papillary-cystic pancreatic tumor in a 2-year-old male patient by Dr. Virginia Kneeland Frantz in 1959 [10]. In 1996, the WHO classified it. Despite being uncommon, an increasing number of instances are becoming public due to breakthroughs in imaging techniques.
Research conducted by Law et al. [3] revealed 2744 patients with SPN, of which over 87% were reported after 2000. This represents a seven-fold increase in cases recorded between 2000 and 2012 compared to the period from 1961 to 1999, with men accounting for 12.2% of the cases. This indicates greater knowledge and improved diagnostic tools rather than necessarily a rise in occurrence.
In the English literature at the time, 718 reported instances of SPN were examined in the review by Papavramidis [11]. The tumor's entire range was found to be between 0.5 and 34.5 cm, with a mean diameter of about 6.08 cm. They also emphasized the tumor's preference for female gender, with a female to male ratio of 9.78: 1. The pancreatic body and tail continue to be the most frequent sites [12]. These tumors have numerous microscopic blood arteries that are not maintained and start off as solid masses, which leads to the formation of the pseudo papillary pattern [13]. The cells closest to the vessels remain intact, resulting in characteristic cystic alterations and creation of the classic pseudo papillary pattern associated with the tumor, while the cells furthest from the vessels incur necrosis owing to nutrient deprivation. Therefore, cystic alterations are uncommon in SPN and are typically observed in larger masses that have outgrown their blood supply and experienced necrosis and degenerative changes [14].
Beltrame et al. [15] conducted a single-institution study on 451 patients with pancreatic cystic tumors. Of these, 18 (3.7%) had pancreatic SPN based on histological analysis, and only one patient had an elevated serum CA 19-9 level of 92 U/ml (normal range: 0 to 37 U/ml).
The majority of cancers are discovered by accident during imaging. Surgical tumor enucleation or excision is nearly invariably curative [16]. Although metastatic disease is uncommon, its presence does not preclude surgical excision [17]. Long-term disease-free survival is nevertheless compatible with resection of metastases, either in addition to or instead of resection of the main. The liver is the most often metastasized location [18].
An analysis of 16 male SPN patients revealed that, compared to female patients, the male patients were older and had a better prognosis following surgery, with no deaths or recurrences during the follow-up period. By comparison, a recent study conducted by Wu et al [19].
Even when there is local invasion or distant metastasis, the cornerstone of treatment for every patient with SPN is complete surgical resection. Over 95% of patients survive for at least five years. Aggressive early therapy provides great long-term survival even in cases of severe disease [20].
Chen J et al studies retrospectively of 486 patients with SPNs who were diagnosed. A total of 21 patients experienced recurrence or metastasis postoperatively. Tumor size, lymph vascular invasion, and the Ki-67 index were independent predictors of relapse, Risk factors included 3 parameters: tumor size (>9 cm), lymph vascular invasion status (presence), and Ki-67 index (>1%). SPNs are low-grade malignant neoplasm’s that rarely metastasize, and the 3 selected pathologic parameters can be used to predict their behavior [21].
Sun G et al showed cases of 118 patients with SPN. The mean tumor size was 5.9 (standard deviation, 2.9) cm. Fourteen patients (11.9%) presented aggressive pathologic behavior, which was correlated to the incomplete tumor capsule. At a median follow-up of 59.2 months, the recurrence rate was 1.8% and the overall 5-year survival rate was 97.7%. This study also concludes that Solid pseudo papillary neoplasm of the pancreas is a potentially low-grade malignant tumor that most frequently found in young females [22].
Guo J study revealeda 45-year-old female presented with a right upper abdominal mass and abdominal pain for 3 and 1 months as chief complaints. It’s interesting to note that the tumor invaded both the right wall of the inferior vena cava and the right side of the liver, with its origins revealed to be in the retro peritoneum. The pathology results verified the tumor's identification as an extra pancreatic SPN, and the procedure went smoothly. After a follow-up of 15 months, the patient was still asymptomatic. The recommended course of action for extra pancreatic SPN patients is still surgery. The preoperative misdiagnosis emphasizes the significance of precise diagnosis and the creation of suitable treatment plans for hepatic tumors [23].
This rare tumor, which accounts for 0.3% to 2.7% of all pancreatic neoplasm’s, has an unclear histogenesis and goes by various names. It is also referred to as a Frantz tumor, after the person who originally characterized it [24]. The incidence rate that is more common among young women is highlighted by the author.
Even in pancreatic disease referral institutions with significant patient volumes, available data on the behavior and prognosis of this tumor are limited, and published experiences are based on single case reports or a small number of patients [25]. However, no particular risk factors or connections to functional endocrine disorders have been found. SPN exhibits a unimodal behavior in men at 64 years old and a bimodal incidence in women with two peaks at 28 and 64 years old [26].
Following are some Literature base on the SPN literature, Table 1.
Conclusion
SPN is an uncommon pancreatic tumor that primarily affects young girls and has a very good prognosis. It is yet unclear what causes the tumor and how women are more susceptible to it, therefore more research is necessary to fully understand the mechanisms involved.
Summary
Data Availability
- Presented a distinctive case of solid pseudo papillary neoplasm as seen in a 45-year-old woman.
- Pancreatic mass shows appear variegated with solid grey-brown areas, necrosis and hemorrhagic areas noted.
- Spleen measuring 8.0x7.0x5.0cms, the mass appears congested.
- Large heterogeneous lesion was seen in left hypochondrium. Well defined soft tissue density lesion measuring 12.0x8.0x6.0cmsnoted, involving the distal body and tail of pancreas
- Patient was shifted to MICU started on IVF, IV antibiotics and other supportive treatment.1 pint PRBC was transfusion done
- This studied shows pancreatic tissue with tumors exhibiting heterogeneous, variable admixture of solid and pseudo papillary areas.
- The patient was discharged after five days in a satisfactory condition with advice and post treatment.
Data is available on the patient’s medical records.
Conflicts of Interest
The authors declare that there is no conflict of interest regarding the publication of this article.
- Z Słowik-Moczydłowska, M Gogolewski, S Yaqoub, et al. (2015) Solidpseudopapillary tumor of the pancreas (Frantz’s tumor): two case reports and a review of the literature, J. Med. Case Rep. 9: 268.
- N Guo, QB Zhou, RF Chen, et al. (2011) Diagnosis and surgical treatment of solidpseudopapillary neoplasm of the pancreas: analysis of 24 cases, Can. J. Surg. 54: 368-74.
- JK Law, A Ahmed, VK Singh, et al. (2014) A systematic review ofsolid-pseudopapillary neoplasms: are these rare lesions? Pancreas, 43: 331-7.
- Papavramidis T, Papavramidis S (2005) Solid pseudopapillary tumors of the pancreas: review of 718 patients reported in English literature. J Am Coll Surg. 200: 965-72.
- Kosmahl M, Seada LS, Janig U, Harms D, Kloppel G (2000) Solid-pseudopapillary tumor of the pancreas: its origin revisited. Virchows Arch. 436: 473-80.
- Kawamoto S, Scudiere J, Hruban RH, Wolfgang CL, Cameron JL, Fishman EK (2011) Solid-pseudopapillary neoplasm of the pancreas: spectrum of findings on multidetector CT. Clin Imaging, 35: 21-8.
- JS Estrella, L Li, A Rashid et al. (2014) “Solid pseudopapillary neoplasm of the pancreas,” The American Journal of Surgical Pathology, 38: 147-57.
- CM Vollmer Jr, E Dixon, DR Grant (2003) “Management of a solid pseudopapillary tumor of the pancreas with liver metastases,” HPB, 5: 264-7.
- CM Kang, SH Choi, SC Kim et al. (2014) “Predicting recurrence of pancreatic solid pseudopapillary tumors after surgical resection,” Annals of Surgery, 260: 348-55.
- JR Carlotto, FR Torrez, AM Gonzalez et al. (2016) “Solid pseudopapillary neoplasm of the pancreas,” Arquivos Brasileiros de Cirurgia Digestiva, 29: 93-6.
- JK Law, A Ahmed, VK Singh et al. (2014) “A systematic review of solid-pseudopapillary neoplasms,” Pancreas, 43: 331-7.
- T Papavramidis, S Papavramidis (2005) “Solid pseudopapillary tumors of the pancreas: review of 718 patients reported in English literature,” Journal of the American College of Surgeons, 200: 965-72.
- SE Lee, JY Jang, DW Hwang, KW Park, SW Kim (2008) “Clinical features and outcome of solid pseudopapillary neoplasm,” Archives of Surgery, 143: 1218-21.
- BA Liu, ZM Li, ZS Su, XL She (2010) “Pathological differential diagnosis of solid-pseudopapillary neoplasm and endocrine tumors of the pancreas,” World Journal of Gastroenterology, 16: 1025-30.
- S Sunkara, TR Williams, DT Myers, ON Kryvenko (2012) “Solid pseudopapillary tumours of the pancreas: spectrum of imaging findings with histopathological correlation,” The British Journal of Radiology, 85: e1140-4.
- V Beltrame, G Pozza, E Dalla Bona, A Fantin, M Valmasoni, C Sperti (2016) “Solid-pseudopapillary tumor of the pancreas: a single center experience,” Gastroenterology Research and Practice, 2016: 4289736-7.
- T Guo, L Wang, P Xie, Z Zhang, Y Yu (2020) “Diagnosis and surgical treatment and pathological findings of solid pseudopapillary tumor of the pancreas: a single-institution experience,” Cancer Management and Research, 12: 581-8.
- JCU Coelho, MAR da Costa, EJB Ramos, AR Torres, MC Savio, CMP Claus (2018) “Surgical management of solid pseudopapillary tumor of the pancreas,” Journal of the Society of Laparoendoscopic Surgeons, 22: 4.
- H Zhang, TB Liang, WL Wang, Y Shen, GP Ren, SS Zheng (2006) “Diagnosis and treatment of solid-pseudopapillary tumor of the pancreas,” Hepatobiliary & Pancreatic Diseases International, 5: 454-8.
- J Wu, Y Mao, Y Jiang et al. (2020) “Sex differences in solid pseudopapillary neoplasm of the pancreas: a population-based study,” Cancer Medicine, 9: 6030-41.
- Igbinosa O (2011) Pseudopapillary tumor of the pancreas.An algorithmic approach. JOP, 12: 262-5.
- Chen J, Zong L, Wang P, Liu Y, Zhang H, et al. (2023) Solid pseudopapillary neoplasms of the pancreas: clinicopathologic analysis and a predictive model. Modern Pathology, 36: 100141.
- Sun G, Fang K, Fu X, Peng L, Shu J, Tu Y, Li Y, Xiao W (2023) Solid Pseudopapillary Neoplasm of the Pancreas: A Multi-Institution Study of 118 Cases. Pancreas, 52: e121-6.
- Guo J, Zhao Q, Qin L, Xie S, Lu S, et al. (2024) Massive extrapancreatic solid pseudopapillary neoplasm misdiagnosed as hepatic tumor: a case report and literature review. Frontiers in Oncology, 14: 1342400.
- Chen H, Huang Y, Yang N, Yan W, Yang R, et al. (2021) Solid-Pseudopapillary Neoplasm of the Pancreas: A 63-Case Analysis of Clinicopathologic and Immunohistochemical Features and Risk Factors of Malignancy. Cancer Manag Res. 13: 3335-43.
- Torres OJM, Rezende MB, Waechter FL, Neiva RF, Moraes-Junior JMA, et al. (2019) Pancreatoduodenectomy for solid pseudopapillary tumor of the pancreas: A multi-institution study. Arq Bras Cir Dig. 32: e1442.
- Flores RL, Rossi R, Castiblanco A, Gallardo A, Schiappacasse G (2021) Solid bifocal pseudopapillary neoplasm of the pancreas: A case report. Int J Surg Case Rep. 84: 106131.
- Deniz K, Arıkan TB, Başkol M, Karahan Öİ (2021) Solid Pseudopapillary Neoplasm of the Pancreas. Journal of Gastrointestinal Surgery. 25: 322-4.
- Singh S, Zhao Q, Sachs TE, Hartshorn K (2023) Reporting a Case of Solid Pseudopapillary Neoplasm of the Pancreas in a 44-Year-Old Woman with Parallel Analysis of Literature. Case Reports in Oncological Medicine, 2023: 1768926.
- Sandlas G, Tiwari C (2017) Solid pseudopapillary tumor of pancreas: A case report and review of literature. Indian J Med Paediatr Oncol, 38: 207-9.

Tables at a glance
Figures at a glance