Clinical Features of Hemophilia with Cerebral Hemorrhage
Received Date: January 03, 2025 Accepted Date: February 03, 2025 Published Date: February 06, 2025
doi:10.17303/jspcr.2025.7.101
Citation: Jianting Lin,Kuai Yang, Penglei Zhu, Buyi Zheng (2025) Clinical Features of Hemophilia with Cerebral Hemorrhage. J Surg Proce Case Rep 7: 1-8
Abstract
Background: Intracranial hemorrhage, the most serious complications of hemophilia, is a common disease of nervous system. Reports of hemophilia with cerebral hemorrhage have increased in recent years. However, the clinical features and pathogenesis of hemophilia with cerebral hemorrhage patients are unclear.
Methods: From January 2007 to August 2019, patients of hemophilia with cerebral hemorrhage in the Wenzhou People’s hospital were selected. The clinical manifestations, laboratory measurements, and neurological images of these patients were collected and analyzed retrospectively.
Result: There are 21 patients recorded in this research. All of these patients are male and the average age of these 21 patients was 27.8 years old. Among these patients, conventional vascular risk factors were identified in 3 patients. Coagulation was observed in 18 patients, and increased D-dimer level was recorded in 16 patients. The hemorrhage developed in the brain lobe solely (n=5), ventricle (n=2), basal ganglia region (n=2), subdural hemorrhage (n=4), subarachnoid hemorrhage (n=3), brainstem (n=1), and combination of multiple hemorrhage (n=4). The average time from diagnosis of hemophilia to the occurrence of cerebral hemorrhage was 18.6 years.
Conclusion: Most hemophilia patients with cerebral hemorrhage were male, which could occur anywhere in the skull. The clinical presentation including headache, coma, fatigue, vomiting, dizziness and epilepsy. Coagulation is usually normal or mildly impaired. Transfusion of coagulation factors could help improve prognosis.
Keywords: Hemophilia; Cerebral Hemorrhage; Clinical Feature; Skull; Bleeding Disorders
Introduction
Hemophilia A and B are X-linked recessive bleeding disorders, associated with mutations affecting the factor VIII or factor IX gene located on the X chromosome; it is classified into type A (factor VIII) and type B (factor IX) [1]. Being linked to the X chromosome, the incidence is higher in men, and women are carriers without developing the disease [2]. The clinical phenotype is dominated by the residual endogenous factor level, resulting in severe (<1%), moderate (1% to 5%), and mild (5% to 50 %) forms [3]. Although joint and muscle bleeds are the most common symptoms of hemophilia, intracranial hemorrhage (ICH) can be considered the most severe complication. Intracranial bleeds may form serious life-threatening events and have been associated with considerable neurological sequelae in survivors [4-6]. The incidence of ICH is 10-20 fold higher in hemophilia patients than in the general population and the risk factors are head trauma, thrombocytopenia, hypertension, previous ICH, or nonsteroidal anti-inflammatory drugs (NSAID) use [7,8]. Due to non-specific initial symptoms of headache and vomiting, the diagnosis of ICH may often be delayed. This paper aims to reveal the clinical characteristics of hemophilia patients with intracerebral hemorrhage, so as to guide the clinical work.
Methods
General Information
All patients were selected from January 2007 to August 2019 in the Wenzhou People’s hospital, which clinically diagnosed of hemophilia and developed intracranial hemorrhage in final. The clinical manifestations, laboratory measurements, and neurological images of these patients were collected and analyzed retrospectively.
Diagnostic Criteria
The diagnostic criteria of Hemophilia was adapted from Chinese guidelines on the treatment of hemophilia (version 2020) [9].The diagnosis of cerebral hemorrhage was made based on the guidelines for the diagnosis and treatment of spontaneous intracerebral hemorrhage released by the American Heart Association in 2022 [10]. Coagulopathy was diagnosed when any of the following situations occurred: prothrombin time (PT) >15 seconds, activated partial thromboplastin time (APTT) >45 seconds, international normalized ratio (INR) >1.5,D-dimer >0.5mg/L [11].
Inclusion Criteria
Hemophilia patients with cerebral hemorrhage were identified by a panel of hematologist, neuroimaging experts, and neurologists. Briefly, patients in any of the following situations were included into the study: (1) patients who were admitted to the Wenzhou People’s hospital due to acute cerebral hemorrhage and had a history of Hemophilia; (2) patients who were admitted to the hospital presented headache or epilepsy (e.g.fatigue, coma), and the cerebral hemorrhage was confirmed by brain computed tomography (CT) or magnetic resonance imaging (MRI) scan.
Exclusion Criteria
Patients in any of the situations below were excluded from the study: (1) patients who had serious systemic diseases, such as other hematologic disease or cancer; (2) patients with central nervous system complications such as intracranial venous system thrombosis, cerebral infarction, or traumatic brain injury; (3) cases with intracranial aneurysm or intracranial vascular malformation confirmed by brain DSA, magnetic resonance angiography (MRA), and computed tomography angiography (CTA) examinations; and (4) patients with incomplete medical records.
Collection of Clinical Data
General demographic characteristics such as gender and age were recorded. Previous history such as hypertension, diabetes were collected. The main symptoms, consciousness levels, hemorrhagic locations, and treatment methods of the cerebral hemorrhage were also collected.
The hematoma volume was calculated by much field formula, the level of consciousness was evaluated according to the Glasgow Coma Scale (GCS). The functional outcome of patients on the day of discharge was evaluated based on the Glasgow Outcome Scale (GOS). The main collection of laboratory analysis was coagulation indexes, include international normalized ratio (INR); prothrombin time(PT) ; activated partial thromboplastin time (APTT); plasma D-dimer. Brain CT, CTA, brain MRI, MRA, and total brain DSA.
All subjects and/or their parents gave informed consent to participate in this study, which was approved by the Medical Research Ethics Committee of Wenzhou People's Hospital.
Results
21 patients were included in this study. All of these patients were male and the average age of them was 27.8 years old (range: 1-85 years old; Table 1). Hemophilia a patients were the main type in this study, accounting for 95.2%. And hemophilia B had a mere 5.2%. The average time from diagnosis of hemophilia to the occurrence of cerebral hemorrhage was 18.6 years. According to the laboratory tests, coagulopathy was recorded in 18 (85.7%) patients, high D-dimer level was detected in 16 (76.2%) patients. According to factor VIII level, 4 (19%) patients were identified as severe hemophilia and 17 (81%) patients were regarded as mild hemophilia. 3 patients suffered from hypertension and 2 patients suffered from diabetes before cerebral hemorrhage (Table 1).
As shown in Figure 1, during the acute onset of cerebral hemorrhage, hemorrhage developed in the brain lobe solely (n=5), basal ganglia (n=2), ventricle (n=2), subdural (n=4), subarachnoid hemorrhage (n=3), brainstem (n=1). The most common symptoms were headache, which occurred in 12 (57.1%) patients, and the other symptoms were coma in 4 (19%), epilepsy in 1 (4.8%), fatigue in 2 (9.5%), vomiting in 2 (9.5%), and dizziness in 1 (4.8%) patient. Only 2 patients accepted surgery, among which, and one patient chose automatic discharge final while the other recovered well and discharged successfully. 4 (19%) patients developed cerebral hemorrhage in multiple parts, which caused poor prognosis. The average hematoma volume was 17.8ml (range:4-80ml; Table 2). It was worth to mention that GCS score in most patients were not below 8. At the same time, GOS score in most patients were 4 and above (Table 2).
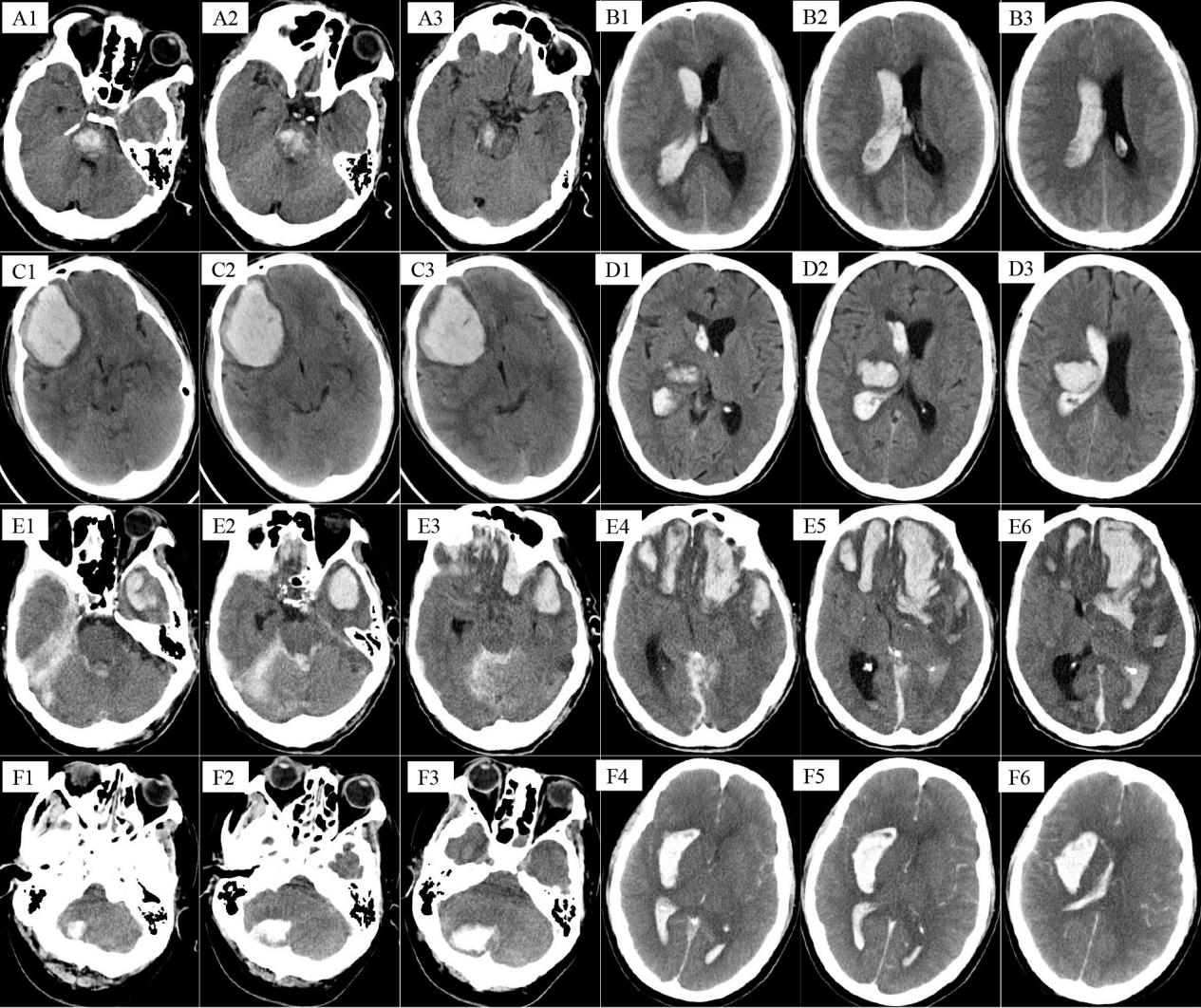
A1-A3 were obtained from a 31-years-old male who had no traditional vascular risk factors, but developed cerebral hemorrhage (a lesion in the brainstem) at 20 years after diagnosis of hemophilia. B1-B3 were obtain from a 50-years-old male who had no traditional vascular risk factors, but developed cerebral hemorrhage (a lesion in the right paracele) at 10 years after diagnosis of hemophilia. C1-C3 were obtain from a 34-years-old male who had no traditional vascular risk factors, but developed cerebral hemorrhage (a lesion in the right frontal lobe) at 26 years after diagnosis of hemophilia. D1-D3 were obtain from a 59-years-old male who had no traditional vascular risk factors, but developed cerebral hemorrhage (a lesion in the right basal ganglia) at 35 years after diagnosis of hemophilia. E1-E6 was obtained from an 85-years-old male who had hypertension and diabetes for more than 20 years. There was a history of head trauma which caused cerebral hemorrhage in bilateral frontal lobe, left temporal lobe, ventricle and subdural. F1-F6 was obtained from a 41-years-old male who had hypertension for more than 10 years and developed a lesion in the right cerebellum, right basal ganglia and ventricle.
Discussion
In the literature there is very limited clinical characteristic information about hemophilia with cerebral hemorrhage. As a rare disorder, the global prevalence of hemophilia A is about 1 in 5000 male births, and 1 in 30,000 male births for hemophilia B. These figures are similar worldwide, regardless of ancestry or ethnicity [2]. Hemarthrosis and bleeding at the muscular level are one of the main clinical features, which will be the first clinical symptom usually [12]. Cerebral hemorrhage was an uncommon but severe complication of hemophilia [13]. A literature review concluded that 3%-10% of patients with hemophilia experience an intracranial bleeding during life [14]. Due to the low incidence of hemophilia and the initial characteristic were not typical, intracranial hemorrhage could not be detected in the first time. In the present study, we analyzed the main clinical symptoms of 21 patients. Headache, vomiting, fatigue and other non-specific symptoms accounted for the main part, reached 81%. This made intracranial bleeding more difficult to recognize and delay diagnosis and treatment in final. Otherwise, the diagnosis of hemophilia depended mainly on the level of FVIII and FIX. Laboratory tests usually showed normal platelet count, normal prothrombin time (PT) full form, and prolonged activated partial thromboplastin time (APTT) full form [15]. Sometimes, activated partial thromboplastin time full form could be a normal high value or slight prolonged, which leaded to clinical neglect of the diagnosis of hemophilia. In this study, 19% patients manifested a normal activated partial thromboplastin time and 28.6% slightly prolonged. This could be difficult for clinicians, especially younger doctors, to associate with hemophilia.
According to previous reports, the most common sites of intracranial hemorrhage were subdural and brain lobes [7]. Just as it reported, 9 (42.9%) patients in this study occurred hemorrhage at these sites. But it is worthy to notice that 7 (33.3%) patients developed subarachnoid hemorrhage and multiple hematoma in the study, which also accounted for a considerable part. It suggested that clinicians should considered the possibility of hemophilia in various sites of cerebral hemorrhage, especially when clotting function in the normal range or slightly impaired for treatment. Neurosurgery in hemophiliac patients is challenging due to the increased risk of perioperative bleeding. The various steps to prevent this complication include perioperative clotting factor replacement, maintenance of hemodynamics, and special care to achieve meticulous hemostasis during the intraoperative period [16]. It is critical to replenish clotting factor both pre-operatively and post-operatively to optimize the coagulation profile and reduce procedure-related complications including bleeding [17]. 2 patients in this study accepted surgery. Before the operation, FVIII and frozen plasma were transfused under the guidance of professional hematologists. One patient (patient B) was discharged voluntarily due to a cerebral infraction final, because he was in a deep coma with dilated pupils when he came in hospital. Another patient (patient C) who underwent surgery made a good recovery and was eventually discharged from the hospital with no significant effect on muscle strength and conscious. There was a literature suggested that factor VIII activity levels decreased precipitously following surgery when without corrective infusions [18]. This underscores the need to continue treatment with factor VIII in both the acute and subacute postoperative periods [18,19]. In cases where factor levels are not available, an initial dose of 50 units/kg of factor VIII should be given for hemophilia A or 100-120 units/kg of factor IX for hemophilia B [20]. For our 2 patients above, after continuous postoperative transfusion of coagulation factors, both maintained a relatively normal level of clotting factors and no further bleeding occurred after the operation. However, due to the complexity of cerebral hemorrhage treatment, more factors need to be taken into account, like intracranial pressure (ICP). It is important to prevent ICP elevations during the postoperative period, preferably guided by monitoring devices, as raised ICP is one of the important predictors of mortality [21]. But because of the emergency condition and lack of relevant experience at that time, intracranial pressure monitoring was not carried out, which was our shortcoming. In addition, on account of the limitation of case number, characteristic of this summery could only be a reference. More cases need to be collect to make the results more reliable.
Conclusions
In summary, hemophilia with cerebral hemorrhage were overwhelmingly male and could occur anywhere in the skull. Coagulation is usually normal or mild impaired. The most common clinical presentation is headache, follow by coma, fatigue, vomiting, dizziness and epilepsy were rare. According to GCS scores and GOS scores, most patients had a good prognosis. For patients requiring surgery, transfusion of coagulation factors preoperative and postoperative was important to reduce postoperative rehemorrhage. Because of the limitation of case number, more clinical feature and better treatment options need to be found in a large number of clinical cases.
- Fijnvandraat K, Cnossen M, Leebeek F, Peters M (2012) Diagnosis and management of haemophilia. BMJ (Clinical research ed). 344: e2707.
- Ramos-Petersen L, Rodríguez-Sánchez J, Cortés-Martín J, Reinoso-Cobo A, Sánchez-García J, Rodríguez-Blanque R, et al. (2023) A Qualitative Study Exploring the Experiences and Perceptions of Patients with Hemophilia Regarding Their Health-Related Well-Being, in Salamanca. Journal of clinical medicine. 12.
- Moreira A, Das H (2018) Acute Life-Threatening Hemorrhage in Neonates with Severe Hemophilia A: A Report of 3 Cases. Journal of investigative medicine high impact case reports. 6: 2324709618800349.
- de Tezanos Pinto M, Fernandez J, Perez Bianco P (1992) Update of 156 episodes of central nervous system bleeding in hemophiliacs. Haemostasis. 22: 259-67.
- Andersson N, Auerswald G, Barnes C, Carcao M, Dunn A, Fijnvandraat K, et al. (2017) Intracranial haemorrhage in children and adolescents with severe haemophilia A or B - the impact of prophylactic treatment. British journal of haematology. 179: 298-307.
- Andersson N, Wu R, Carcao M, Claeyssens-Donadel S, Kobelt R, Liesner R, et al. (2020) Long-term follow-up of neonatal intracranial haemorrhage in children with severe haemophilia. British journal of haematology. 190: e101-4.
- Stieltjes N, Calvez T, Demiguel V, Torchet M, Briquel M, Fressinaud E, et al. (2005) Intracranial haemorrhages in French haemophilia patients (1991-2001): clinical presentation, management and prognosis factors for death. Haemophilia : the official journal of the World Federation of Hemophilia. 11: 452-8.
- Chopra P, Singh M, Singh A, Masi A, Yurkofsky J, Zaita B, et al. (2023) Perioperative Management of Spontaneous Intracranial Hemorrhage in a Patient With Hemophilia A in a Resource Limited Country. Cureus. 15: e43485.
- Chinese guidelines on the treatment of hemophilia (2020). Zhonghua xue ye xue za zhi = Zhonghua xueyexue zazhi. 41: 265-71.
- Greenberg S, Ziai W, Cordonnier C, Dowlatshahi D, Francis B, Goldstein J, et al. (2022) Guideline for the Management of Patients With Spontaneous Intracerebral Hemorrhage: A Guideline From the American Heart Association/American Stroke Association. Stroke. 53: e282-361.
- Yang M, Pan X, Liang Z, Huang X, Duan M, Cai H, et al. (2019) Clinical Features of Nephrotic Syndrome with Cerebral Hemorrhage. Medical science monitor : international medical journal of experimental and clinical research. 25: 2179-85.
- Roosendaal G, Lafeber F (2006) Pathogenesis of haemophilic arthropathy. Haemophilia : the official journal of the World Federation of Hemophilia. 117-21.
- Nagel K, Pai M, Paes B, Chan A (2013) Diagnosis and treatment of intracranial hemorrhage in children with hemophilia. Blood coagulation & fibrinolysis : an international journal in haemostasis and thrombosis. 24: 23-7.
- Ljung R (2008) Intracranial haemorrhage in haemophilia A and B. British journal of haematology. 140: 378-84.
- Mannucci P, Tuddenham E (2001) The hemophilias--from royal genes to gene therapy. The New England journal of medicine. 344: 1773-9.
- Gyanesh P, Dhiraaj S (2013) Anesthetic management of a patient with hemophilia A with spontaneous acute subdural hematoma. Journal of anaesthesiology, clinical pharmacology. 29: 117-20.
- Hegde A, Nair R, Upadhyaya S (2016) Spontaneous intracerebral hemorrhage in hemophiliacs-A treatment dilemma. International journal of surgery case reports. 29: 17-9.
- Sethi M, Gurha P (2017) Perioperative management of a patient with haemophilia-A for major abdominal surgery. Indian journal of anaesthesia. 61: 354-5.
- Srivastava A, Santagostino E, Dougall A, Kitchen S, Sutherland M, Pipe S, et al. (2020) WFH Guidelines for the Management of Hemophilia, 3rd edition. Haemophilia : the official journal of the World Federation of Hemophilia. 1-158.
- Peyvandi F, Garagiola I, Young G (2016) The past and future of haemophilia: diagnosis, treatments, and its complications. Lancet (London, England). 388: 187-97.
- Gurer B, Kertmen H, Yilmaz E, Dolgun H, Hasturk A, Sekerci Z (2017) The Surgical Outcome of Traumatic Extraaxial Hematomas Causing Brain Herniation. Turkish neurosurgery. 27: 37-52.

Tables at a glance
Figures at a glance